By Kat Fu, M.S., M.S. / May 1, 2026
If your bloodwork came back showing elevated CRP and your doctor cannot fully explain it, your sleep may be a factor the lab requisition did not account for. CRP is one of the most widely used inflammatory biomarkers, and research now shows it is associated with disturbed sleep in population studies and can shift after severe sleep loss in some controlled studies.
This article covers how poor sleep can raise inflammatory markers, how inflammation may interact with sleep in return, the sex differences in the CRP-sleep relationship, and what lowers sleep-related inflammatory risk. It does not cover acute-phase CRP elevations from infection or injury. For the broader framework, see: Inflammatory Sleep Disruption.
How Quickly Does Poor Sleep Raise CRP?
The threshold is relevant, but not fully settled. A 2016 meta-analysis of more than 70 studies found that sleep disturbance was associated with elevated CRP and IL-6. However, the same meta-analysis did not find a reliable pooled association between experimental sleep deprivation or sleep restriction and CRP, IL-6, or TNF-alpha.
The dose that mirrors everyday life: sleeping 6 hours instead of 8 hours across 7 nights raised IL-6 in both sexes and TNF-alpha in men. A separate partial-sleep-restriction study using 4 hours per night for 10 days found IL-6 elevation, while CRP showed a nonsignificant between-group trend. Another small laboratory study using about 4 hours per night for 10 days reported hs-CRP elevation.
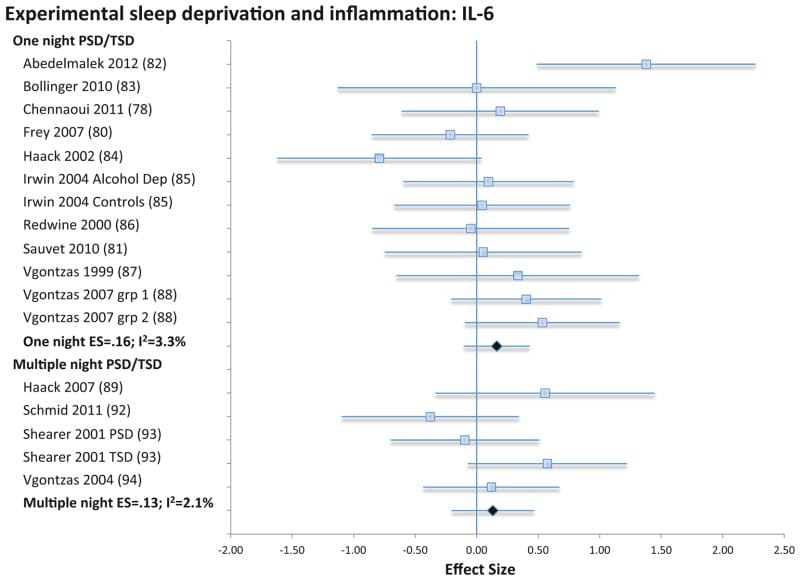
Chronic insomnia produces the same pattern at a lower magnitude.
The same meta-analysis found that sleep disturbance was associated with elevated CRP, with an overall CRP effect size of 0.12 and an IL-6 effect size of 0.20. In young adults, insomnia symptoms were independently associated with elevated CRP after controlling for depression, anxiety, and neuroticism, although this was a small observational sample. In contrast, the large Norwegian HUNT study did not find consistent associations between insomnia symptoms and hs-CRP, so it should not be used as primary support for a strong insomnia-CRP link.
Does High CRP Make Sleep Worse?
In the CARDIA study, sleep disturbance was a significant predictor of CRP and IL-6 five years later, and the association held after controlling for baseline CRP levels. That finding supports sleep disturbance as a longitudinal predictor of inflammation. It does not, by itself, prove a bidirectional CRP-sleep loop. In people with inflammatory bowel disease, sleep disturbances were documented independent of disease activity, which supports a broader link between inflammatory conditions and poor sleep continuity.
If your CRP is elevated and your sleep is poor, the two may share overlapping drivers, including adiposity, infection, autoimmune activity, sleep apnea, circadian disruption, stress physiology, or chronic insomnia.
Sleep remains a plausible contributor, but CRP is not specific enough to identify sleep as the cause by itself.
Are CRP Responses to Sleep Loss Different for Men and Women?
The Whitehall II Study found that women sleeping 5 hours or fewer per night had elevated hs-CRP after controlling for BMI, physical activity, alcohol, smoking, and other covariates. Men sleeping 5 hours or fewer showed no hs-CRP association.
Whitehall II did not show significant sleep-duration variation in inflammatory markers among men.
In young adults from the Add Health cohort, persistent short sleep duration was associated with elevated hs-CRP in males, while persistent long sleep duration was associated with elevated hs-CRP in females. In a Korean population study, women showed hs-CRP associations with difficulty initiating sleep, poor sleep quality, and some maintaining-sleep categories. Men showed no significant sleep-quality association with hs-CRP; the clearest male association was excessive sleep duration of 10 hours or more.
These are population-level associations, and individual variation exists. The pattern is not “women always respond more.” A more precise reading is that sex modifies the CRP-sleep association, and the direction depends on sleep duration, sleep quality, age, and population.
Does Improving Sleep Lower CRP?
The data comes from a randomized controlled trial of CBT-I in older adults with insomnia. Compared with a sleep education control, CBT-I reduced the odds of CRP > 3.0 mg/L at 16 months. This is important because 3.0 mg/L is the AHA/CDC high cardiovascular-risk threshold for hs-CRP.
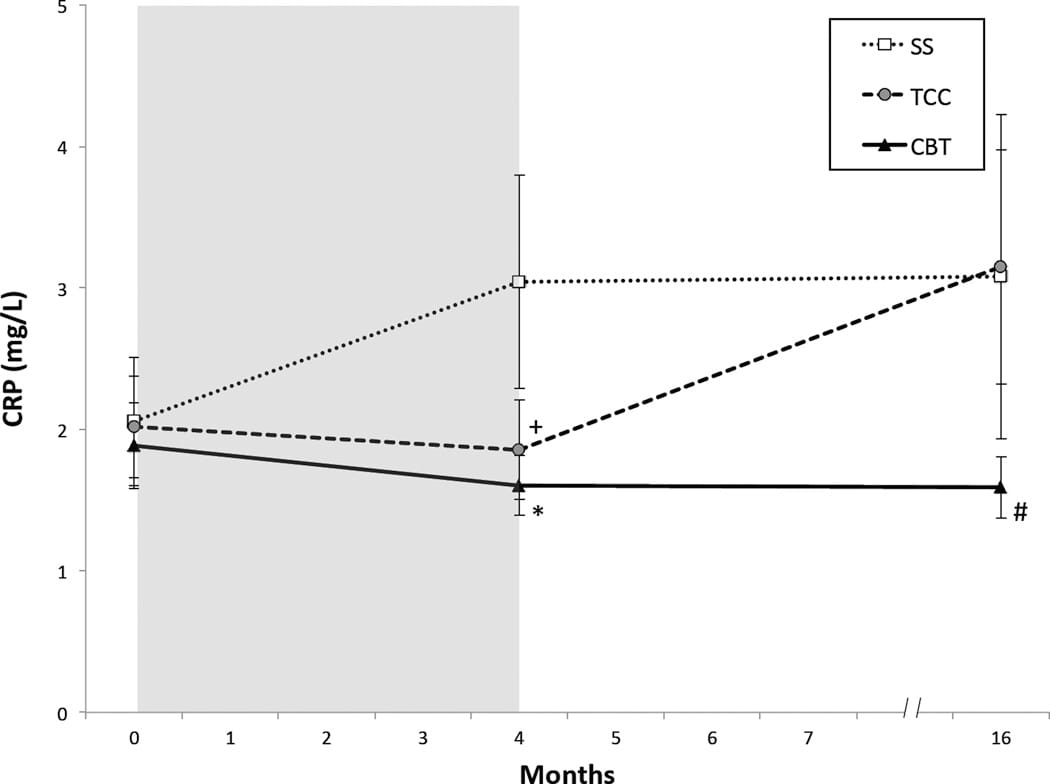
A systematic review of randomized controlled trials found that CBT-I was associated with improved CRP and glucose regulation across RCTs. However, many included trials had high risk of bias, so the direction of evidence is encouraging rather than definitive.
Weekend catch-up sleep tells a more conditional story. A cross-sectional study of > 17,000 adults found moderate catch-up sleep, defined as 1 to less than 3 extra hours on weekends, was associated with 13% lower odds of being in the highest hs-CRP quartile. This pattern was strongest in people with consistent weekday-weekend bedtimes. Severe catch-up sleep, defined as 3 or more additional hours, did not show the same benefit. Because the study was cross-sectional, it cannot prove that catch-up sleep lowered CRP. It does suggest that sleep regularity may matter as much as total weekend recovery sleep.
Elevated CRP from poor sleep is one inflammatory factor, but it may compound with sleep apnea, circadian disruption, infection, autoimmune activity, metabolic disease, adiposity, or other inflammatory drivers. Identifying which causes are active helps prioritize where to start.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
What Is the Difference Between CRP and hs-CRP?
The AHA/CDC cardiovascular-risk thresholds use hs-CRP: below 1.0 mg/L is low risk, 1.0-3.0 mg/L is moderate risk, and above 3.0 mg/L is high risk.
Is CRP Elevated With Sleep Apnea?
Intermittent hypoxia and short sleep duration may act as independent inflammatory stressors. In patients with coronary artery disease, nocturnal intermittent hypoxia and short sleep duration were independently associated with elevated CRP.
What Does Elevated CRP Feel Like?
This range is still clinically relevant because it carries higher long-term cardiovascular risk in population studies. More on the fatigue-insomnia paradox: Inflammation: Exhausted But Can’t Sleep.
What CRP Level Should You Be Concerned About?
In context, sleep-related CRP shifts can occur within the low-grade range, so the absolute value matters less than persistence, clinical context, and other inflammatory causes. CBT-I trials suggest that treating insomnia can reduce the odds of remaining above the 3.0 mg/L high-risk threshold.
If your hs-CRP is persistently above 1.0 mg/L and you have poor sleep, sleep quality is a plausible contributing factor. It should be considered alongside body composition, infection, autoimmune disease, sleep apnea, smoking, medications, oral health, and other medical causes.
Related Reading
- Inflammatory Sleep Disruption — full cause overview (pillar)
- Histamine and 3am Waking
- Histamine Intolerance and Sleep
- Why Is Inflammation Worse at Night?
- Leaky Gut and Insomnia
- Brain Fog and Poor Sleep: Neuroinflammation
- The Glymphatic System and Sleep
- Chronic Inflammation and Insomnia
- Why Do Men Sleep Worse After 50?
- Autoimmune Insomnia
- Inflammation and 3am Wakeups
- Inflammation: Exhausted But Can’t Sleep
- Stress, Inflammation, and Insomnia
- Gut Bacteria and Insomnia
- Anti-Inflammatory Diet and Sleep
- Inflammaging and Sleep
- NLRP3 Inflammasome and Sleep
- Prostaglandins and Sleep
- Mast Cell Activation and Sleep
- Does Circadian Disruption Cause Inflammation
References
Cho, H. J., Seeman, T. E., Kiefe, C. I., Lauderdale, D. S., & Irwin, M. R. (2015). Sleep disturbance and longitudinal risk of inflammation: Moderating influences of social integration and social isolation in the Coronary Artery Risk Development in Young Adults (CARDIA) study. Brain, Behavior, and Immunity, 46, 319-326. https://pubmed.ncbi.nlm.nih.gov/25733101/
Bray, C., Bell, L. N., Liang, H., Haykal, R., Kaiksow, F., Mazza, J. J., & Yale, S. H. (2016). Erythrocyte sedimentation rate and C-reactive protein measurements and their relevance in clinical medicine. Wisconsin Medical Journal, 115(6), 317-321. https://pubmed.ncbi.nlm.nih.gov/29094869/
Lee, H.-W., Yoon, H.-S., Yang, J. J., Song, M., Lee, J.-K., Lee, S.-A., Choi, J.-Y., & Kang, D. (2020). Association of sleep duration and quality with elevated hs-CRP among healthy Korean adults. PLoS One, 15(8), e0238053. https://pubmed.ncbi.nlm.nih.gov/32841297/
Slavish, D. C., Graham-Engeland, J. E., Engeland, C. G., Taylor, D. J., & Buxton, O. M. (2018). Insomnia symptoms are associated with elevated C-reactive protein in young adults. Psychology & Health, 33(11), 1396-1415. https://pubmed.ncbi.nlm.nih.gov/30358412/
Woo, S.-J., Chae, C.-H., & Lim, J.-W. (2022). Association between shift work and inflammatory markers in workers at an electronics manufacturing company. Annals of Occupational and Environmental Medicine, 34, e35. https://pubmed.ncbi.nlm.nih.gov/36544883/
Haack, M., Sanchez, E., & Mullington, J. M. (2007). Elevated inflammatory markers in response to prolonged sleep restriction are associated with increased pain experience in healthy volunteers. Sleep, 30(9), 1145-1152. https://pmc.ncbi.nlm.nih.gov/articles/PMC1978405/
Irwin, M. R., Olmstead, R., Carrillo, C., Sadeghi, N., Nicassio, P., Ganz, P. A., & Bower, J. E. (2015). Cognitive behavioral therapy vs. tai chi for late life insomnia and inflammatory risk. Biological Psychiatry, 78(6), 856-864. https://pubmed.ncbi.nlm.nih.gov/25748580/
Irwin, M. R., Olmstead, R., & Carroll, J. E. (2016). Sleep disturbance, sleep duration, and inflammation: A systematic review and meta-analysis. Biological Psychiatry, 80(1), 40-52. https://pubmed.ncbi.nlm.nih.gov/26140821/
Savin, K. L., Clark, T. L., Perez-Ramirez, P., Allen, T. S., Tristão Parra, M., & Gallo, L. C. (2023). The effect of cognitive behavioral therapy for insomnia (CBT-I) on cardiometabolic health biomarkers: A systematic review of randomized controlled trials. Behavioral Sleep Medicine, 21(6), 671-694. https://pubmed.ncbi.nlm.nih.gov/36476211/
Miller, M. A., Kandala, N. B., Kivimaki, M., Kumari, M., Brunner, E. J., Lowe, G. D., Marmot, M. G., & Cappuccio, F. P. (2009). Gender differences in cross-sectional relationships between sleep duration and markers of inflammation: Whitehall II study. Sleep, 32(7), 857-864. https://pubmed.ncbi.nlm.nih.gov/19639748/
Bakour, C., Schwartz, S., O’Rourke, K., Wang, W., Sappenfield, W., Couluris, M., & Chen, H. (2017). Sleep duration trajectories and systemic inflammation in young adults: Results from the National Longitudinal Study of Adolescent to Adult Health (Add Health). Sleep, 40(11), zsx156. https://pubmed.ncbi.nlm.nih.gov/29155987/
Fukuoka, R., Kohno, T., Kohsaka, S., Yanagisawa, R., Kawakami, T., Hayashida, K., Kanazawa, H., Yuasa, S., Maekawa, Y., Sano, M., & Fukuda, K. (2017). Nocturnal intermittent hypoxia and short sleep duration are independently associated with elevated C-reactive protein levels in patients with coronary artery disease. Sleep Medicine, 29, 29-34. https://pubmed.ncbi.nlm.nih.gov/28153212/
Ranjbaran, Z., Keefer, L., Farhadi, A., Stepanski, E., Sedghi, S., & Keshavarzian, A. (2007). Impact of sleep disturbances in inflammatory bowel disease. Journal of Gastroenterology and Hepatology, 22(11), 1748-1753. https://pubmed.ncbi.nlm.nih.gov/17914945/
Li, K., Wei, P., Qin, Y., & Wei, Y. (2017). Is C-reactive protein a marker of obstructive sleep apnea?: A meta-analysis. Medicine (Baltimore), 96(19), e6850. https://pubmed.ncbi.nlm.nih.gov/28489776/
Laugsand, L. E., Vatten, L. J., Bjørngaard, J. H., Hveem, K., & Janszky, I. (2012). Insomnia and high-sensitivity C-reactive protein: the HUNT study, Norway. Psychosomatic Medicine, 74(5), 543-553. https://pubmed.ncbi.nlm.nih.gov/22685243/
Vgontzas, A. N., Zoumakis, E., Bixler, E. O., Lin, H. M., Follett, H., Kales, A., & Chrousos, G. P. (2004). Adverse effects of modest sleep restriction on sleepiness, performance, and inflammatory cytokines. Journal of Clinical Endocrinology and Metabolism, 89(5), 2119-2126. https://pubmed.ncbi.nlm.nih.gov/15126529/
Park, S., Kang, D. Y., Ahn, H., Kim, N., Yoon, J.-H., & Yang, B. R. (2022). Effect of weekend catch-up sleep on high-sensitivity C-reactive protein levels according to bedtime inconsistency: a population-based cross-sectional study. Scientific Reports, 12, 21619. https://doi.org/10.1038/s41598-022-25787-x
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 17 references · Third-party fact-checked and revised 2026-05-01
