The common understanding of stress-related insomnia is that excess cortisol keeps the brain wired. The mechanism is less intuitive. Chronic stress changes how immune cells respond to cortisol at the receptor level, allowing inflammatory cytokine production to run unchecked. That inflammatory output then disrupts sleep architecture in measurable ways. And the fragmented sleep feeds right back into the same inflammatory pathway.
This article traces the full cycle — stress to inflammation, inflammation to sleep disruption, sleep disruption back to inflammation — with the human evidence behind each link. It does not cover the metabolic cortisol-blood-sugar pathway to 3am waking (that is a separate mechanism covered in the metabolic cluster). For the broader overview of inflammatory sleep disruption, see the parent article: Inflammatory Sleep Disruption.
How Does Chronic Stress Cause Inflammation?
The mechanistic foundation comes from Cohen et al. (2012), who ran two linked studies. In the first (n=276), participants with long-duration stressful life events showed higher susceptibility to developing a confirmed cold after rhinovirus exposure. In the second (n=79), the mechanism was identified: peripheral blood mononuclear cells from chronically stressed participants showed blunted dexamethasone suppression of LPS-stimulated cytokine production (p<0.05). Dexamethasone is a synthetic glucocorticoid — a proxy for cortisol — so blunted suppression means the immune cells were no longer responding to cortisol's anti-inflammatory instruction. Among infected subjects, greater glucocorticoid receptor resistance predicted higher local production of IL-1β, TNF-α, and IL-6. The causal chain: stress produces glucocorticoid receptor resistance, which amplifies inflammatory output.
Miller, Cohen, and Ritchey (2002) documented this in caregivers. Parents caring for children with cancer showed flatter diurnal cortisol slopes — reduced morning rise, maintained evening levels — AND diminished dexamethasone suppression of IL-6 production. Total daily cortisol output did not differ from controls. This rules out the “too much cortisol” explanation. Glucocorticoid receptor resistance is a receptor-level phenomenon: the cortisol is present, but the cells have stopped listening to it. The flattened cortisol slope documented here mirrors the pattern found in chronic insomnia populations — reduced circadian contrast between morning and evening cortisol.
Miller et al. (2008) provided the genomic proof. Monocytes from chronically stressed caregivers of brain-cancer patients showed reduced transcription of genes bearing glucocorticoid response elements (where cortisol exerts its anti-inflammatory effects) and increased transcription of NF-κB target genes — IL-1β, IL-6, IL-8, and TNF pathway genes. Serum CRP and IL-1 receptor antagonist were elevated, confirming the genomic change translated to measurable circulating inflammation. Despite similar plasma cortisol between groups, the stressed group’s immune cells were selectively unresponsive to cortisol at the transcriptional level.
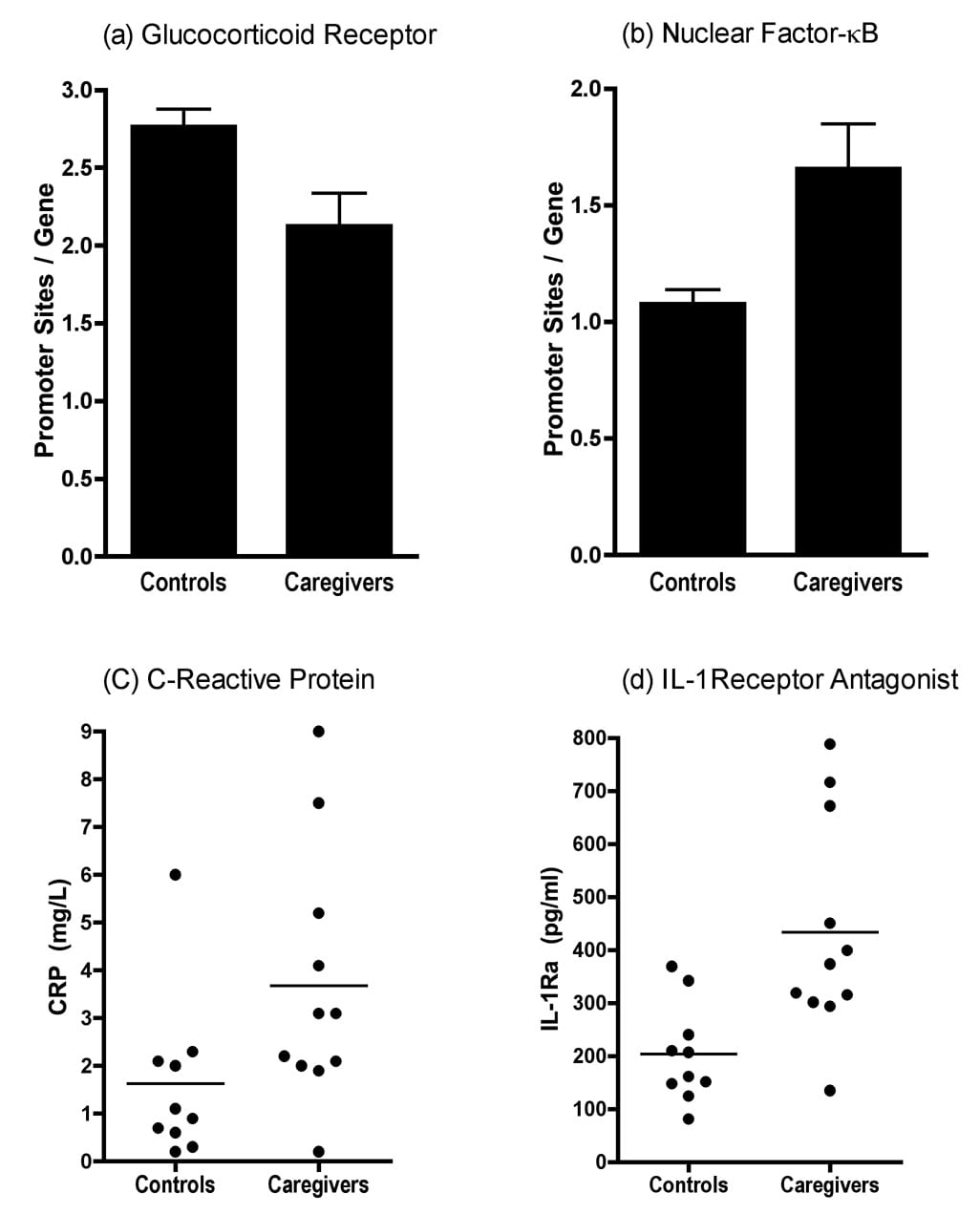
The logical chain: stress does not equal “too much cortisol.” Chronic stress breaks cortisol’s ability to suppress inflammation at the receptor level. The immune response escapes its primary anti-inflammatory regulator.
How Does Stress-Induced Inflammation Disrupt Sleep?
Vgontzas et al. (2002) conducted the defining cytokine-timing study. Comparing 11 chronic insomnia participants with 11 age- and BMI-matched controls, cosinor analysis showed the IL-6 secretion peak moved from 4am (controls) to 7pm (insomnia group), p<0.05. This is a full circadian inversion. TNF-α rhythm was blunted nocturnally and fragmented into a 4-hour daytime pulse pattern. Both cytokine timing inversions correlated with daytime fatigue. The insomnia group also showed 24-hour hypersecretion of corticotropin-releasing hormone (CRH, the hypothalamic hormone that drives cortisol production) and cortisol itself — around-the-clock hyperarousal, not confined to nighttime.
This cytokine timing inversion produces the “wired at 3am” phenotype through a defined sequence: evening IL-6 elevation activates the HPA axis (the hypothalamic-pituitary-adrenal cascade that produces cortisol), elevated evening cortisol impairs sleep initiation, fragmented sleep further dysregulates the next night’s cytokine rhythm. The wakefulness is not unpatterned — it is the downstream output of an inflammatory-neuroendocrine timing inversion.
Passos et al. (2023) confirmed the cortisol-insomnia link in a PSG-verified (polysomnography — overnight sleep recording) chronic insomnia population (n=34). Insomnia Severity Index scores correlated with morning salivary cortisol (r=0.37, p=0.03) and with anxiety and depression scores. N3 sleep — slow-wave deep sleep, the restorative stage — negatively correlated with tension-anxiety (r=-0.36, p=0.04). This pattern indicates that inflammatory arousal suppresses deep sleep while preserving lighter stages. Sleep quantity may look adequate on a report. Sleep quality — the proportion of restorative deep sleep — erodes.
The distinction between this pathway and other inflammatory insomnia mechanisms matters. The cytokine timing inversion described here is driven upstream by glucocorticoid receptor resistance from chronic psychological stress. Other inflammatory insomnia mechanisms — such as gut permeability-driven LPS translocation or histamine-mediated arousal — involve different upstream triggers producing inflammation through separate pathways. For an overview of how NF-κB and the circadian clock interact during the pre-dawn hours, see why inflammation worsens at night.
Why Does Poor Sleep Make Stress-Related Inflammation Worse?
Piber et al. (2023) dissociated sleep continuity from sleep duration as independent drivers of inflammatory activation. In a cohort of 262 older adults, greater wake time after sleep onset (WASO — the total minutes spent awake between initial sleep onset and final awakening) predicted higher NF-κB activation in peripheral blood mononuclear cells (beta=0.42, p<0.01) after full covariate adjustment. Total sleep time showed no association with NF-κB or STAT protein activation. It is the continuity of sleep, not its duration, that drives inflammatory transcription factor activation.
Among female participants, elevated WASO also predicted higher STAT1, STAT3, and STAT5 activation (p<0.05 for STAT1 and STAT3; p<0.01 for STAT5) — transcription factors that amplify cytokine production cascades, feeding back into IL-6 and IL-1β output. No comparable associations were found in male participants. This is the first study to identify sleep fragmentation as an independent driver of transcriptional inflammatory activation in humans — separate from total sleep loss.
Knight et al. (2021) provided the population-scale evidence. Using structural equation modeling in 914 adults from the national MIDUS 2 cohort, they confirmed a statistically relevant indirect pathway: perceived psychological stress flattens the diurnal cortisol slope, and that flattened slope elevates IL-6, CRP, fibrinogen, E-Selectin, and ICAM-1. The pathway replicated across all five inflammatory markers. Cortisol was measured via ambulatory salivary sampling across multiple days — not a single-point laboratory draw — providing ecologically valid evidence for diurnal cortisol flattening under chronic stress.

The self-reinforcing cycle in plain terms: stress breaks cortisol’s anti-inflammatory regulation (glucocorticoid receptor resistance), escaped inflammation disrupts sleep architecture (cytokine timing inversion), and fragmented sleep activates the NF-κB pathway that started the cascade. Each pass through the cycle deepens glucocorticoid receptor resistance, worsens the cytokine timing inversion, and further flattens the cortisol slope. For a broader overview of how NF-κB activation from poor sleep amplifies inflammation, see the bidirectional inflammation-insomnia cycle.
Can Breaking the Stress-Inflammation Cycle Improve Sleep?
Breaking at the stress link. The Knight et al. (2021) structural equation model identified the flattened diurnal cortisol slope — blunted morning rise, maintained evening levels — as the cortisol pattern that mediates the connection between perceived stress and elevated inflammatory markers. Approaches that restore cortisol rhythm contrast (high morning, low evening) address this mechanism. The measurable target is the diurnal cortisol slope itself, not overall cortisol reduction. This matters because total daily cortisol output may be normal even when the slope is flattened — as Miller, Cohen, and Ritchey (2002) documented in chronically stressed caregivers.
Breaking at the inflammation link. The genomic fingerprint from Miller et al. (2008) identified NF-κB gene upregulation as the transcriptional driver of stress-related inflammation. Approaches that reduce NF-κB activity address the molecular mechanism. The Piber et al. (2023) finding that sleep fragmentation (not duration) drives NF-κB activation means that sleep continuity improvements are mechanistically precise — they target the inflammatory pathway rather than reducing inflammation through an indirect route.
Breaking at the sleep link. Sleep continuity improvement reduces NF-κB activation (Piber et al., 2023). This means sleeping without frequent awakenings — not sleeping longer. The Vgontzas et al. (2002) cytokine timing data suggests that restoring consolidated nocturnal sleep may allow IL-6 to return to its normal nighttime pattern, removing the daytime fatigue driver while restoring the nighttime consolidation contribution.
Each entry point feeds back into the others. Restored cortisol rhythm reduces inflammatory output, which improves sleep continuity, which further reduces NF-κB activation. The same self-reinforcing property that makes this cycle difficult to escape also means that interrupting one link can cascade benefits to the other two.
The question for someone caught in this cycle is which link offers the best starting point — the stress exposure, the inflammatory load, or the sleep fragmentation — and that depends on individual circumstances the article cannot determine.
Stress-driven inflammation is one path to sleep disruption, but it can compound with hormonal changes, metabolic patterns, circadian misalignment, or autonomic dysregulation. Identifying which causes are active helps direct the response.
Find out which causes may be driving your 3am wakeups →
Frequently Asked Questions
Does Inflammation Elevate Cortisol?
This is the paradox Cohen et al. (2012) documented: stressed subjects had functional cortisol production, but their immune cells showed blunted response to it. The result is elevated cortisol AND elevated inflammation at the same time — a combination that seems contradictory until glucocorticoid receptor resistance is understood. Cortisol is present and being produced, but the immune cells that should be responding to its anti-inflammatory action have downregulated their sensitivity.
Does Cortisol Reduce Inflammation or Cause It?
The Miller et al. (2008) genomic study showed this — stressed individuals had normal circulating cortisol but reduced glucocorticoid response element-driven gene transcription and increased NF-κB target gene expression. Cortisol’s relationship with inflammation is duration-dependent, not dose-dependent.
What Is the Cortisol Awakening Response?
A blunted CAR is associated with the flattened diurnal cortisol slope that Knight et al. (2021) linked to elevated IL-6, CRP, and fibrinogen in a national sample. Because the flattened slope also impairs evening cortisol decline — maintaining cortisol at levels that interfere with sleep onset — a blunted CAR can indicate that the stress-inflammation cycle is already active.
Does Magnesium Reduce Cortisol at Night?
Some studies show magnesium supplementation improves subjective sleep quality in populations with low baseline magnesium, but the mechanism is not demonstrated to be cortisol-mediated. This is a plausible indirect effect — better sleep leading to lower cortisol — rather than a demonstrated pharmacological pathway from magnesium to cortisol reduction. Anyone considering magnesium for sleep should understand this distinction: it may help through a parallel mechanism rather than by interrupting the stress-inflammation cycle described in this article.
Related Reading
- Inflammatory Sleep Disruption — the full cause overview
- Chronic Inflammation and Insomnia — the bidirectional cytokine-insomnia loop
- Why Is Inflammation Worse at Night? — NF-κB circadian mechanism referenced in this article
- Brain Fog and Poor Sleep — neuroinflammatory consequences of sleep fragmentation
- Histamine and 3am Waking — a separate inflammatory arousal mechanism
- Leaky Gut and Insomnia — gut permeability as an independent inflammatory driver
- The Glymphatic System and Sleep — how sleep fragmentation impairs brain waste clearance
- Can Histamine Intolerance Cause Sleep Problems?
- Why Do Men Sleep Worse After 50?
- What Is Autoimmune Insomnia and Why Does It Resist Standard Sleep Approaches?
- Can Inflammation Cause 3am Wakeups?
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- Gut Bacteria and Insomnia: Which Microbes Affect Your Sleep (and Which Ones Help)
- Does an Anti-Inflammatory Diet Improve Sleep?
References
Cohen, S., Janicki-Deverts, D., Doyle, W. J., Miller, G. E., Frank, E., Rabin, B. S., & Turner, R. B. (2012). Chronic stress, glucocorticoid receptor resistance, inflammation, and disease risk. Proceedings of the National Academy of Sciences, 109(16), 5995–5999. https://pubmed.ncbi.nlm.nih.gov/22474371/
Knight, E. L., Jiang, Y., Rodriguez-Stanley, J., Almeida, D. M., Engeland, C. G., & Zilioli, S. (2021). Perceived stress is linked to heightened biomarkers of inflammation via diurnal cortisol in a national sample of adults. Brain, Behavior, and Immunity, 93, 206–213. https://pubmed.ncbi.nlm.nih.gov/33515741/
Miller, G. E., Cohen, S., & Ritchey, A. K. (2002). Chronic psychological stress and the regulation of pro-inflammatory cytokines: A glucocorticoid-resistance model. Health Psychology, 21(6), 531–541. https://pubmed.ncbi.nlm.nih.gov/12433005/
Miller, G. E., Chen, E., Sze, J., Marin, T., Arevalo, J. M. G., Doll, R., Ma, R., & Cole, S. W. (2008). A functional genomic fingerprint of chronic stress in humans: Blunted glucocorticoid and increased NF-kappaB signaling. Biological Psychiatry, 64(4), 266–272. https://pubmed.ncbi.nlm.nih.gov/18440494/
Passos, G. S., Youngstedt, S. D., Rozales, A. R. C., Ferreira, W. S., De-Assis, D. E., De-Assis, B. P., & Santana, M. G. (2023). Insomnia severity is associated with morning cortisol and psychological health. Sleep Science, 16(1), 92–96. https://pubmed.ncbi.nlm.nih.gov/37151768/
Piber, D., Olmstead, R., Cho, J. H., & Irwin, M. R. (2023). Disturbance of sleep maintenance, but not sleep duration, activates nuclear factor-κB and signal transducer and activator of transcription family proteins in older adults: Sex differences. Sleep, 46(10), zsad130. https://pubmed.ncbi.nlm.nih.gov/37140651/
Vgontzas, A. N., Zoumakis, M., Papanicolaou, D. A., Bixler, E. O., Prolo, P., Lin, H. M., Vela-Bueno, A., Kales, A., & Chrousos, G. P. (2002). Chronic insomnia is associated with a shift of interleukin-6 and tumor necrosis factor secretion from nighttime to daytime. Metabolism, 51(7), 887–892. https://pubmed.ncbi.nlm.nih.gov/12077736/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
