Every wellness site has a version of “anti-inflammatory foods for better sleep.” The peer-reviewed literature does support a connection between dietary inflammation and sleep — but the findings are more specific, more limited, and more useful than a list of salmon and blueberries. There are now over a dozen studies linking the Dietary Inflammatory Index (DII) to individual sleep metrics, a half-million-participant meta-analysis on the Mediterranean diet and insomnia, and a randomized-controlled-trial-only meta-analysis on omega-3 and sleep outcomes. This article walks through the research by evidence type — population data, dietary pattern analyses, and supplement trials — and draws a line between what the data supports and where popular coverage overclaims. The pillar article covers the full inflammatory sleep disruption picture; this article focuses on the dietary evidence.
What Does the Research Say About Anti-Inflammatory Diets and Sleep?
The DII scores 45 dietary components — nutrients, flavonoids, spices, and other food constituents — on a scale from pro-inflammatory to anti-inflammatory, calculated from dietary recall data and energy-adjusted for body size. It was developed as a standardized research tool so that studies across different populations use the same metric for dietary inflammation (Farrell et al., 2024). Nearly every study in this field uses some version of the DII or its energy-adjusted variant (E-DII) to quantify the inflammatory potential of a person’s diet.
The largest US population study on this question is Kase et al. (2021), drawing on NHANES data from 2005-2016 across 30,121 adults aged 20 and older. Adults in the highest E-DII quintile — meaning the most pro-inflammatory dietary pattern — had 40% greater odds of short sleep duration of six hours or less compared to the lowest quintile (OR=1.40, 95% CI: 1.21-1.61). Long sleep duration of nine hours or more was also elevated in higher E-DII quintiles (OR=1.23), creating a U-shaped relationship where both too-short and too-long sleep associate with inflammatory diets. Sleep disturbance risk rose in the highest quintile as well (OR=1.14, 95% CI: 1.02-1.27).
That study is cross-sectional, meaning it cannot establish that diet caused the sleep problems — the association could run in either direction. Jiang et al. (2025) addressed this limitation. Using NHANES data from 2007-2018 (n=24,780) combined with two-sample Mendelian randomization — a method that uses genetic variants as proxies to reduce confounding and test causal direction — they found each one-unit DII increase was associated with 12.9% elevated sleep disorder risk (OR=1.129, 95% CI: 1.079-1.182). The Mendelian randomization analysis supports a causal direction from diet to sleep disruption rather than the reverse. At the nutrient level, their analysis identified selenium as protective against sleep apnea (OR=0.92, 95% CI: 0.86-0.98) and vitamin C and monounsaturated fatty acids as protective against sleep-wake disorders.
A limitation: NHANES relies on a single 24-hour dietary recall per participant, which may not capture habitual eating patterns. The Mendelian randomization component partially compensates for the cross-sectional design, but the MR assumptions — that the genetic instruments are valid and only affect sleep through diet — cannot be fully verified.
Does the Mediterranean Diet Improve Sleep?
Arab et al. (2024) conducted the largest meta-analysis to date on dietary patterns and insomnia, pooling 37 observational studies involving 591,223 participants across multiple countries and age groups. Mediterranean diet adherence was associated with a 14% reduction in insomnia risk (OR: 0.86; 95% CI: 0.79-0.93; p<0.001). High overall diet quality — measured by indices that overlap with but extend beyond Mediterranean diet scoring — showed an even larger 34% reduction (OR: 0.66; 95% CI: 0.48-0.90; p=0.010). This study was co-authored by Frank Scheer, a circadian researcher at Harvard, and insomnia was assessed using validated instruments including the Insomnia Severity Index and the Athens Insomnia Scale, which reduces measurement bias.
Godos et al. (2024) published the broadest review focused on the Mediterranean diet and sleep, synthesizing 23 reports across European, North American, and Middle Eastern populations. Higher adherence was associated with lower likelihood of poor sleep quality, inadequate sleep duration, excessive daytime sleepiness, and insomnia across the majority of included studies. Studies conducted outside the traditional Mediterranean region showed the same beneficial associations — the findings are not explained by climate or lifestyle factors specific to Southern Europe.
Here is where the evidence becomes more nuanced than popular coverage suggests. Of the three randomized controlled trials included in the Godos review, two showed no statistically meaningful improvement in sleep outcomes. The observational data is consistent and comes from large samples, but the evidence from trials — where researchers assign people to a Mediterranean diet and measure whether sleep improves — is weaker than what health sites typically imply.
There is also a bidirectional complication. Poor sleep impairs food choices — sleep-deprived people tend to eat more calorie-dense, processed food. This means the observational association between Mediterranean diet adherence and better sleep partially reflects reverse causation: people who sleep well may eat better because they sleep well, rather than sleeping well because they eat better. Observational designs cannot fully untangle this.
The practical takeaway: the Mediterranean diet is a reasonable dietary approach supported by strong population-level data. But “proven to fix your sleep” overstates what the current evidence base shows. The gap between observational and trial-level evidence is where overclaiming happens on health sites.
Do Omega-3 Fatty Acids Improve Sleep?
Shimizu et al. (2024) conducted a meta-analysis restricted to randomized controlled trials — 8 trials comparing omega-3 long-chain polyunsaturated fatty acid supplementation against placebo. This RCT-only design makes it the highest-quality evidence available for any single anti-inflammatory nutrient and sleep. Prior meta-analyses mixed randomized trials with observational data, which inflates apparent effect sizes.
The primary finding: sleep efficiency was higher in omega-3 groups compared to placebo groups — a consistent result across the included trials. Sleep latency (time to fall asleep) and total sleep duration did not differ between groups at the pooled level. This pattern echoes the broader anti-inflammatory diet literature: quality metrics respond, quantity metrics do not.
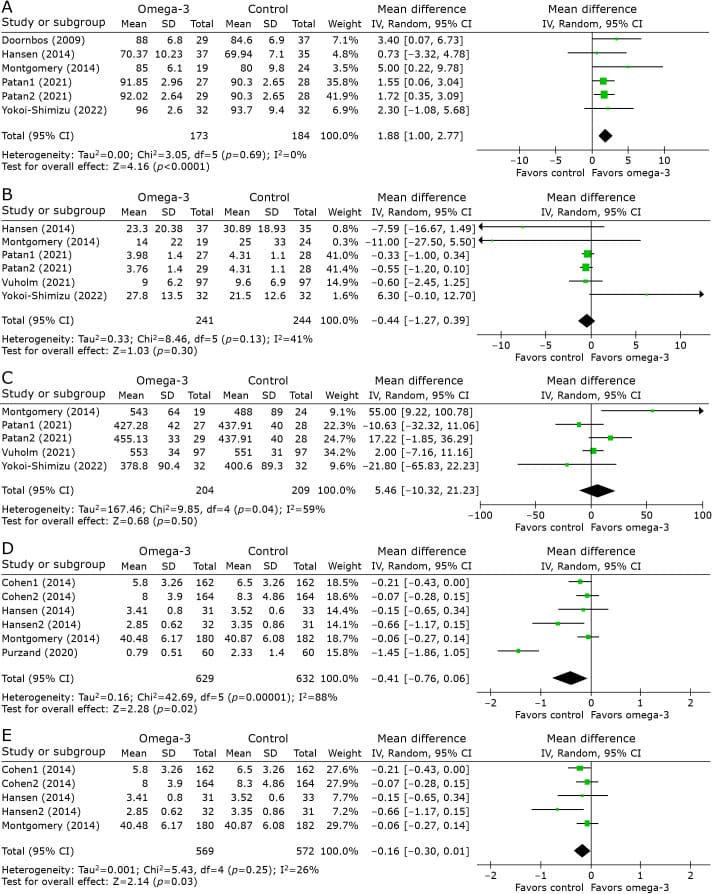
DHA (docosahexaenoic acid) shows more consistent benefit than EPA (eicosapentaenoic acid) in subgroup analyses. This aligns with DHA’s known role in cell membrane composition and serotonin receptor function — DHA is concentrated in neural tissue in a way that EPA is not.
Dose matters. Subjectively assessed sleep quality showed improvement overall, though initial statistical heterogeneity was high. When the analysis was stratified by supplementation dose, heterogeneity dropped and the improvement became consistent, suggesting that reaching an adequate dose is a factor in whether omega-3 affects sleep.
Effect sizes are modest. Omega-3 supplementation is not equivalent to a pharmacological sleep aid in magnitude. This is a real, replicable effect on a meaningful metric (sleep efficiency), but anyone expecting the same impact as a prescription medication will be disappointed. The value of this evidence is its quality — RCTs with placebo controls — rather than its magnitude.
What Does Anti-Inflammatory Eating Improve About Sleep — and What Doesn’t It Change?
Farrell et al. (2024) published the definitive DII-sleep review, authored by the group that developed and validated the Dietary Inflammatory Index as a research tool (Hebert, Shivappa). Across all 12 included studies — cross-sectional, longitudinal, and one trial — more anti-inflammatory diets were associated with better sleep in at least one domain. The metrics that showed the most consistent and reproducible associations were sleep efficiency and WASO. The metric that did not associate with DII across studies: total sleep duration.
This distinction is the central finding of the DII-sleep literature and the one that popular health articles miss. Anti-inflammatory eating is associated with sleeping more efficiently — less time awake during the night, fewer disruptions, higher percentage of time in bed spent asleep. It is not associated with sleeping longer. Popular articles claiming that anti-inflammatory diets help you “sleep longer” or “get more hours” are not supported by the current evidence base.
The strongest individual study supporting this pattern is Farrell et al. (2023), a prospective longitudinal cohort that tracked 427 adults across four timepoints over three years. Sleep was measured with actigraphy (objective measurement via wearable sensor), and DII was calculated from three unannounced 24-hour dietary recalls at each wave — stronger methodology than the single-recall approach used in NHANES. For each one-unit increase in DII (more pro-inflammatory), WASO increased by approximately one minute (beta=0.80, p=0.02). Longitudinally, every one-unit increase in DII change predicted a WASO increase (beta=1.00, p=0.01) and a sleep efficiency decrease (beta=-0.16, p<0.05).
An additional finding rarely mentioned in popular coverage: pro-inflammatory dietary changes also predicted later bedtime and later waketime — a circadian timing effect. The diet-sleep connection may extend beyond inflammation into circadian regulation, though the current evidence cannot determine whether this runs through inflammatory pathways, circadian mechanisms, or both.
Race modified these associations. African American participants showed stronger bedtime-delay effects from pro-inflammatory diets than European American participants, indicating the diet-sleep relationship is not uniform across populations.
Studies using rigorous measurement — multiple dietary recalls combined with actigraphy — showed stronger and more consistent results than those relying on single dietary recalls or subjective sleep questionnaires alone. Measurement quality matters. The weakest associations came from studies with the weakest measurement tools, which suggests the true effect may be larger than what lower-quality studies detect.
Diet-related inflammation is one contributor to sleep disruption, but it can compound with hormonal changes, metabolic patterns, circadian timing, or autonomic dysregulation. Multiple causes might be active at once — dietary change alone might not be enough if other drivers are present.
Find out which causes might be driving your 3am wakeups
Frequently Asked Questions
Can an Anti-Inflammatory Diet Improve Sleep After 50?
The DII-sleep associations in NHANES data include adults aged 20 and older across age strata. Kase et al. (2021) captured age as a covariate, and the DII-sleep association persisted across age groups. This makes physiological sense: as inflammatory burden increases with age, the relative contribution of dietary inflammation to overall inflammatory load becomes more pronounced. Someone at 55 with elevated baseline inflammation from aging-related immune changes has a higher starting point — dietary changes that reduce that load have more room to produce a measurable effect. The practical message is that dietary change becomes more relevant, not less, as age-related inflammatory burden accumulates. For more on the intersection of aging-related inflammation and sleep, see Inflammaging and Sleep.
Does a High Glycemic Diet Worsen Sleep?
This is the flip side of the Mediterranean diet finding. High-glycemic diets are pro-inflammatory in nature — they trigger blood sugar spikes followed by compensatory cortisol and insulin responses, and the downstream inflammatory load accumulates over time. The 16% increase in insomnia risk from high glycemic index diets (Arab et al., 2024) represents a meaningful population-level effect. It is not only that anti-inflammatory eating patterns associate with better sleep — pro-inflammatory eating patterns associate with worse sleep. The two directions of evidence reinforce each other and make the overall finding more credible than either direction alone.
How Long Does It Take for Dietary Changes to Affect Sleep?
The Farrell et al. (2023) prospective cohort measured DII and sleep at baseline, one year, two years, and three years — the changes in WASO and sleep efficiency associated with DII changes emerged across these intervals. Shorter-term data from supplement trials (including the omega-3 RCTs in Shimizu et al., 2024) measured outcomes at 4 to 12 weeks. There is no study showing that adopting an anti-inflammatory diet produces measurable sleep changes within days. A minimum of one month appears necessary for detectable effects, and the prospective data suggests that sustained dietary change over months to years produces the most consistent associations. People who change their diet and expect improved sleep by the following week will be disappointed — setting realistic expectations reduces the chance of abandoning a strategy that does have long-term evidence behind it.
Related Reading
- Inflammatory Sleep Disruption: The Immune-Driven Pathway Behind Fragmented Sleep
- Histamine and 3am Waking
- Histamine Intolerance and Sleep
- Why Is Inflammation Worse at Night?
- Leaky Gut and Insomnia
- Brain Fog and Poor Sleep: The Neuroinflammation Connection
- The Glymphatic System and Sleep
- Chronic Inflammation and Insomnia
- Inflammaging and Sleep
- Gut Microbiome and Sleep Inflammation
- Omega-3 and Sleep Quality
- What Is Autoimmune Insomnia and Why Does It Resist Standard Sleep Approaches?
- Can Inflammation Cause 3am Wakeups?
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- Can Chronic Stress Cause Insomnia Through Inflammation?
References
1. Arab, A., Karimi, E., Garaulet, M., & Scheer, F. A. J. L. (2024). Dietary patterns and insomnia symptoms: A systematic review and meta-analysis. Sleep Medicine Reviews, 75, 101936. https://pubmed.ncbi.nlm.nih.gov/38714136/
2. Farrell, E. T., Wirth, M. D., McLain, A. C., Hurley, T. G., Shook, R. P., Hand, G. A., Hebert, J. R., & Blair, S. N. (2023). Associations between the Dietary Inflammatory Index and sleep metrics in the Energy Balance Study (EBS). Nutrients, 15(2), 419. https://pubmed.ncbi.nlm.nih.gov/36678290/
3. Farrell, E. T., Hebert, J. R., Heflin, K., Davis, J. E., Turner-McGrievy, G. M., & Wirth, M. D. (2024). Dietary inflammatory index (DII) and sleep quality, duration, and timing: A systematic review. Sleep Medicine Reviews, 77, 101964. https://pubmed.ncbi.nlm.nih.gov/38833836/
4. Godos, J., Ferri, R., Lanza, G., Caraci, F., Rojas Vistorte, A. O., Yelamos Torres, V., Grosso, G., & Castellano, S. (2024). Mediterranean diet and sleep features: A systematic review of current evidence. Nutrients, 16(2), 282. https://pubmed.ncbi.nlm.nih.gov/38257175/
5. Jiang, J., Huang, S., Yao, W., Yuan, Y., Huang, T., & Xia, Z. (2025). Exploring the link between dietary inflammatory index and sleep disorders: Insights from NHANES and Mendelian randomization approach. Medicine (Baltimore), 104(27), e43170. https://pubmed.ncbi.nlm.nih.gov/40629578/
6. Kase, B. E., Liu, J., Wirth, M. D., Shivappa, N., & Hebert, J. R. (2021). Associations between dietary inflammatory index and sleep problems among adults in the United States, NHANES 2005-2016. Sleep Health, 7(2), 273-280. https://pubmed.ncbi.nlm.nih.gov/33071202/
7. Shimizu, K., Kuramochi, Y., & Hayamizu, K. (2024). Effect of omega-3 fatty acids on sleep: A systematic review and meta-analysis of randomized controlled trials. Journal of Clinical Biochemistry and Nutrition, 75(3), 204-212. https://pubmed.ncbi.nlm.nih.gov/39583980/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
