You are so tired your body aches, but the moment you lie down your brain refuses to switch off. This happens across chronic health conditions and acute illness alike, and it has a neurochemical explanation. It is not a willpower problem, and it is not anxiety. It is a direct consequence of how the inflammatory immune response reorganizes brain chemistry. Two prostaglandins — PGD2 and PGE2 — pull the brain in opposite directions at the same time. Inflammatory cytokines (immune-messaging proteins including TNF-alpha and IL-1beta) reshape when and how the brain receives sleep and wake inputs. This article covers the prostaglandin paradox, the cytokine contribution, and the feedback loop that makes the pattern self-perpetuating. For the broader inflammatory sleep picture, see Inflammatory Sleep Disruption.
Why Does Inflammation Cause Fatigue?
During inflammatory states, PGD2 is produced in the arachnoid membrane (one of the protective membranes surrounding the brain) and the choroid plexus (the tissue that produces cerebrospinal fluid) by an enzyme called lipocalin-type PGD synthase. From there, PGD2 is released into the cerebrospinal fluid, where it circulates through the ventricular and subarachnoid spaces. When PGD2 reaches the basal forebrain, it binds DP1 receptors and triggers local release of adenosine — a paracrine molecule that accumulates during wakefulness and drives sleep pressure. This adenosine activates A2A receptors on VLPO (ventrolateral preoptic area) sleep neurons, which then send inhibitory GABAergic projections to the tuberomammillary nucleus, suppressing its histamine-driven arousal output (Urade & Hayaishi, 2011). Blocking any step in this cascade — with PGD synthase inhibitors, DP1 antagonists, or caffeine (an adenosine A2A receptor antagonist) — reduces both NREM and REM sleep, which confirms the pathway is required for normal sleep.
The subjective experience is deep, pervasive tiredness. But PGD2 is only half the story.
Inflammatory cytokines — TNF-alpha and IL-1beta — amplify the fatigue through a separate route: sickness behavior, an evolutionarily conserved neuroimmune adaptation where the immune response reorganizes physiology to prioritize pathogen defense (Zhang et al., 2025). In people with chronic insomnia, the circadian rhythm of these cytokines moves from its normal nighttime peak to daytime hours. Vgontzas et al. (2002) measured this displacement in 11 chronic insomniacs versus 11 healthy controls: both IL-6 and TNF-alpha release moved to daytime, producing inflammatory fatigue at hours when the brain should be alert. TNF-alpha, which normally promotes NREM sleep at night, instead promotes sleepiness at the wrong time — while contributing to nighttime arousal. The result is a timing mismatch: fatigue inputs arrive during the day, and arousal inputs block sleep at night.
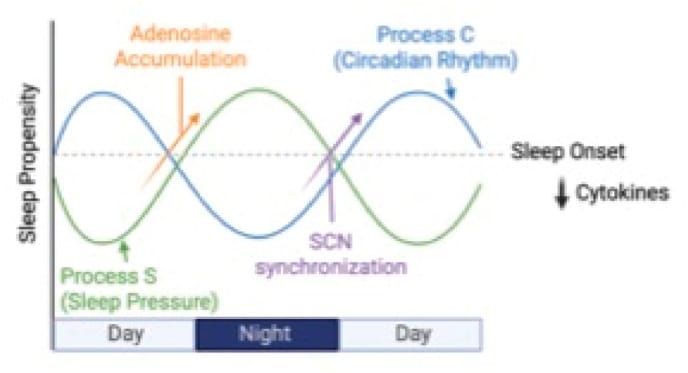
Why Does Inflammation Also Prevent Sleep?
While PGD2 pushes the brain toward sleep, PGE2 does the opposite. Huang et al. (2003) perfused PGE2 into the tuberomammillary nucleus of rats at doses of 100, 200, and 400 pmol/min for two hours. The result was a dose-dependent increase in histamine release in both the medial preoptic area and the frontal cortex, accompanied by increased wakefulness and suppression of both NREM and REM sleep. Of the four PGE2 receptor subtypes (EP1 through EP4), only the EP4 agonist replicated this wake-promoting effect, and EP4 mRNA was identified in histaminergic neurons of the tuberomammillary nucleus.
The anatomical separation matters. PGE2’s wake-promoting action occurs in the posterior hypothalamus (the tuberomammillary nucleus), while its fever-inducing action occurs in the anterior preoptic area. Arousal and fever are mechanistically separable consequences of the same inflammatory mediator — a person can experience PGE2-driven wakefulness independent of fever.
This is what makes the paradox neurochemically precise: PGE2 activates the same histaminergic neurons that PGD2 works to suppress through the VLPO inhibition cascade. During inflammation, both prostaglandins are elevated simultaneously from the same arachidonic acid precursor. The sleep-promoting arm and the wake-promoting arm are both running at once.
TNF-alpha and IL-1beta add a second dimension to the wakefulness side. Zhang et al. (2025) documented that these cytokines promote NREM sleep pressure while simultaneously activating arousal pathways — corticotropin-releasing hormone (CRH), norepinephrine, and histamine. The effect is fragmented sleep architecture: the brain accumulates NREM pressure but cannot consolidate it into sustained sleep episodes. IL-6, when displaced to nighttime in inflammatory states, flattens the consolidation window that normally supports uninterrupted sleep (Vgontzas et al., 2002).
What Creates the “Exhausted But Unable to Sleep” State?
Both prostaglandins are synthesized from the same arachidonic acid pool. During inflammation, COX enzymes (cyclooxygenase-1 and cyclooxygenase-2) convert arachidonic acid into both PGD2 and PGE2 in increased quantities. The sleep-promoting and wake-promoting arms of the prostaglandin response escalate together — creating a state where the harder the body pushes toward sleep, the harder the brain pushes back toward wakefulness (Urade & Hayaishi, 2011).
The human evidence for this paradox comes from Raison et al. (2010), who studied 31 participants — 19 receiving chronic interferon-alpha therapy (which induces sustained inflammatory cytokine activation through TNF-alpha, IL-6, and NF-kB) and 12 controls. Polysomnography showed that those receiving interferon-alpha developed increased wake after sleep onset (WASO), decreased Stage 3/4 slow-wave sleep, reduced sleep efficiency, and extended REM latency. Their sleep was objectively fragmented and non-restorative. At the same time, fatigue scores rose during the interferon-alpha course — these participants were measurably more tired. Yet when given standardized daytime nap opportunities, the interferon-alpha group showed reduced propensity to fall asleep compared to controls. They were objectively more fatigued and objectively less able to sleep. The paradox was measured directly: deep tiredness coexisting with a brain that would not permit sleep.
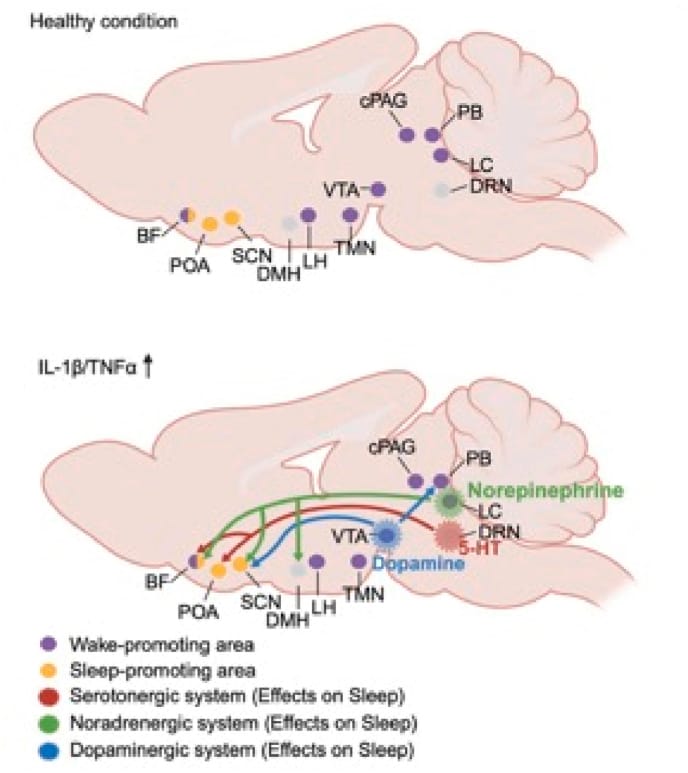
This is not a psychological phenomenon. The inflammatory immune response reorganizes sleep architecture during infection as a conserved survival mechanism — redirecting resources toward pathogen defense at the cost of sleep consolidation (Zhang et al., 2025). When inflammation becomes chronic, that reorganization persists without resolution.
The feedback loop makes it worse. Haack et al. (2009) measured urinary PGE2 metabolite levels in 15 participants undergoing 88 hours of total sleep deprivation, compared to 9 controls. By the third night without sleep, PGE2 metabolite rose by approximately 30%. Sleep loss elevates the prostaglandin that blocks sleep. Each sleepless night raises PGE2, which drives histaminergic wakefulness, which prevents the next night’s sleep — a cycle that feeds itself.
Does Sleep Loss Make Inflammation-Induced Fatigue Worse?
Zhang et al. (2023) conducted a review and Mendelian randomization meta-analysis — a method that uses genetic variants as instruments to establish causal direction — across 44 publications and 51,879 participants. Chronic insomnia participants showed elevated serum CRP (C-reactive protein), IL-1beta, IL-6, and TNF-alpha compared to healthy controls. The Mendelian randomization analysis found that genetically elevated CRP and IL-6 causally alter sleep duration phenotypes. This goes beyond correlation — the genetic evidence establishes that inflammatory proteins influence sleep architecture.
The relationship between inflammatory markers and sleep follows a U-shaped curve: both short and long sleep associate with elevated inflammation, but through different cytokine profiles. Daytime sleepiness alone did not show the same inflammatory differentiation, which is consistent with the prostaglandin paradox — inflammation drives insomnia, not drowsiness. IL-1beta showed the strongest association with insomnia severity, consistent with its known role in activating CRH neurons and the HPA (hypothalamic-pituitary-adrenal) axis, which promotes arousal (Zhang et al., 2023).
Meanwhile, each night of poor sleep raises PGE2 (the wake-promoting prostaglandin), increases NF-kB (nuclear factor kappa-B, a transcription factor that drives inflammatory gene expression) activation, and displaces cytokine rhythms further from their normal nighttime alignment (Haack et al., 2009). The person who cannot sleep because of inflammation develops worse inflammation because they cannot sleep. Each cycle elevates PGE2 further, strengthens histaminergic arousal, and deepens the displacement of TNF-alpha and IL-6 from their nighttime peak.
Breaking this cycle requires addressing the inflammation. Sedation does not resolve the prostaglandin imbalance or restore displaced cytokine rhythms — it forces the brain into a sleep state while leaving the inflammatory drive unchanged. The PGD2/PGE2 ratio, the cytokine circadian alignment, and the NF-kB activation state all persist through pharmacological sedation.
Many people have more than one cause contributing to their sleep disruption. The exhausted-but-unable-to-sleep pattern might be driven by inflammatory prostaglandins, but it might also be shaped by metabolic, hormonal, or autonomic contributors running in parallel. Identifying which causes might be active in your case is a useful next step.
Find out which causes might be driving your 3am wakeups
Frequently Asked Questions
Does Ibuprofen Affect Sleep Through Prostaglandins?
Ibuprofen and other non-selective NSAIDs block both COX-1 and COX-2, reducing the synthesis of both sleep-promoting PGD2 and wake-promoting PGE2. Some people report easier sleep onset after taking ibuprofen — consistent with PGE2 reduction lowering arousal. Others report lighter or more disrupted sleep — consistent with PGD2 reduction weakening the sleep drive. COX-2 selectivity changes this equation: COX-2-selective inhibitors preferentially reduce PGE2 (which is upregulated during inflammation through COX-2 induction) while preserving more constitutive PGD2 production. The practical takeaway is that NSAID effects on sleep are not uniform — they depend on whether PGD2 or PGE2 is the dominant prostaglandin in a given person’s inflammatory state. See Why Does Inflammation Cause Fatigue? and Why Does Inflammation Also Prevent Sleep? for the underlying mechanisms.
Why Do Non-Steroidal Anti-Inflammatory Drugs Affect Sleep Quality?
Taking ibuprofen before bed may reduce pain-related arousal — which can improve sleep for people whose waking is driven by nociceptive input — but it simultaneously weakens the brain’s PGD2-to-adenosine sleep-promoting cascade. The net result depends on whether the pain reduction or the sleep drive reduction has the larger effect. COX-2-selective inhibitors may preserve more of the PGD2 sleep pathway because COX-2 preferentially drives the inflammatory PGE2 production that is elevated during illness. This is why NSAID effects on sleep are not reducible to a “helps or hurts” answer — the same drug suppresses both sides of the prostaglandin balance described throughout this article.
Why Do You Feel Exhausted But Cannot Sleep When You Are Sick?
During acute infection, TNF-alpha, IL-1beta, and IL-6 all rise to concentrations that would be unusual in chronic conditions. This cytokine surge drives both PGD2 production (through increased arachidonic acid metabolism) and PGE2 production simultaneously. The sickness behavior program — fatigue, muscle ache, withdrawal from activity, reduced appetite — is an evolutionarily conserved response that redirects metabolic resources toward immune function (Zhang et al., 2025). At the same time, PGE2 activates histaminergic arousal neurons, and arousal-pathway cytokines maintain wakefulness to support immune surveillance. The subjective experience — feeling physically destroyed but lying awake for hours — is the acute, self-limiting version of the same PGD2/PGE2 paradox that becomes chronic when sustained inflammatory conditions prevent resolution.
Related Reading
- Inflammatory Sleep Disruption — the full cause overview covering all inflammatory mechanisms
- Why Does Histamine Wake You Up at 3am? — histaminergic arousal and the mast cell circadian peak
- Can Histamine Intolerance Cause Sleep Problems? — histamine degradation, DAO deficiency, and sleep disruption
- Why Is Inflammation Worse at Night? — the NF-kB circadian rhythm and nocturnal inflammatory vulnerability
- How Does Leaky Gut Affect Sleep? — gut permeability, LPS translocation, and neuroinflammation
- Does Poor Sleep Cause Brain Inflammation? — neuroinflammation, brain fog, and sleep loss
- How Does the Glymphatic System Work During Sleep? — waste removal and deep sleep dependency
- What Is the Connection Between Chronic Inflammation and Insomnia? — the bidirectional cytokine-insomnia cycle
- Why Do Men Sleep Worse After 50?
- What Is Autoimmune Insomnia and Why Does It Resist Standard Sleep Approaches?
- Can Inflammation Cause 3am Wakeups?
- Can Chronic Stress Cause Insomnia Through Inflammation?
- Gut Bacteria and Insomnia: Which Microbes Affect Your Sleep (and Which Ones Help)
- Does an Anti-Inflammatory Diet Improve Sleep?
References
1. Haack, M., Lee, E., Cohen, D., & Mullington, J. M. (2009). Activation of the prostaglandin system in response to sleep loss in healthy humans: Potential mediator of increased spontaneous pain. Pain, 145(1-2), 136-141. https://pubmed.ncbi.nlm.nih.gov/19560866/
2. Huang, Z.-L., Sato, Y., Mochizuki, T., Okada, T., Qu, W.-M., Yamatodani, A., Urade, Y., & Hayaishi, O. (2003). Prostaglandin E2 activates the histaminergic system via the EP4 receptor to induce wakefulness in rats. Journal of Neuroscience, 23(14), 5975-5983. https://pubmed.ncbi.nlm.nih.gov/12853415/
3. Raison, C. L., Rye, D. B., Woolwine, B. J., Vogt, G. J., Bautista, B. M., Spivey, J. R., & Miller, A. H. (2010). Chronic interferon-alpha administration disrupts sleep continuity and depth in patients with hepatitis C: Association with fatigue, motor slowing, and increased evening cortisol. Biological Psychiatry, 68(10), 942-949. https://pubmed.ncbi.nlm.nih.gov/20537611/
4. Urade, Y., & Hayaishi, O. (2011). Prostaglandin D2 and sleep/wake regulation. Sleep Medicine Reviews, 15(6), 411-418. https://pubmed.ncbi.nlm.nih.gov/22024172/
5. Vgontzas, A. N., Zoumakis, M., Papanicolaou, D. A., Bixler, E. O., Prolo, P., Lin, H.-M., Vela-Bueno, A., Kales, A., & Chrousos, G. P. (2002). Chronic insomnia is associated with a shift of interleukin-6 and tumor necrosis factor secretion from nighttime to daytime. Metabolism, 51(7), 887-892. https://pubmed.ncbi.nlm.nih.gov/12077736/
6. Zhang, Y., Zhao, W., Liu, K., Chen, Z., Fei, Q., Ahmad, N., & Yi, M. (2023). The causal associations of altered inflammatory proteins with sleep duration, insomnia and daytime sleepiness. Sleep, 46(10), zsad207. https://pubmed.ncbi.nlm.nih.gov/37535878/
7. Zhang, N., Park, K., Chung, S., & Yim, Y. S. (2025). IL-1beta and TNF-alpha-driven sleep alterations: Neuroimmune mechanisms and behavioral implications. Brain, Behavior, & Immunity — Health, 50, 101139. https://pubmed.ncbi.nlm.nih.gov/41323350/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
