People with autoimmune conditions describe a frustration that follows a recognizable pattern: “I can’t heal unless I sleep, but I can’t sleep because I haven’t healed yet.” They have tried sleep hygiene, melatonin, magnesium, and sometimes CBT-I — and hit a wall. The assumption is usually that pain or medication is causing the insomnia. The research points to a different primary driver: the cytokines themselves act on sleep-regulatory circuits in the brain, independently of pain.
This article covers the cytokine-mediated mechanism behind autoimmune insomnia, the evidence that blocking cytokines repairs sleep before disease activity improves, and which autoimmune conditions carry the highest insomnia risk. For the broader overview of how inflammation fragments sleep, see the parent article: Inflammatory Sleep Disruption.
How Do Autoimmune Diseases Cause Insomnia?
The assumption that autoimmune insomnia is caused by pain makes intuitive sense — inflamed joints hurt, and pain disrupts sleep. But the polysomnography data tells a different story. The cytokines elevated in autoimmune disease reshape sleep architecture through direct neurological action, separate from any pain pathway.
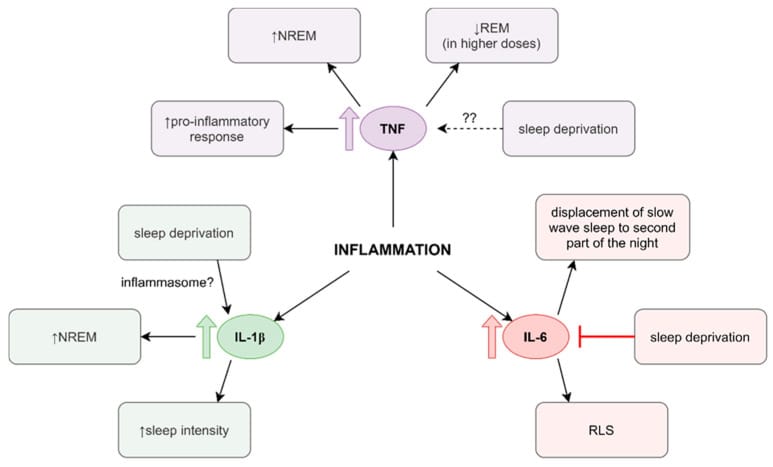
TNF-alpha, IL-6, and IL-1beta are the three cytokines at the center of this mechanism. At the chronic low-grade concentrations present in rheumatoid arthritis, lupus, and multiple sclerosis, these molecules produce fragmented, non-restorative sleep rather than the consolidated deep sleep seen during acute infection (Zhang et al., 2025). During a short-term illness, the same cytokines promote deep NREM sleep to support immune recovery. But in chronic autoimmune disease, where cytokine elevation is sustained for months or years, this adaptive mechanism becomes pathological.
The neuroscience maps to each cytokine individually. IL-1beta suppresses serotonergic neuronal firing in the dorsal raphe nucleus (a brainstem region that regulates sleep-wake transitions) while enhancing serotonin release in sleep-promoting hypothalamic regions — a push-pull disruption that destabilizes sleep architecture from two directions. TNF-alpha modulates noradrenergic and dopaminergic arousal circuits, increasing NREM depth at moderate concentrations but increasing fragmentation at the chronically elevated concentrations found in autoimmune disease (Zhang et al., 2025).
The strongest human evidence for this mechanism comes from a polysomnography study by Bjurstrom et al. (2017). They studied 72 participants — 24 with rheumatoid arthritis and 48 matched controls — using overnight sleep recordings combined with monocytic cytokine assays taken in the evening and morning. The results were directional and cytokine-dependent:
- RA participants showed lower sleep efficiency than controls (83.8% vs. 88.1%).
- Evening TNF production correlated with sleep maintenance that night — the cytokine measured before bed predicted how well sleep held together overnight.
- Evening IL-6 predicted increased slow-wave sleep (the deepest stage of NREM), which in turn counteracted morning inflammatory upswings.
- The cytokine-sleep relationships were bidirectional — IL-6 and TNF operated through distinct temporal windows and produced opposing downstream effects.
The Bjurstrom findings demonstrate that the inflammatory cycle — not pain — is the primary driver of sleep fragmentation architecture in rheumatoid arthritis. Evening cytokine concentrations predicted that night’s sleep structure in ways that pain scores did not match.
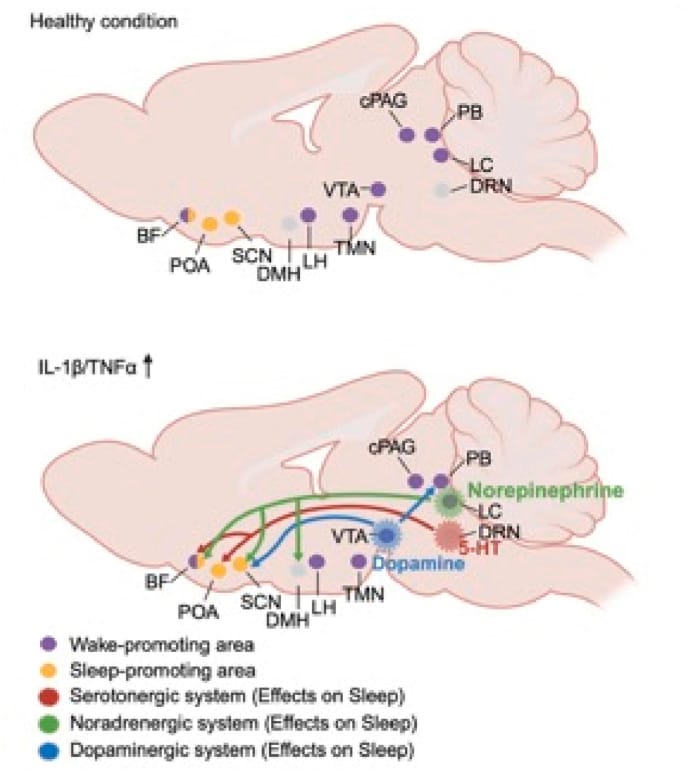
The evolutionary context explains why this happens. These cytokine-sleep interactions are an adaptive host-defense response — during acute infection, IL-1beta and TNF-alpha promote deep NREM sleep to support immune recovery. In chronic autoimmune disease, where cytokine elevation is sustained rather than temporary, this adaptive mechanism never turns off. The brain keeps receiving pro-inflammatory input that would normally drive deep sleep, but the chronic nature of the exposure disrupts the circuits that input acts on, producing fragmentation instead of consolidation (Zhang et al., 2025).
Can Blocking Inflammatory Cytokines Improve Autoimmune Insomnia?
This is the section that moves the autoimmune insomnia argument from correlation to mechanism. If blocking a single cytokine receptor repairs sleep before the disease itself improves, then the cytokine is driving the insomnia directly.
Fragiadaki et al. (2012) studied 15 people with rheumatoid arthritis and confirmed sleep disturbances. Each received six monthly infusions of tocilizumab at 8 mg/kg — tocilizumab is an IL-6 receptor antagonist, meaning it blocks IL-6 from binding to its receptor throughout the body, including in the brain. The sleep results arrived early:
- Sleep quality improved and daytime sleepiness decreased at the first-month assessment — before measurable reductions in disease activity (DAS-28 scores) occurred.
- Sleep improvements did not correlate with changes in DAS-28 scores, ruling out pain relief as the mediating mechanism.
- Sleep improvements did correlate with improvements in functional disability (HAQ scores), suggesting IL-6 acts on sleep through functional neurological pathways separate from joint inflammation.
The mechanistic explanation is direct: IL-6 crosses the blood-brain barrier and acts on hypothalamic sleep-regulatory circuits. When tocilizumab blocks IL-6 receptor binding, it interrupts this neuroimmune pathway. The sleep improvement appearing before disease improvement demonstrates that pain relief was not driving better sleep — disease activity would have needed to decrease first.
This finding is not isolated. Ditmer et al. (2022) documented a broader pattern: anti-cytokine biologics (anti-TNF, anti-IL-6) improve sleep quality in rheumatoid arthritis and inflammatory bowel disease beyond what pain reduction or disease activity improvement explains. The sleep benefit appears to be a direct neurological effect of reducing cytokine action on brain circuits, not an indirect consequence of feeling better.
The implication is direct. Blocking a single cytokine receptor repairs sleep — before the underlying disease improves — meaning the immune response is driving the insomnia through a mechanism that is independent of pain, disability, or psychological distress.
Which Autoimmune Conditions Carry the Highest Insomnia Risk?
The general population insomnia rate falls between 10.5% and 22.6%. In every major autoimmune condition studied, the rate is two to seven times higher — and the excess risk persists after accounting for known confounders like pain, medication use, and depression.
Rheumatoid arthritis carries an insomnia prevalence of 25.6% to 70.9% (Ditmer et al., 2022). The range reflects different measurement methods and populations, but the lower bound alone exceeds the general population ceiling. George et al. (2025) provided the largest recent epidemiological evidence: in a population-based study of 1,826 participants (913 with RA, 913 matched comparators), RA participants had a 34% increased hazard of developing any sleep disorder (HR 1.34; 95% CI 1.11-1.61). The risk of insomnia was also elevated 34% (HR 1.34; 95% CI 1.03-1.73). This elevated risk persisted after adjusting for obesity, cardiovascular disease, depression, and anxiety — indicating that RA itself is the independent risk driver, not the comorbidities that accompany it.
Lupus (erythematosus) shows an insomnia prevalence of 33.3% to 71.1% (Ditmer et al., 2022). Faraguna et al. (2024) provided objective data using actigraphy in 73 participants (40 with SLE, 33 healthy controls). The findings revealed a counterintuitive pattern: SLE participants had higher total sleep time (7.55 vs. 6.60 hours) but worse sleep quality — more time in bed, less restorative sleep. Glucocorticoid dose independently correlated with sleep maintenance impairment, while perceived psychological stress was associated with a separate short-sleep phenotype. This means at least two mechanistically separate insomnia types exist in lupus: one driven by medication effects on sleep maintenance, and one driven by stress and immune activation producing shortened sleep.
Multiple sclerosis has a sleep disorder prevalence of approximately 60% — a rate two to five times higher than the general population (Sakkas et al., 2019). The mechanism in MS includes a factor not present in RA or lupus: demyelinating lesions (areas where the protective coating around nerve fibers has been damaged) in hypothalamic and brainstem sleep centers directly disrupt sleep-wake regulatory circuitry, independent of peripheral pain. Elevated TNF-alpha, IL-1beta, and IFN-gamma levels in progressive MS alter sleep homeostasis by acting on adenosine and serotonergic sleep-promoting pathways (Sakkas et al., 2019). People with MS show reduced slow-wave sleep and REM disruption that correlates with fatigue severity independently of disability scores.
The cross-disease pattern is consistent: across rheumatoid arthritis, lupus, and multiple sclerosis, insomnia prevalence exceeds the general population by two to seven times. The mechanism is not explained by pain, medication, or psychological factors alone. Each condition has cytokine-mediated neurological pathways that fragment sleep architecture through direct immune action on the brain.
Why Don’t Standard Sleep Approaches Work Well for Autoimmune Insomnia?
If you have an autoimmune condition and have worked through the standard sleep recommendations without lasting improvement, the research explains why.
CBT-I is the gold standard for primary insomnia. It works by restructuring sleep behavior (sleep restriction, stimulus control) and reducing conditioned hyperarousal — the learned association between bed and wakefulness. In autoimmune insomnia, the hyperarousal is not conditioned. It is driven by cytokines acting on brainstem and hypothalamic circuits. The behavioral approach addresses a different part of the problem. CBT-I may still provide partial benefit by improving the behavioral component of sleep, but it does not reach the immune driver.
Melatonin addresses circadian timing — it helps the brain recognize when it is time to initiate sleep. Magnesium supports GABA-mediated relaxation, reducing neural excitability. Neither addresses the TNF-alpha, IL-6, or IL-1beta acting on sleep-regulatory circuits at the neurological level. These approaches work on mechanisms that are intact in primary insomnia but are being overridden by immune activity in autoimmune insomnia.
Ditmer et al. (2022) documented this mismatch directly: standard insomnia approaches show limited efficacy in autoimmune populations because they do not address the immunological root cause. The sleep disturbances in these conditions are mechanistically distinct from insomnia in the general population. The review authors call for immune-targeted sleep approaches in autoimmune conditions as a distinct category — recognizing that the standard toolkit was developed for a different mechanism.
The direction the research points toward is addressing the inflammatory driver as the primary focus. This is a conversation for a rheumatologist or immunologist who understands both the autoimmune condition and its sleep consequences. The Fragiadaki tocilizumab data shows that biologics and disease-modifying therapies may have sleep benefits beyond their primary indication — meaning the medication managing the autoimmune condition might also be the more effective sleep approach. Discussing sleep outcomes when evaluating or adjusting autoimmune therapy is a step that the research supports.
Autoimmune inflammation might be one contributor to sleep disruption, but it rarely acts alone. Metabolic changes, hormonal fluctuations, circadian misalignment, and autonomic dysregulation might all be active at the same time, each amplifying the others. Identifying which causes might be driving your pattern is a useful next step before deciding where to focus.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can an Overactive Immune Response Cause Sleep Problems?
Bjurstrom et al. (2017) demonstrated this with polysomnography in 72 participants: the cytokine concentrations measured in the evening predicted sleep structure that night with a directionality that pain scores did not match. Evening TNF production correlated with sleep maintenance, and evening IL-6 predicted slow-wave sleep depth. The immune-sleep relationship unfolds on a time scale of hours — not the weeks or months that characterize disease flares.
Does Addressing Insomnia Lower Inflammation?
This does not mean behavioral sleep approaches are irrelevant in autoimmune conditions. They may still improve the behavioral and conditioned components of insomnia. But the research suggests that the immune-to-sleep direction carries more weight in autoimmune populations than the sleep-to-immune direction — blocking IL-6 repaired sleep before disease improved (Fragiadaki et al., 2012), while behavioral approaches alone have not produced equivalent results in these populations (Ditmer et al., 2022).
Can Cytokines Cause Insomnia?
The Zhang et al. (2025) review maps the molecular detail of each pathway. IL-1beta acts through NF-kB and prostaglandin E2 pathways. TNF-alpha modulates adenosine receptor activity. At acute concentrations (during a short-term illness), these cytokines promote consolidated deep sleep. At the chronic low-grade concentrations found in autoimmune disease, the same molecules fragment the sleep architecture they normally protect — a dose-dependent reversal from sleep-promoting to sleep-disrupting.
Related Reading
- Inflammatory Sleep Disruption: How Chronic Inflammation Fragments Your Sleep Architecture — the full cause overview covering all five inflammatory mechanisms
- Histamine and 3am Waking: Why Your Immune System Won’t Let You Sleep at Night
- Histamine Intolerance and Sleep: When Standard Sleep Remedies Make Everything Worse
- Why Is Inflammation Worse at Night? The Body Clock Connection Nobody Explains
- Leaky Gut and Insomnia: How Gut Inflammation Reaches the Brain at Night
- Brain Fog and Poor Sleep: When Neuroinflammation Breaks Both
- The Glymphatic System and Sleep: How Your Brain Detoxes Overnight (and What Goes Wrong)
- Chronic Inflammation and Insomnia: The Cycle That Keeps You Wired at 3am
- Can Inflammation Cause 3am Wakeups? The Science Behind the Viral Question
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- Gut Bacteria and Insomnia: Which Microbes Affect Your Sleep (and Which Ones Help)
- Why Do Men Sleep Worse After 50?
- Can Chronic Stress Cause Insomnia Through Inflammation?
- Does an Anti-Inflammatory Diet Improve Sleep?
References
Bjurstrom, M. F., Olmstead, R., & Irwin, M. R. (2017). Reciprocal relationship between sleep macrostructure and evening and morning cellular inflammation in rheumatoid arthritis. Psychosomatic Medicine, 79(1), 24-33. https://pubmed.ncbi.nlm.nih.gov/27428854/
Ditmer, M., Gabryelska, A., Turkiewicz, S., Bialasiewicz, P., Malecka-Wojciesko, E., & Sochal, M. (2022). Sleep problems in chronic inflammatory diseases: Prevalence, treatment, and new perspectives: A narrative review. Journal of Clinical Medicine, 11(1), 67. https://pubmed.ncbi.nlm.nih.gov/35011807/
Faraguna, U., Porciani, C., Colitta, A., Bruno, S., Frumento, P., Stagnaro, C., Tani, C., Vagelli, R., & Mosca, M. (2024). Actigraphic and self-reported characterization of sleep in systemic lupus erythematosus patients. Rheumatology (Oxford), 63(4), 1076-1083. https://pubmed.ncbi.nlm.nih.gov/37432350/
Fragiadaki, K., Tektonidou, M. G., Konsta, M., Chrousos, G. P., & Sfikakis, P. P. (2012). Sleep disturbances and interleukin 6 receptor inhibition in rheumatoid arthritis. Journal of Rheumatology, 39(1), 60-62. https://pubmed.ncbi.nlm.nih.gov/22133618/
George, R. J., Kumar, R., Achenbach, S. J., Lovering, E., Lennon, R. J., Davis, J. M. 3rd, Carvalho, D. Z., Crowson, C. S., & Myasoedova, E. (2025). Sleep disorders in rheumatoid arthritis: Incidence, risk factors and association with dementia. Seminars in Arthritis and Rheumatism, 73, 152722. https://pubmed.ncbi.nlm.nih.gov/40245587/
Sakkas, G. K., Giannaki, C. D., Karatzaferi, C., & Manconi, M. (2019). Sleep abnormalities in multiple sclerosis. Current Treatment Options in Neurology, 21(1), 4. https://pubmed.ncbi.nlm.nih.gov/30701337/
Zhang, N., Park, K., Chung, S., & Yim, Y. S. (2025). IL-1beta and TNF-alpha-driven sleep alterations: Neuroimmune mechanisms and behavioral implications. Brain, Behavior, & Immunity – Health, 50, 101139. https://pubmed.ncbi.nlm.nih.gov/41323350/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
