If you have a chronic inflammatory condition — rheumatoid arthritis, autoimmune disease, persistent pain — you may have noticed that your worst hours are not during the day. Joint stiffness is worst on waking. Pain intensifies in the second half of the night. Sleep fragments at the same hours, not at unpredictable times.
This is not coincidental. Immunity runs on a 24-hour clock, and inflammatory activity peaks in the late-night and early-morning hours. The molecular reasons for this involve the interaction between cortisol, the transcription factor NF-κB, and the core circadian clock protein BMAL1 — a relationship that, once disrupted, reinforces itself. Inflammation is one of several causes of 3am waking; metabolic, autonomic, hormonal, and circadian factors can produce the same pattern through different mechanisms.
This article covers the inflammatory mechanism. For the broader overview of how inflammation disrupts sleep architecture, see the parent article: Inflammatory Sleep Disruption.
Why Does Inflammation Get Worse at Night?
Cortisol is the body’s primary endogenous anti-inflammatory molecule. It suppresses NF-κB — the transcription factor that activates genes encoding pro-inflammatory cytokines like IL-6, TNF-α, and IL-1β. During daytime hours, cortisol concentrations are high enough to keep NF-κB activity restrained.
But cortisol follows a circadian rhythm. It peaks in the early morning (around 6-8am), declines through the day, and reaches its lowest concentration between approximately 2am and 4am (Srinivasan & Walker, 2022). At that trough, NF-κB is no longer suppressed.
In healthy people, this overnight NF-κB rise is moderate — part of normal immune maintenance. But in people with chronic inflammation, the rise overshoots. NF-κB activates more inflammatory gene transcription than normal, producing elevated concentrations of cytokines during the pre-dawn hours.
This was first documented in humans in 1994. Arvidson et al. measured serum IL-6 across 24-hour cycles in 48 individuals with rheumatic diseases and 10 healthy controls. IL-6 was undetectable in the healthy controls but elevated in those with rheumatoid arthritis, with concentrations peaking in the early morning hours. The temporal pattern correlated directly with the severity of morning stiffness — worst at the time of highest cytokine load (Arvidson et al., 1994).
More recent data shows the relationship works in the other direction too. Lee and Park (2024) reported that three nights of inadequate sleep in healthy human subjects caused IL-6 mRNA to increase three-fold and TNF-α mRNA to increase two-fold. Poor sleep itself amplifies the overnight inflammatory load, which then fragments sleep further.
Does Immunity Follow a Circadian Rhythm?
The circadian regulation of immunity is not limited to cortisol. Wang et al. (2022) reviewed the circadian architecture of immune function and found that clock genes are expressed in virtually every immune cell lineage — T cells, B cells, macrophages, neutrophils, natural killer cells. Each has its own internal clock, and the timing of their activity follows a predictable 24-hour pattern.
Leukocyte trafficking — the movement of immune cells through the bloodstream and into tissues — peaks at night. Circulating immune cell numbers are highest during sleep hours. This means immune mobilization is at its peak at the same time cortisol-mediated suppression is lowest (Lee & Park, 2024).
One of the more detailed mechanisms comes from Tang et al. (2022), who studied nocturnal asthma — a condition where airway inflammation worsens at night. They found that BMAL1 (the core circadian clock protein) suppresses IL-6 production in airway epithelial cells by upregulating a transcription factor called FOXA2. When BMAL1 is depleted, IL-6 increases and loses its normal circadian oscillation.
In their study cohort, asthmatic individuals with nocturnal flares had serum IL-6 concentrations of 7.45 ± 6.57 pg/mL, compared to 2.81 ± 1.49 pg/mL in those without nighttime episodes — a 2.6-fold difference. The molecular pathway is: reduced BMAL1 → reduced FOXA2 → unchecked IL-6 production → nocturnal inflammatory flares.
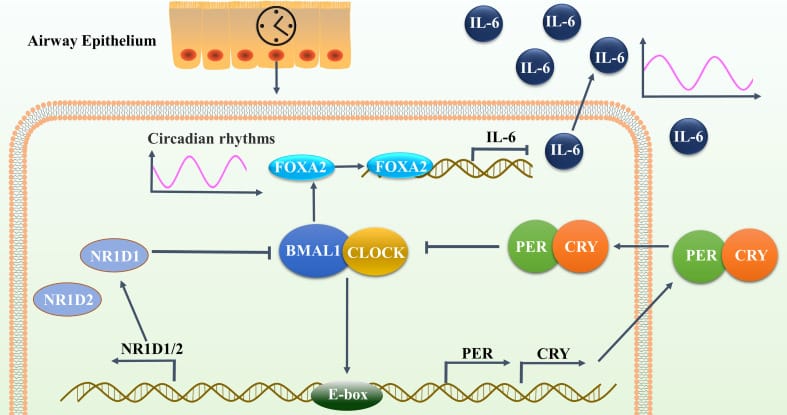
How Does Inflammation Disrupt the Body Clock?
This is where the relationship between inflammation and the body clock becomes self-reinforcing.
Hong et al. (2018) showed that NF-κB is not just modulated by the clock — it is necessary for the clock to function. In transgenic mice, removing NF-κB activity impaired rhythmic activity patterns and altered core clock gene expression. But when NF-κB was chronically activated (as it is in persistent inflammation), it redirected the clock’s machinery toward inflammatory gene transcription.
Here is what happens at the molecular level: NF-κB activation causes CLOCK/BMAL1 protein complexes to relocate across the genome to NF-κB-bound sites. Instead of driving the normal circadian gene expression program (Period, Cryptochrome, Rev-erb — the genes that form the clock’s negative feedback loop), CLOCK/BMAL1 is redirected toward inflammatory gene transcription. The clock’s negative feedback loop — the cycle that keeps circadian timing on schedule — is suppressed.
Hong et al. also found that a high-fat diet triggered NF-κB activation patterns comparable to those seen with bacterial lipopolysaccharide (LPS), suggesting that metabolic stress disrupts circadian timing through this same pathway.
The consequence: once chronic inflammation activates NF-κB, NF-κB disrupts the circadian clock, the disrupted clock can no longer time immune responses properly, and unscheduled immune activity generates more NF-κB. The loop sustains itself.
Pu et al. (2025) provided pharmacological confirmation of this relationship. They developed a small molecule (Core Circadian Modulator, or CCM) that targets the PASB domain of BMAL1, restoring its transcriptional activity. In macrophages, CCM produced dose-dependent downregulation of inflammatory and phagocytic pathways — demonstrating that BMAL1 activity directly controls macrophage inflammatory output. When NF-κB suppresses BMAL1 at night, the result is the inverse: elevated macrophage-driven inflammation (Pu et al., 2025).
Does Cortisol Block Inflammation at Night?
The cortisol-NF-κB relationship is documented across both human and animal literature. Cortisol induces a protein called GILZ (glucocorticoid-induced leucine zipper), which prevents the NF-κB subunit p65 from translocating into the nucleus and activating inflammatory genes. When cortisol is high, GILZ keeps p65 out. When cortisol drops, GILZ expression falls and p65 is free to enter the nucleus and drive transcription of IL-6, TNF-α, and IL-1β (Srinivasan & Walker, 2022).
There is also a second factor at this time point. The CLOCK protein acetylates glucocorticoid receptors, reducing their ability to suppress inflammation. This means that even the cortisol that is present at the overnight nadir is less effective — its receptors are less responsive to it. The combination of low cortisol concentration and reduced receptor sensitivity compounds the effect (Srinivasan & Walker, 2022).
Kikyo (2023) reviewed the evidence for this window in rheumatoid arthritis. RA morning stiffness correlates with elevated pro-inflammatory cytokines and melatonin in the early morning hours, while cortisol remains insufficient to suppress them until later in the morning. Joint damage markers (TNF-α, IL-1, IL-6) all show circadian expression patterns, meaning that the rate of joint destruction is not constant — it is highest during the nocturnal inflammatory peak.
This understanding has led to chronotherapy — timing medication to the inflammatory window rather than giving it at standard intervals. Modified-release prednisone administered at bedtime (so that drug concentrations peak at the 2-4am cytokine surge) has demonstrated effectiveness in RA, validating the nocturnal inflammatory window as a measurable, targetable phenomenon (Kikyo, 2023).
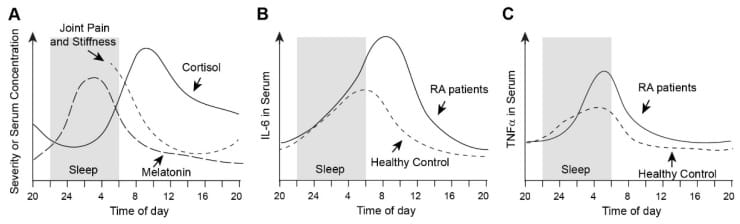
The circadian inflammatory pattern described above might be one of several causes contributing to disrupted sleep. Inflammatory and circadian dysregulation can compound with autonomic, metabolic, or hormonal factors — meaning multiple mechanisms might be active at the same time. Identifying which causes might be driving your pattern is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Do Cytokines Peak at Night?
The circadian pattern of cytokine production is documented across multiple inflammatory conditions. Arvidson et al. (1994) first showed this in RA: IL-6 follows a consistent overnight rise that persists even after corticosteroid administration reduces overall concentrations. The rhythm is maintained — the entire curve is lower, but the timing of the peak does not change.
TNF-α follows a similar pattern, though with different peak timing depending on the condition. In RA, both IL-6 and TNF-α contribute to the early-morning stiffness window. The cytokine peak is a temporal pattern with a defined molecular driver: the NF-κB/cortisol interaction described above.
Why Does Arthritis Pain Get Worse at Night?
Kikyo (2023) reviewed this relationship in the context of joint destruction. The osteoclastogenesis inducers — TNF-α, IL-1, and IL-6 — all show circadian expression patterns. This means that the activity of osteoclasts (the cells that break down bone tissue in RA) is not constant across the day. It is highest when these cytokines peak, during the late-night and early-morning hours.
This is why people with RA consistently report that stiffness and pain are worst upon waking and improve through the morning as cortisol rises. The inflammatory load in the joints tracks the same molecular clock that governs NF-κB and cortisol across the rest of the body.
Does Melatonin Reduce Inflammation?
Melatonin’s interaction with inflammation is concentration-dependent and cell-type-dependent. At physiological concentrations, it tends to reduce NF-κB nuclear translocation and lower cytokine output. But melatonin also peaks during the same overnight hours when cytokines are elevated — meaning it coexists with, rather than prevents, the nocturnal inflammatory window.
In RA, Kikyo (2023) noted that melatonin is elevated alongside pro-inflammatory cytokines in the early morning hours. Whether supplemental melatonin helps or worsens nocturnal inflammation depends on the underlying condition, the dose, and which immune pathways are dominant. This is not a question with a universal answer.
Does Disrupted Sleep Cause Immune Impairment?
Lee and Park (2024) reviewed the human evidence for this bidirectional relationship. The three-night sleep restriction data (three-fold IL-6 increase, two-fold TNF-α increase) shows how rapidly sleep loss amplifies inflammatory load. But the chronic data tells a different story: in people with chronic insomnia, the IL-6 secretion peak can redistribute from 4am to 7pm, meaning the circadian timing of immune activity is no longer aligned with the sleep-wake cycle.
NF-κB is involved on both sides of this relationship. It activates the transcription of pro-inflammatory cytokines (IL-1β, TNF-α) that also function as sleep-regulatory substances — they promote NREM sleep duration. Immune function relies on sleep as a repair and consolidation window. When NF-κB is chronically overactivated, it disrupts the sleep it needs to resolve the inflammation it is producing.
Related Reading
- Inflammatory Sleep Disruption — the full cause overview covering all five inflammatory mechanisms
- Why Does Histamine Wake You Up at 3am? — how brain-resident mast cells and the circadian histamine peak drive early-morning waking
- Can Histamine Intolerance Cause Sleep Problems? — how DAO deficiency and mast cell activation compound histamine-driven insomnia
- How Does Leaky Gut Affect Sleep? — how gut permeability and LPS translocation trigger neuroinflammation that fragments sleep
- Does Poor Sleep Cause Brain Inflammation? — the bidirectional loop between neuroinflammation, brain fog, and sleep loss
- How Does the Glymphatic System Work During Sleep? — how the brain’s waste clearance pathway depends on deep sleep
- What Is the Connection Between Chronic Inflammation and Insomnia? — the wired-but-tired cycle of cytokines, sleep loss, and 24-hour hyperarousal
- Why Do Men Sleep Worse After 50?
- What Is Autoimmune Insomnia and Why Does It Resist Standard Sleep Approaches?
- Can Inflammation Cause 3am Wakeups?
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- Can Chronic Stress Cause Insomnia Through Inflammation?
- Gut Bacteria and Insomnia: Which Microbes Affect Your Sleep (and Which Ones Help)
- Does an Anti-Inflammatory Diet Improve Sleep?
References
1. Arvidson, N. G., Gudbjörnsson, B., Elfman, L., Rydén, A. C., Tötterman, T. H., & Hällgren, R. (1994). Circadian rhythm of serum interleukin-6 in rheumatoid arthritis. Annals of the Rheumatic Diseases, 53(8), 521–524. https://pubmed.ncbi.nlm.nih.gov/7944637/
2. Hong, H. K., Maury, E., Ramsey, K. M., Perelis, M., Fez-Cerón, B., Vijayaraghavan, P., Kuber, A. N., Bhaskaran, N., Xu, X., & Bass, J. (2018). Requirement for NF-κB in maintenance of molecular and behavioral circadian rhythms in mice. Genes & Development, 32(21–22), 1367–1379. https://pubmed.ncbi.nlm.nih.gov/30366905/
3. Kikyo, N. (2023). Circadian regulation of macrophages and osteoclasts in rheumatoid arthritis. International Journal of Molecular Sciences, 24(15), 12307. https://pubmed.ncbi.nlm.nih.gov/37569682/
4. Lee, Y., & Park, K. I. (2024). The relationship between sleep and innate immunity. Encephalitis, 4(4), 69–75. https://pubmed.ncbi.nlm.nih.gov/38769055/
5. Pu, H., Bailey, L. C., Bauer, L. G., Voronkov, M., Baxter, M., Huber, K. V. M., Khorasanizadeh, S., Ray, D., & Rastinejad, F. (2025). Pharmacological targeting of BMAL1 modulates circadian and immune pathways. Nature Chemical Biology. https://pubmed.ncbi.nlm.nih.gov/40133642/
6. Srinivasan, M., & Walker, C. (2022). Circadian clock, glucocorticoids and NF-κB signaling in neuroinflammation — implicating glucocorticoid induced leucine zipper as a molecular link. ASN Neuro, 14, 17590914221120190. https://pubmed.ncbi.nlm.nih.gov/36317290/
7. Tang, L., Liu, L., Sun, X., Hu, P., Zhang, H., Wang, B., Zhang, X., Jiang, J., Zhao, X., & Shi, X. (2022). BMAL1/FOXA2-induced rhythmic fluctuations in IL-6 contribute to nocturnal asthma attacks. Frontiers in Immunology, 13, 947067. https://pubmed.ncbi.nlm.nih.gov/36505412/
8. Wang, C., Lutes, L. K., Barnoud, C., & Scheiermann, C. (2022). The circadian immune system. Science Immunology, 7(72), eabm2465. https://pubmed.ncbi.nlm.nih.gov/35658012/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 8 references cited
