“Waking up at 3am” is among the highest-volume sleep search terms in 2026. The prevailing explanations cover cortisol spikes, blood sugar crashes, and anxiety. The inflammatory mechanism — documented in published research describing an immune vulnerability window at those hours — is absent from all of them.
This article does three things. First, it explains what the immune apparatus is doing at 3am and why that hour constitutes the vulnerability window. Second, it maps four inflammatory pathways — each covered individually in the Inflammatory Sleep Disruption pillar — to the 3am phenomenon. Third, it provides a practical framework for distinguishing inflammatory 3am waking from metabolic, hormonal, and circadian causes.
This is a synthesis. Each pathway described below has a dedicated Tier 1 article with full mechanistic detail. This article connects them to one experience: waking up at 3am and not being able to fall back asleep.
What Happens in Your Immune Apparatus at 3am?
The circadian immune rhythm is governed by the interaction between NF-κB and the core clock protein BMAL1. Hong et al. (2018) demonstrated that NF-κB is not modulated by the clock — it is necessary for the clock to function. When NF-κB is chronically activated, it causes CLOCK/BMAL1 protein complexes to relocate across the genome to NF-κB-bound sites, redirecting the clock’s machinery toward inflammatory gene transcription. NF-κB activation also suppresses Period, Cryptochrome, and Rev-erb genes — the repressors that normally form the clock’s negative feedback loop. This creates a self-reinforcing cycle: chronic inflammation dismantles the circadian timing that would normally contain it.
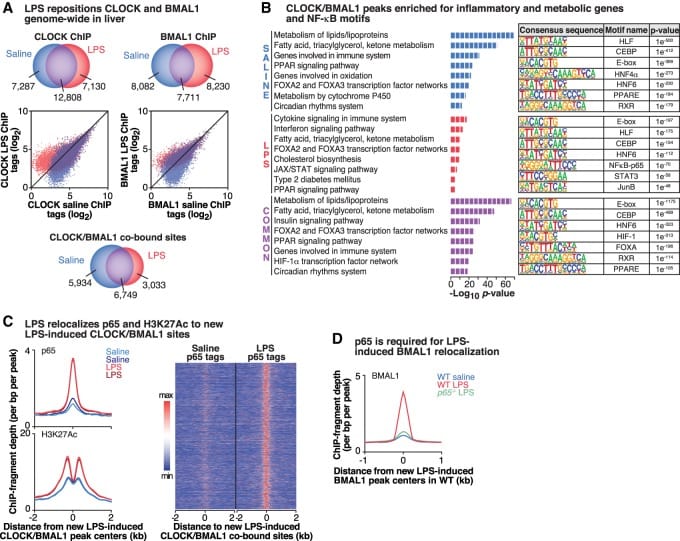
When BMAL1 is suppressed by NF-κB, downstream consequences follow. Tang et al. (2022) showed that BMAL1 suppresses IL-6 production in airway epithelial cells through a transcription factor called FOXA2. When BMAL1 is depleted, IL-6 increases and loses its normal circadian oscillation. In their cohort of asthmatic individuals, those with nocturnal flares had serum IL-6 of 7.45 ± 6.57 pg/mL compared to 2.81 ± 1.49 pg/mL in those without nighttime episodes — a 2.6-fold difference. The pathway is: NF-κB suppresses BMAL1, BMAL1 can no longer upregulate FOXA2, FOXA2 can no longer suppress IL-6, and IL-6 spikes at night.
Cortisol’s suppressive role at this time point involves a molecular intermediary. Cortisol induces GILZ (glucocorticoid-induced leucine zipper), a protein that prevents the NF-κB subunit p65 from entering the cell nucleus and activating inflammatory genes. Between 2am and 4am, when cortisol is at its lowest overnight concentration, GILZ expression falls and p65 translocates freely into the nucleus (Srinivasan & Walker, 2022). At the same time, the CLOCK protein acetylates glucocorticoid receptors, reducing their anti-inflammatory efficacy. Low cortisol plus impaired receptor sensitivity creates a double vulnerability at the pre-dawn trough.
The first rigorous human documentation of this circadian inflammatory pattern came from Arvidson et al. (1994), who measured serum IL-6 across 24-hour cycles in 48 individuals with rheumatic diseases and 10 healthy controls. IL-6 was undetectable in healthy controls but elevated in RA participants, peaking in the early morning hours. The temporal pattern correlated with morning stiffness severity. More recent evidence confirms the same mechanism acts through sleep disruption: Lee and Park (2024) reported that three nights of inadequate sleep caused IL-6 mRNA to increase three-fold and TNF-α mRNA to increase two-fold in healthy subjects.
Which Inflammatory Pathways Can Wake You Up at 3am?
Histamine and mast cells — nighttime histamine dump. Brain-resident mast cells release histamine on a circadian schedule. Histaminergic neurons in the tuberomammillary nucleus (TMN) fire during wakefulness and are silenced during sleep — they are the brain’s primary wake-lock (Thakkar, 2011). When mast cell activation is elevated, the natural nocturnal histamine peak overshoots the threshold for sleep maintenance, engaging the TMN wake circuit at 2-4am. This is why antihistamines induce drowsiness — they suppress the same circuit that inflammatory histamine activates. The full mechanism is covered in Histamine and 3am Waking and Histamine Intolerance and Sleep.
Gut lipopolysaccharides — barrier breakdown peaks at night. When intestinal barrier integrity is compromised, bacterial lipopolysaccharides (LPS) enter the bloodstream and activate TLR4/NF-κB transcription. The vagus nerve relays gut-derived inflammatory input to the brainstem — vagotomy abolished the sleep-deprivation-amplified cytokine response in animal studies (Zhang et al., 2021). Because LPS-triggered inflammation adds to the existing nocturnal NF-κB peak, the combined load can breach the arousal threshold at 3am. For the full gut-sleep mechanism, see Leaky Gut and Insomnia.
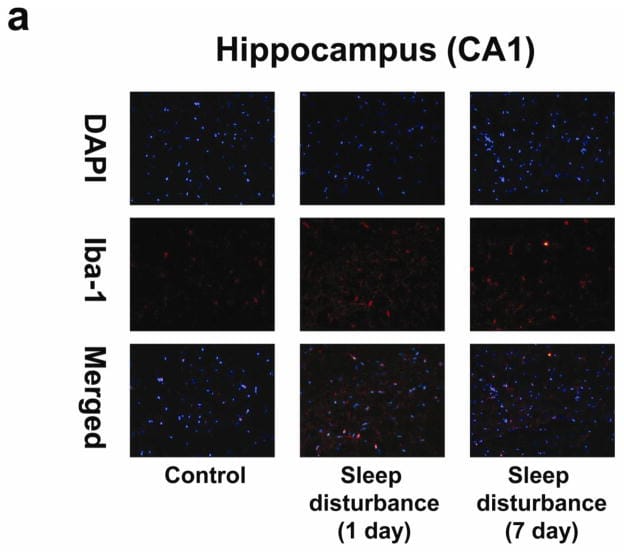
Neuroinflammation — microglial activation disrupting sleep maintenance. Sleep deprivation activates microglia — the brain’s resident immune cells — in the hippocampus (CA1 and CA3 regions), where they release IL-6 into sleep-regulatory circuits (Zhu et al., 2012). Mitochondrial DNA leakage from stressed microglia triggers NF-κB activation in brain tissue, creating a local neuroinflammatory cascade that disrupts sleep from within the brain itself (Hu et al., 2024). This pathway may explain why inflammatory 3am waking feels like the brain switching on — not the body. The full neuroinflammation-sleep connection is in Brain Fog and Poor Sleep: Neuroinflammation.
Pro-inflammatory cytokines — IL-6 and TNF-α arousal redistribution. In chronic insomnia, the normal circadian pattern of IL-6 secretion inverts — from a 4am peak (which supports sleep consolidation) to a 7pm peak (which drives daytime fatigue and hyperarousal). TNF-α loses its circadian rhythm altogether (Vgontzas et al., 2002). This circadian misalignment means the brain receives wakefulness input at 3am without the consolidation factor that would normally keep sleep intact. The result is the “wired but tired” phenotype — exhaustion that cannot translate into sustained sleep. The full bidirectional mechanism is in Chronic Inflammation and Insomnia.
How Do You Know If Inflammation Is the Reason You Wake Up at 3am?
Inflammatory 3am waking feels like the brain switched on. You wake alert, mind activated, sometimes with joint stiffness or sinus pressure. Falling back asleep takes 30-90+ minutes. Daytime fatigue often worsens as the day progresses rather than improving. This pattern occurs more frequently in people with chronic inflammatory conditions, histamine sensitivity, gut permeability issues, or persistent brain fog.
Metabolic 3am waking (blood sugar crash) feels like the body is requesting fuel. Hunger, shakiness, heart racing. Eating something resolves it within 15-20 minutes.
Hormonal 3am waking (cortisol spike or night sweats) feels like the body’s thermostat malfunctioned. Hot, sweaty, heart pounding. Common during perimenopause and andropause.
Circadian 3am waking (advanced sleep phase) feels like morning arrived early. Awake, calm, not distressed but unable to fall back asleep. Common after 50 and in early-rise chronotypes.
The meta-analytic evidence supports the inflammatory angle as measurable. Irwin et al. (2016) reviewed 72 studies (n > 50,000 participants) and found that chronic sleep disturbance is associated with elevated CRP and IL-6. High-sensitivity CRP (hs-CRP) is the accessible blood test that tracks with the inflammatory sleep phenotype. Dressle et al. (2022) added that people with chronic insomnia show moderately elevated cortisol (standardized mean difference = 0.50) consistent with a 24-hour hyperarousal state — a finding that overlaps with but is distinct from the inflammatory pattern.
The inflammatory pattern should be considered when 3am waking co-occurs with any chronic inflammatory condition, histamine sensitivity, gut issues, or persistent brain fog — and when the standard explanations (blood sugar, anxiety, cortisol) have been addressed without resolution.
This is not a way to identify a cause. It is a framework for asking better questions.
Inflammation is one of several causes that can drive 3am waking. Metabolic blood sugar patterns, hormonal changes, autonomic nervous imbalances, and circadian timing disruption can all produce middle-of-the-night arousal. The inflammatory pathway is the one that rarely gets checked.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can Inflammation Cause Insomnia?
The relationship is bidirectional. Inflammation causes insomnia by activating arousal pathways and fragmenting sleep architecture. Insomnia causes inflammation by elevating NF-κB, which drives cytokine transcription that compounds the original sleep disruption. Once the loop is established, each direction reinforces the other. The full mechanism — including the NF-κB/cytokine cascade and the evidence for reversibility — is covered in Chronic Inflammation and Insomnia.
Does Inflammation Make You Tired But Unable to Sleep?
The prostaglandin balance may also contribute. PGD2 promotes sleep while PGE2 promotes wakefulness, and inflammatory states can redistribute this balance. The exhaustion-without-sleep phenotype is covered in more depth in Why Does Inflammation Make You Exhausted But Unable to Sleep?.
Does Reducing Inflammation Improve Sleep?
On the behavioral side, CBT-I (a non-pharmacological insomnia approach) reduced CRP and reversed NF-κB activation at 4-month and 16-month follow-ups in a randomized controlled trial of 123 older adults (Irwin et al., 2015). The evidence supports the principle that reducing inflammatory load improves sleep, but the approach depends on which pathway is dominant. Direction, not prescription — the pathway matters.
What Inflammatory Markers Are Associated with Poor Sleep?
IL-6 was the first cytokine documented in a circadian pattern in RA participants (Arvidson et al., 1994). Its effect size of 0.42 after multi-night partial sleep restriction reflects a moderate but consistent elevation (Ballesio et al., 2026).
TNF-α loses its circadian rhythm altogether in chronic insomnia; mRNA increases two-fold after three nights of inadequate sleep (Lee & Park, 2024).
CRP shows the largest effect size (0.76) after multi-night partial sleep restriction (Ballesio et al., 2026). High-sensitivity CRP (hs-CRP) is the accessible blood test for tracking this marker.
Additionally, 10 nights of 4-hour sleep restriction produced IL-6 elevations that correlated with increased pain sensitivity (r = 0.67, p < 0.01) (Haack et al., 2007) — demonstrating that these markers track with physical consequences, not just laboratory measurements.
Related Reading
- Inflammatory Sleep Disruption: How Chronic Inflammation Fragments Your Sleep
- Histamine and 3am Waking
- Histamine Intolerance and Sleep
- Why Is Inflammation Worse at Night?
- Leaky Gut and Insomnia
- Brain Fog and Poor Sleep: Neuroinflammation
- Glymphatic System and Sleep
- Chronic Inflammation and Insomnia
- Why Men Sleep Worse After 50
- Autoimmune Insomnia
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- The Stress-Inflammation-Insomnia Cycle
- Gut Bacteria and Insomnia
- Anti-Inflammatory Diet and Sleep
References
Arvidson, N.G., Gudbjornsson, B., Elfman, L., Ryden, A.C., Totterman, T.H., & Hallgren, R. (1994). Circadian rhythm of serum interleukin-6 in rheumatoid arthritis. Annals of the Rheumatic Diseases, 53(8), 521–524. https://pubmed.ncbi.nlm.nih.gov/7944637/
Ballesio, A., Fiori, V., & Lombardo, C. (2026). Effects of experimental sleep deprivation on peripheral inflammation: An updated meta-analysis of human studies. Journal of Sleep Research, e70099. https://pubmed.ncbi.nlm.nih.gov/40474574/
Dressle, R.J., Feige, B., Spiegelhalder, K., Schmucker, C., Benz, F., Mey, N.C., & Riemann, D. (2022). HPA axis activity in patients with chronic insomnia: A systematic review and meta-analysis of case-control studies. Sleep Medicine Reviews, 62, 101588. https://pubmed.ncbi.nlm.nih.gov/35091194/
Fragiadaki, K., Tektonidou, M.G., Konsta, M., Chrousos, G.P., & Sfikakis, P.P. (2012). Sleep disturbances and interleukin 6 receptor inhibition in rheumatoid arthritis. Journal of Rheumatology, 39(1), 60–62. https://pubmed.ncbi.nlm.nih.gov/22133618/
Haack, M., Sanchez, E., & Mullington, J.M. (2007). Elevated inflammatory markers in response to prolonged sleep restriction are associated with increased pain experience in healthy volunteers. Sleep, 30(9), 1145–1152. https://pubmed.ncbi.nlm.nih.gov/17910386/
Hong, H.K., Maury, E., Ramsey, K.M., Bhaskaran, N., Xu, X., et al. (2018). Requirement for NF-κB in maintenance of molecular and behavioral circadian rhythms in mice. Genes & Development, 32(21-22), 1367–1379. https://pubmed.ncbi.nlm.nih.gov/30366905/
Hu, Y., Wang, Y., Wang, Y., Zhang, Y., Wang, Z., Xu, X., … & Long, J. (2024). Sleep deprivation triggers mitochondrial DNA release in microglia to induce neural inflammation. Antioxidants, 13(7), 833. https://pubmed.ncbi.nlm.nih.gov/39061901/
Irwin, M.R., Wang, M., Ribeiro, D., Cho, H.J., Olmstead, R., Breen, E.C., … & Cole, S. (2008). Sleep loss activates cellular inflammatory signaling. Biological Psychiatry, 64(6), 538–540. https://pubmed.ncbi.nlm.nih.gov/18561896/
Irwin, M.R., Olmstead, R., & Carroll, J.E. (2016). Sleep disturbance, sleep duration, and inflammation: A systematic review and meta-analysis of cohort studies and experimental sleep deprivation. Biological Psychiatry, 80(1), 40–52. https://pubmed.ncbi.nlm.nih.gov/26140821/
Irwin, M.R., Olmstead, R., Breen, E.C., Witarama, T., Carrillo, C., Sadeghi, N., … & Cole, S. (2015). Cognitive behavioral therapy and tai chi reverse cellular and genomic markers of inflammation in late-life insomnia: A randomized controlled trial. Biological Psychiatry, 78(10), 721–729. https://pubmed.ncbi.nlm.nih.gov/25748580/
Lee, Y., & Park, K.I. (2024). The relationship between sleep and innate immunity. Encephalitis, 4(4), 69–75. https://pubmed.ncbi.nlm.nih.gov/38769055/
Srinivasan, M., & Walker, C. (2022). Circadian clock, glucocorticoids and NF-κB signaling in neuroinflammation — implicating glucocorticoid induced leucine zipper as a molecular link. ASN Neuro, 14, 17590914221120190. https://pubmed.ncbi.nlm.nih.gov/36317290/
Tang, L., Liu, L., Sun, X., Hu, P., Zhang, H., Wang, B., … & Shi, X. (2022). BMAL1/FOXA2-induced rhythmic fluctuations in IL-6 contribute to nocturnal asthma attacks. Frontiers in Immunology, 13, 947067. https://pubmed.ncbi.nlm.nih.gov/36505412/
Thakkar, M.M. (2011). Histamine in the regulation of wakefulness. Sleep Medicine Reviews, 15(1), 65–74. https://pubmed.ncbi.nlm.nih.gov/20851648/
Vgontzas, A.N., Zoumakis, M., Papanicolaou, D.A., Bixler, E.O., Prolo, P., Lin, H.M., … & Chrousos, G.P. (2002). Chronic insomnia is associated with a shift of interleukin-6 and tumor necrosis factor secretion from nighttime to daytime. Metabolism, 51(7), 887–892. https://pubmed.ncbi.nlm.nih.gov/12077736/
Zhang, Y., Xie, B., Chen, X., Zhang, J., & Yuan, S. (2021). A key role of gut microbiota-vagus nerve/spleen axis in sleep deprivation-mediated aggravation of systemic inflammation after LPS administration. Life Sciences, 265, 118736. https://pubmed.ncbi.nlm.nih.gov/33176177/
Zhu, B., Dong, Y., Xu, Z., Gompf, H.S., Ward, S.A.P., Xue, Z., … & Xie, Z. (2012). Sleep disturbance induces neuroinflammation and impairment of learning and memory. Neurobiology of Disease, 48(3), 348–355. https://pubmed.ncbi.nlm.nih.gov/22776332/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 17 references cited
