If you wake up between 2am and 4am — fully alert, heart pounding, unable to fall back asleep — there is a neurotransmitter involved. Histamine is not just the molecule behind allergies and itchy eyes. It is the brain’s primary arousal driver, produced on a circadian rhythm that peaks in the late sleep period. And the source of that nocturnal histamine is not limited to neurons — immune cells called mast cells release histamine on their own circadian schedule, with implications for anyone dealing with inflammation or immune activation. This article covers the histamine-wakefulness pathway, the role of mast cells in nocturnal histamine release, and why the pre-dawn cortisol rise compounds the problem. For the broader picture of how inflammation disrupts sleep through multiple pathways, see Inflammatory Sleep Disruption.
How Does Histamine Control Wakefulness in the Brain?
The tuberomammillary nucleus, a small cluster of neurons in the posterior hypothalamus, is the brain’s sole source of neuronal histamine (Scammell et al., 2019). These neurons project diffusely to the cortex, thalamus, and every major wake-promoting region. During wakefulness, they fire continuously. During both NREM and REM sleep, they stop firing.
This on-off behavior is maintained by a mutual inhibition circuit. The ventrolateral preoptic area (VLPO), the brain’s primary sleep-promoting region, releases GABA onto tuberomammillary neurons to suppress histamine output and initiate sleep. In the opposite direction, histamine from the tuberomammillary nucleus inhibits the VLPO to maintain wakefulness. This mutual inhibition creates a stable toggle — the brain is either in a wake-dominant state or a sleep-dominant state, with rapid transitions between them (Scammell et al., 2019).
The H1 receptor is the downstream effector that converts histamine into conscious wakefulness. In mice lacking the H1 receptor, the arousal-promoting effect of histamine is abolished — even when histamine release is pharmacologically increased, the animals show no wakefulness response (Huang et al., 2006). This is also why first-generation antihistamines (diphenhydramine, doxylamine) cause drowsiness: they cross the blood-brain barrier and block H1 receptors on cortical neurons, reducing the wake-promoting input from the tuberomammillary nucleus.
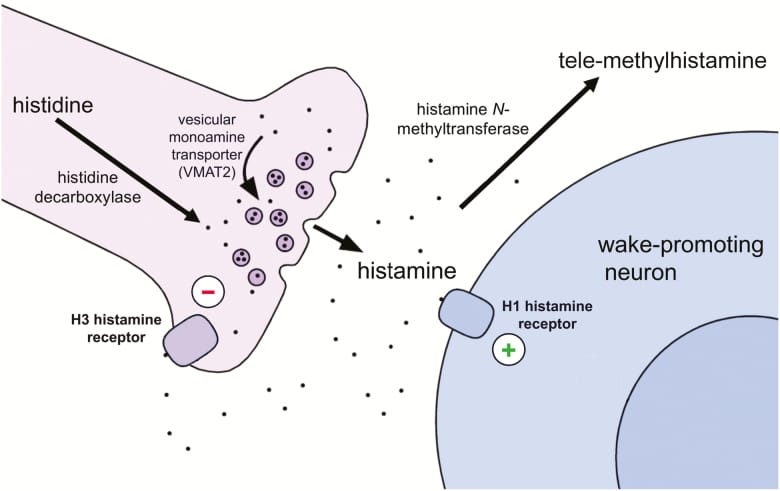
What makes histamine different from other wake-promoting neurotransmitters is its function during states of heightened arousal. Histamine neurons show their highest activity during attentive wakefulness — exploration, danger assessment, and situations requiring heightened vigilance — rather than during calm rest (Mochizuki, 2022). The brain’s histamine response is an ancient alerting mechanism. When mast cells release histamine during sleep, the brain responds as though it has detected something requiring urgent attention.
What Is a Histamine Dump at Night?
The tuberomammillary nucleus is not the only source of histamine in the brain. Mast cells — immune cells embedded in brain tissue — contribute a large share of total brain histamine. When mast cells are absent in animal models, both wakefulness and histamine-mediated arousal responses are reduced, and H1-receptor-blocking antihistamines lose their sleep-promoting effect (Chikahisa et al., 2013). This means mast cell-derived histamine is not a minor contributor to brain arousal — it is a functionally necessary part of the wakefulness response.
What makes the nocturnal histamine peak predictable is that mast cells have their own internal molecular clock. Nakamura et al. (2017) demonstrated that mast cells contain clock genes (CLOCK, BMAL1, Period2) that drive circadian oscillation of histamine release through transcriptional control of OCT3, a histamine transporter. In mast cell-deficient mice, plasma histamine lost its circadian rhythm. When these mice were reconstituted with mast cells carrying a mutant Clock gene, the circadian histamine pattern did not return. The mast cell clock is not optional for the histamine rhythm — it is the source of it.
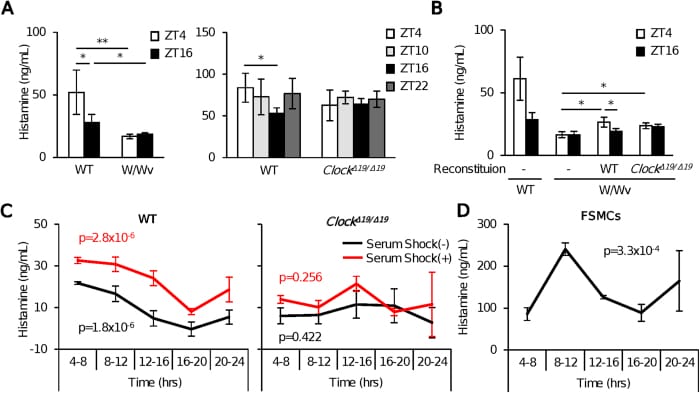
In healthy people, plasma histamine peaks in the early morning hours (Nakamura et al., 2017). This peak coincides with the timing of allergic flares in asthmatics and the timing of histamine-mediated waking. During normal conditions, the peak stays below the arousal threshold and sleep continues. But when mast cell activation is elevated — from chronic inflammation, immune imbalance, or prolonged stress — the amplitude of the peak increases. More histamine is released at 2-4am than the sleep-maintenance circuit can override, and the result is sudden, full wakefulness.
Stress desynchronizes this clock. In the same study, mice subjected to chronic restraint stress showed complete loss of circadian histamine oscillation — the normal rhythm flattened, and histamine release became erratic rather than timed (Nakamura et al., 2017). This is relevant because many people who experience 3am waking are also under chronic stress, which may both amplify mast cell activation and disrupt the timing of histamine release.
Does Cortisol Affect Histamine Levels at Night?
The cortisol awakening response — a rapid increase in cortisol output that begins around 3-4am and peaks approximately 30 minutes after waking — is a normal part of the diurnal hormonal rhythm. Its function is to prepare the body for daytime activity: mobilizing glucose, priming the immune response, and increasing alertness.
The problem arises when this cortisol rise intersects with an already-elevated histamine peak. Cortisol is associated with mast cell degranulation — the process by which mast cells release their stored histamine and other inflammatory mediators. In someone with normal mast cell activity, the pre-dawn cortisol rise produces a modest, tolerable increase in histamine. In someone with elevated mast cell activation, cortisol triggers a larger release on top of the circadian histamine peak that is already running high.
This creates a compounding effect at a vulnerable time. Between 2am and 4am, the mast cell clock is driving histamine toward its circadian maximum. Cortisol begins rising in the same window, stimulating additional mast cell degranulation. The two wakefulness-promoting inputs converge, and the combined histamine load exceeds the capacity of the VLPO-TMN sleep-maintenance circuit to hold the brain in sleep (Meckes & Meckes, 2026).
The inflammatory dimension compounds this. During the early morning hours, cortisol is at its lowest point before the pre-dawn rise begins. During this low-cortisol window, the inflammatory transcription factor NF-kB is less suppressed, allowing inflammatory activity to increase. This means the 2-4am period is characterized by both rising inflammation (from reduced cortisol suppression) and rising histamine (from the mast cell clock), followed by a cortisol surge that can trigger additional mast cell degranulation. For someone with chronic inflammation or mast cell activation, this three-hour window concentrates multiple arousal-promoting inputs into the same narrow timeframe.
Meckes & Meckes (2026) documented a case in which a 74-year-old individual with post-viral inflammatory insomnia — with Oura Ring sleep scores in the 30-40 range — showed recovery to scores above 75 within 24 hours of initiating histamine-targeted steps (H1 and H2 receptor blockade plus environmental and dietary histamine reduction). The speed of this response is consistent with a model where elevated histamine was the primary driver of the arousal.
How Common Is Histamine-Driven Insomnia?
Weinstock et al. (2025) studied 553 people with mast cell activation syndrome (MCAS) alongside 558 controls. Among women with MCAS, 58.7% reported severe chronic insomnia, compared to 24.0% of controls — an odds ratio of 4.8. Among men with MCAS, 42.1% reported severe chronic insomnia versus 9.0% of controls — an odds ratio of 7.7. Sleep attacks (sudden, irresistible sleep episodes) occurred in over 40% of women in the MCAS group.
These numbers represent the formally identified end of a broader spectrum. MCAS identification requires meeting defined criteria, and many people with elevated mast cell activity or histamine intolerance do not meet that threshold. The gap between full MCAS and normal mast cell function contains a range of intermediate states where histamine is elevated enough to disrupt sleep but not elevated enough to produce the multi-organ involvement that prompts a full workup.
Several patterns may indicate that histamine is contributing to sleep disruption. Waking between 2am and 4am with sudden full alertness (rather than the gradual, groggy waking of other sleep disturbances) is consistent with a histamine-mediated arousal. Accompanying congestion, nasal swelling, skin flushing, itching, or a racing heart during the waking episode are associated with mast cell degranulation. A pattern that worsens with high-histamine foods consumed in the evening (aged cheese, fermented foods, wine, cured meats) or improves with dietary histamine reduction is another indicator to consider.
The Weinstock et al. (2025) data also showed that antihistamine therapy rated 6.3 out of 10 on a self-reported benefit scale for neuropsychiatric effects across the MCAS population — partial but not complete relief, consistent with histamine being one of several mediators released by activated mast cells.
Many people have more than one cause contributing to their sleep disruption. Histamine-driven waking may compound with autonomic, metabolic, hormonal, or circadian factors. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Histamine Cause Insomnia?
Histamine promotes wakefulness through the H1 receptor on cortical neurons. When histamine is elevated during sleep hours — whether from tuberomammillary neuron activity that does not fully suppress, or from mast cell degranulation — the brain receives a sustained wake-promoting input that opposes sleep maintenance. The circadian peak in histamine release between 2am and 4am means that even a moderate elevation in baseline mast cell activity can become sufficient to break through sleep during this window. In the Weinstock et al. (2025) study, the strong association between mast cell activation and chronic insomnia (odds ratios of 4.8-7.7) provides population-level evidence for histamine as a contributor to sleep disruption.
Does Histamine Prevent Deep Sleep?
The tuberomammillary nucleus must stop firing for both deep NREM and REM sleep to occur. Histamine-deficient mice (HDC knockouts) show altered sleep architecture with changes in both NREM and REM distribution (Scammell et al., 2019). In the other direction, elevated histamine holds the tuberomammillary neurons in a firing state, maintaining cortical arousal and preventing the brain from transitioning into the deeper stages where slow-wave activity and memory consolidation occur. The result is lighter, more fragmented sleep even when total sleep time appears adequate.
How Do You Stop Histamine From Waking You at Night?
Reducing dietary histamine load in the evening (limiting aged cheeses, fermented foods, cured meats, and alcohol) lowers the substrate available for the nocturnal peak. Supporting diamine oxidase (DAO), the enzyme that breaks down ingested histamine in the gut, is another avenue — DAO activity can be impaired by alcohol, certain medications, and gut inflammation. Reducing overall inflammatory load through gut health, stress management, and identifying immune triggers addresses the upstream activation that drives mast cell degranulation. The combination of contributing factors varies between individuals, which is why identifying the specific upstream driver — gut permeability, chronic immune activation, stress — matters more than applying the same dietary or enzyme-support steps to everyone.
Are Mast Cells Connected to Circadian Rhythm?
Nakamura et al. (2017) demonstrated that mast cells are not passive immune cells that release histamine only in response to allergens — they follow an internally generated circadian rhythm of activation. The transcription factor CLOCK binds to the promoter region of OCT3 (a histamine transporter) in mast cells, creating a time-dependent pattern of histamine release that peaks in the early morning hours. When this clock is intact, the histamine peak is predictable and, in healthy individuals, stays below the arousal threshold. When chronic stress or circadian disruption (such as irregular sleep schedules or overnight work rotations) desynchronizes the mast cell clock, histamine release can become erratic, amplified, or mistimed — all of which increase the likelihood of nocturnal waking.
Related Reading
- Inflammatory Sleep Disruption — the full cause overview covering all five inflammatory mechanisms
- Can Histamine Intolerance Cause Sleep Problems? — how DAO deficiency and mast cell activation compound histamine-driven insomnia
- Why Is Inflammation Worse at Night? — how NF-kB and the circadian clock create a pre-dawn inflammatory vulnerability window
- How Does Leaky Gut Affect Sleep? — how gut permeability and LPS translocation trigger neuroinflammation that fragments sleep
- Does Poor Sleep Cause Brain Inflammation? — the bidirectional loop between neuroinflammation, brain fog, and sleep loss
- How Does the Glymphatic System Work During Sleep? — how the brain’s waste clearance pathway depends on deep sleep
- What Is the Connection Between Chronic Inflammation and Insomnia? — the wired-but-tired cycle of cytokines, sleep loss, and 24-hour hyperarousal
- Why Do Men Sleep Worse After 50?
- What Is Autoimmune Insomnia and Why Does It Resist Standard Sleep Approaches?
- Can Inflammation Cause 3am Wakeups?
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- Can Chronic Stress Cause Insomnia Through Inflammation?
- Gut Bacteria and Insomnia: Which Microbes Affect Your Sleep (and Which Ones Help)
- Does an Anti-Inflammatory Diet Improve Sleep?
References
1. Chikahisa, S., et al. (2013). Histamine from brain resident MAST cells promotes wakefulness and modulates behavioral states. PLoS ONE, 8(10), e78434. https://pubmed.ncbi.nlm.nih.gov/24205232/
2. Huang, Z. L., et al. (2006). Altered sleep-wake characteristics and lack of arousal response to H3 receptor antagonist in histamine H1 receptor knockout mice. Proceedings of the National Academy of Sciences USA, 103(12), 4687-4692. https://pubmed.ncbi.nlm.nih.gov/16537376/
3. Meckes, A. J., & Meckes, J. W. (2026). Case report: A case of post-viral inflammatory insomnia: observed sleep restoration associated with histamine-targeted interventions and implications for mast cell pathways. Frontiers in Sleep, 4, 1736866. https://www.frontiersin.org/articles/10.3389/frsle.2025.1736866/full
4. Mochizuki, T. (2022). Histamine as an alert signal in the brain. Current Topics in Behavioral Neuroscience, 59, 413-425. https://pubmed.ncbi.nlm.nih.gov/34448132/
5. Nakamura, Y., et al. (2017). Regulation of plasma histamine levels by the mast cell clock and its modulation by stress. Scientific Reports, 7, 39934. https://pubmed.ncbi.nlm.nih.gov/28074918/
6. Scammell, T. E., et al. (2019). Histamine: neural circuits and new medications. Sleep, 42(1), zsy183. https://pubmed.ncbi.nlm.nih.gov/30239935/
7. Weinstock, L. B., et al. (2025). Prevalence and treatment response of neuropsychiatric disorders in mast cell activation syndrome. Brain, Behavior, and Immunity – Health, 48, 101048. https://pubmed.ncbi.nlm.nih.gov/40686928/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
