Men over 50 notice their sleep deteriorating — more nighttime waking, less deep sleep, earlier morning alertness — and assume it is a normal part of aging. The inflammatory mechanism behind this change is documented in human studies but rarely covered in consumer health content.
This article covers how age-related sterile inflammation — inflammaging — fragments sleep architecture in men. It does not cover the full overview of inflammatory sleep pathways — that is in the parent article, Inflammatory Sleep Disruption.
Inflammaging is one of several mechanisms that disrupt sleep. Hormonal changes, metabolic changes, autonomic dysregulation, and circadian changes may compound the inflammatory pathway. The pillar article covers the broader picture.
Does Aging Cause Chronic Inflammation?
In 2000, immunologist Claudio Franceschi introduced the term inflammaging to describe a specific phenomenon: chronic, low-grade inflammation that rises with age in the absence of infection (Franceschi et al., 2000). Unlike inflammation from an injury or illness, inflammaging originates from the body’s own senescent cells.
The source is senescent cells — cells that have stopped dividing but remain metabolically active. Instead of clearing out, they accumulate in tissues and release a cocktail of inflammatory molecules known as the senescence-associated secretory phenotype (SASP). This cocktail includes IL-6, TNF-a, and other inflammatory mediators that enter circulation. They are aged cells performing a different metabolic role — one that produces a steady rise in baseline inflammation.
A 2016 experimental study demonstrated how sleep disruption accelerates senescent cell accumulation. Carroll et al. exposed 29 community-dwelling adults aged 61-86 to a single night of partial sleep deprivation (restricted to a 3 AM-7 AM sleep window) and measured gene expression changes in their peripheral blood cells. SASP gene expression increased from baseline to the sleep deprivation night (p < 0.05). DNA damage response genes were also upregulated: NFKB2 (p = 0.008), NBS1 (p = 0.004), and CHK2 (p < 0.001). The senescence marker p16(INK4a) was elevated one day after the deprivation (p < 0.01 by microarray) (Carroll et al., 2016).
These changes occurred in peripheral blood mononuclear cells — meaning the activation was not local to one tissue but present throughout the bloodstream. One night of restricted sleep was enough to upregulate the molecular machinery that drives age-related inflammation.
In this experimental model, sleep disruption fed inflammaging — accelerating the same senescent cell accumulation that contributes to sleep disruption in the first place.
How Does Age-Related Inflammation Fragment Sleep?
Vgontzas et al. (2003) provided the first controlled laboratory evidence, comparing 15 younger and 13 older healthy adults with 24-hour blood sampling and polysomnography. Older adults had higher mean 24-hour IL-6 plasma concentrations than younger adults (p < 0.05). Their cortisol levels were also elevated (p < 0.05). On polysomnography, the older group showed increased wake time, more stage 1 (light) sleep, less slow-wave sleep, and reduced total sleep time (all p < 0.05).
Within individuals, the IL-6-sleep relationship held as well. IL-6 plasma concentrations were positively associated with total wake time in both age groups, with the association being stronger in the older cohort (p < 0.05). Vgontzas et al. concluded that age-related overproduction of IL-6 and cortisol is a primary driver of sleep fragmentation in older adults — not a consequence of it (Vgontzas et al., 2003).
Hong et al. (2005) extended this with polysomnographic data from 70 healthy adults. Morning IL-6 levels correlated with specific sleep architecture deficits: increased wake time after sleep onset (WASO; rho = 0.29, p < 0.05), decreased sleep efficiency (rho = -0.36, p < 0.01), decreased slow-wave sleep percentage (rho = -0.26, p < 0.05), and increased REM latency (rho = 0.31, p = 0.01). Stage 1 sleep percentage showed a positive but non-significant trend (rho = 0.23, p = 0.053). After hierarchical regression controlling for race, gender, age, and BMI, IL-6 remained independently associated with REM latency, sleep efficiency, and WASO (Hong et al., 2005).
Slow-wave sleep loss and increased WASO — the two deficits that track with IL-6 elevation across these studies — are also two of the more pronounced sleep architecture changes in aging men.
Longitudinal data from Stahl et al. (2021) showed that this relationship persists over time. In 195 older adults (mean age 74.4) followed for 15 months with blood draws every three months, two IL-6 trajectory groups emerged: a stable lower group (84% of participants, mean IL-6 of 3.2 pg/mL) and a consistently elevated group (16%, mean IL-6 of 9.5 pg/mL — nearly three-fold higher). Poor subjective sleep quality at baseline predicted membership in the elevated group (OR = 1.9; 95% CI 1.03-3.55). The 9.5 pg/mL concentration in the elevated group exceeds thresholds that other aging cohorts have associated with frailty, sarcopenia, and cognitive decline (Stahl et al., 2021).
Poor sleep maintenance predicts sustained, high IL-6 concentrations over months — a chronic inflammatory state consistent with the inflammaging model, persisting across the full 15-month follow-up period.
Why Do Men Experience Inflammatory Sleep Disruption Differently?
Xiao et al. (2022) analyzed data from the MrOS (Osteoporotic Fractures in Men) cohort — over 3,000 older men with multiday actigraphy and fasting blood samples. Lower rest-activity rhythm amplitude, a direct marker of fragmented sleep-wake cycling, was associated with higher CRP, IL-6, TNF-a, and TNF-a soluble receptor II. Men in the lowest quartile of rhythm consolidation had approximately two-fold increased odds of being in the highest quartile of multi-marker inflammation — three or more inflammatory markers simultaneously elevated. These associations held after adjusting for age, BMI, comorbidities, physical activity, and medication use (Xiao et al., 2022).
This is the only study in this article’s reference pool conducted exclusively in older men, measuring multiple inflammatory markers simultaneously against objective sleep-wake data.
Piber et al. (2023) identified a sex difference in the inflammatory pathway that sleep fragmentation activates. In 262 community-dwelling older adults (49.2% male, mean age 71.9), greater wake time after sleep onset was associated with higher NF-kB — nuclear factor kappa-light-chain-enhancer of activated B cells, a transcription factor that drives the production of IL-6, TNF-a, and CRP — after full covariate adjustment (beta = 0.42, p < 0.01). This association held across five successive adjustment models controlling for sociodemographic, behavioral, and socio-emotional factors (Piber et al., 2023).
Total sleep time showed no association with NF-kB. It is sleep fragmentation — wakefulness after falling asleep — that activates this inflammatory pathway. Duration alone does not.
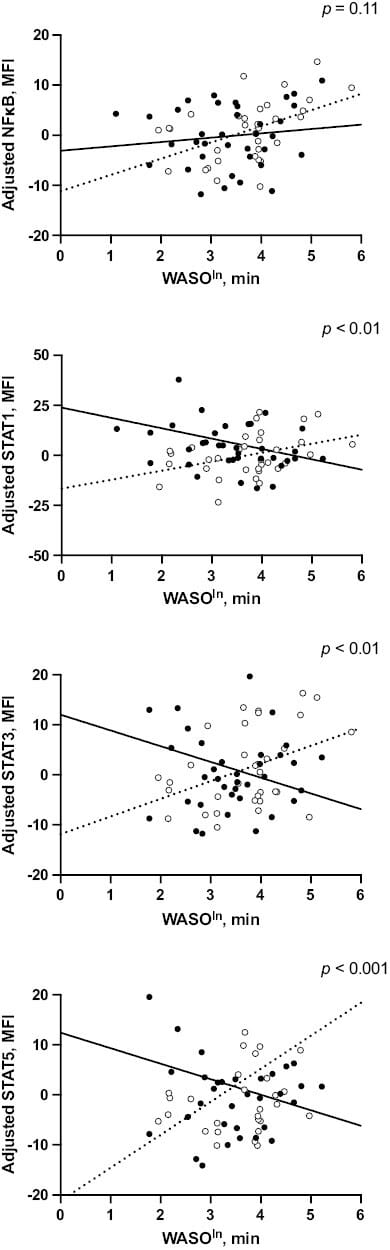
The sex difference appeared in the downstream signaling. In women, WASO was additionally associated with elevated STAT1 (beta = 0.47, p < 0.05), STAT3 (beta = 0.52, p < 0.05), and STAT5 (beta = 0.64, p < 0.01) — cytokine amplifier proteins downstream of IL-6. In men, STAT1/3/5 associations were not statistically meaningful. In this cohort, men's inflammatory response to sleep fragmentation ran primarily through NF-kB; women's engaged both NF-kB and the STAT cascade (Piber et al., 2023).
There is also a recognition gap. When women over 50 report sleep disruption, it is often attributed to estrogen decline and hot flashes — menopause provides a well-understood framework. Men presenting with the same sleep complaints — fragmented sleep, increased waking, less deep sleep — have no equivalent narrative. Their inflammatory pathway goes unidentified because the research has historically defaulted to mixed-sex samples or female-specific framing.
Does Poor Sleep Make Age-Related Inflammation Worse?
Irwin, Olmstead, and Carroll (2016) conducted the definitive meta-analysis: 72 studies encompassing over 50,000 participants. Sleep disturbance was associated with elevated IL-6 (ES = 0.20; 95% CI 0.08-0.31) and CRP (ES = 0.12; 95% CI 0.05-0.19). The IL-6 effect size was larger than CRP, identifying IL-6 as the more responsive marker in the context of disrupted sleep (Irwin et al., 2016).
A distinction in the data matters for men over 50: short sleep duration (under 7 hours) was associated with higher CRP but not with IL-6. Sleep disturbance — fragmentation, poor maintenance, repeated waking — was the driver of IL-6 elevation. This means the quality and continuity of sleep, not just total hours, determines the inflammatory outcome.
The Carroll et al. (2016) experimental data provides the reverse direction. A single night of partial sleep deprivation in adults aged 61-86 activated SASP gene expression and DNA damage response genes — NFKB2 (p = 0.008), p16(INK4a) elevated one day post-deprivation (p < 0.01). This links acute sleep disruption to the accumulation of senescent cell markers (Carroll et al., 2016).
Here is how the self-reinforcing loop between inflammation and sleep fragmentation runs: elevated IL-6, TNF-a, and CRP from senescent cells fragment sleep architecture — reducing slow-wave sleep and increasing wakefulness. Fragmented sleep then activates NF-kB (Piber et al., 2023) and SASP gene expression (Carroll et al., 2016), producing more inflammatory cytokines. Each iteration compounds the previous one.
The consequences of inflammaging-driven sleep fragmentation extend beyond daytime fatigue. The same inflammatory loop intersects with brain waste clearance. Glymphatic clearance — the brain’s overnight process for removing amyloid-beta and tau proteins — requires consolidated deep sleep, the sleep stage that IL-6 elevation reduces first. For the full mechanism, see How Does the Glymphatic System Work During Sleep?.
Can Inflammaging Be Reversed?
Multiple lines of evidence support the modifiability of inflammaging. Regular aerobic exercise lowers circulating IL-6 in older adults across numerous trials. Caloric restriction reduces baseline inflammatory markers in aging populations. Both approaches address the upstream accumulation of senescent cells and the downstream inflammatory load they produce.
Sleep itself may be an underappreciated entry point. The Irwin et al. (2016) meta-analysis established that sleep disturbance — fragmentation and poor maintenance — drives IL-6 elevation. The implication runs in both directions: if fragmented sleep worsens inflammaging, improving sleep continuity should reduce IL-6-driven inflammatory feedback on sleep architecture. The Carroll et al. (2016) data strengthens this — SASP activation was triggered by a single night of restricted sleep, suggesting that even incremental improvements in sleep maintenance could reduce the rate of senescent cell accumulation over time.
Inflammaging is a feature of aging that cannot be eliminated. But the rate at which the self-reinforcing loop between inflammation and sleep fragmentation accelerates can be slowed — and sleep maintenance is one of the more modifiable variables in that loop.
Does Poor Sleep Accelerate Brain Aging?
Glymphatic clearance requires consolidated slow-wave sleep to function. Cerebrospinal fluid flow through brain tissue — the process by which amyloid-beta and tau are removed — increases during deep sleep and decreases during wakefulness and lighter sleep stages. For the full mechanism, see How Does the Glymphatic System Work During Sleep?.
Hong et al. (2005) showed that higher IL-6 correlates with reduced slow-wave sleep percentage (rho = -0.26, p < 0.05) — the sleep stage that glymphatic clearance depends on. When inflammaging reduces deep sleep time, the brain has fewer hours to remove neurotoxic proteins.
The Stahl et al. (2021) elevated IL-6 trajectory group (~9.5 pg/mL, sustained over 15 months) carried inflammatory concentrations that other aging research has associated with accelerated cognitive decline. The compound sequence: inflammaging reduces slow-wave sleep, less slow-wave sleep reduces glymphatic clearance, and reduced clearance allows amyloid-beta and tau to accumulate.
Does Poor Sleep Shorten Lifespan?
The Irwin et al. (2016) meta-analysis — 72 studies, over 50,000 participants — established that sleep disturbance is associated with both IL-6 and CRP elevation. Sustained IL-6 at the concentrations found in the Stahl et al. (2021) elevated trajectory group (~9.5 pg/mL, nearly three-fold higher than the stable group) exceeds thresholds that other aging research has associated with frailty, sarcopenia, and increased mortality risk.
Among the 16% of older adults in the Stahl cohort who maintained these elevated IL-6 concentrations over 15 months, the pattern represents a chronic inflammatory state that persists across months. The connection between sustained inflammation and mortality is epidemiological (it establishes association, not direct causation), but the consistency of the data across large populations and the mechanistic evidence linking IL-6 to multiple aging pathways makes the relationship difficult to attribute to coincidence.
For men over 50 whose sleep is fragmenting, the implication is that the inflammatory consequences of poor sleep maintenance extend beyond daytime fatigue. They intersect with the same inflammatory markers that track with frailty, cognitive decline, and reduced lifespan in aging cohorts.
Inflammaging is one contributor to sleep disruption after 50, but it rarely acts alone. Hormonal changes, metabolic factors, autonomic dysregulation, and circadian changes might all compound the inflammatory pathway. Identifying which contributors might be involved is a practical next step.
Find out which causes might be driving your 3am wakeups →
Related Reading
- Inflammatory Sleep Disruption: How Chronic Inflammation Fragments Your Sleep — Full overview of inflammatory mechanisms that disrupt sleep architecture
- Why Does Histamine Wake You Up at 3am? — How mast cell histamine release drives middle-of-the-night waking
- Can Histamine Intolerance Cause Sleep Problems? — When standard sleep remedies backfire due to histamine overload
- Why Is Inflammation Worse at Night? — The NF-kB/body clock connection that amplifies nighttime inflammation
- How Does Leaky Gut Affect Sleep? — Gut barrier breakdown and its path to sleep-disrupting brain inflammation
- Does Poor Sleep Cause Brain Inflammation? — The neuroinflammation-cognition-sleep connection
- How Does the Glymphatic System Work During Sleep? — Your brain’s overnight waste-clearance system and what disrupts it
- What Is the Connection Between Chronic Inflammation and Insomnia? — The bidirectional cycle linking inflammation to sleeplessness
- Can Inflammation Cause 3am Wakeups?
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- What Is Autoimmune Insomnia and Why Does It Resist Standard Sleep Approaches?
- Can Chronic Stress Cause Insomnia Through Inflammation?
- Gut Bacteria and Insomnia: Which Microbes Affect Your Sleep (and Which Ones Help)
- Does an Anti-Inflammatory Diet Improve Sleep?
References
Franceschi, C., Bonafe, M., Valensin, S., Olivieri, F., De Luca, M., Ottaviani, E., & De Benedictis, G. (2000). Inflamm-aging: An evolutionary perspective on immunosenescence. Annals of the New York Academy of Sciences, 908(1), 244-254. https://pubmed.ncbi.nlm.nih.gov/10911963/
Vgontzas, A. N., Zoumakis, M., Bixler, E. O., Lin, H. M., Prolo, P., Vela-Bueno, A., Kales, A., & Chrousos, G. P. (2003). Impaired nighttime sleep in healthy old versus young adults is associated with elevated plasma interleukin-6 and cortisol levels: Physiologic and therapeutic implications. Journal of Clinical Endocrinology & Metabolism, 88(5), 2087-2095. https://pubmed.ncbi.nlm.nih.gov/12727959/
Hong, S., Mills, P. J., Loredo, J. S., Adler, K. A., & Dimsdale, J. E. (2005). The association between interleukin-6, sleep, and demographic characteristics. Brain, Behavior, and Immunity, 19(2), 165-172. https://pubmed.ncbi.nlm.nih.gov/15664789/
Irwin, M. R., Olmstead, R., & Carroll, J. E. (2016). Sleep disturbance, sleep duration, and inflammation: A systematic review and meta-analysis of cohort studies and experimental sleep deprivation. Biological Psychiatry, 80(1), 40-52. https://pubmed.ncbi.nlm.nih.gov/26140821/
Carroll, J. E., Cole, S. W., Seeman, T. E., Breen, E. C., Witarama, T., Arevalo, J. M. G., Ma, J., & Irwin, M. R. (2016). Partial sleep deprivation activates the DNA damage response (DDR) and the senescence-associated secretory phenotype (SASP) in aged adult humans. Brain, Behavior, and Immunity, 51, 223-229. https://pubmed.ncbi.nlm.nih.gov/26336034/
Stahl, S. T., Smagula, S. F., Rodakowski, J., Dew, M. A., Karp, J. F., Albert, S. M., Butters, M., Gildengers, A., & Reynolds, C. F. 3rd. (2021). Subjective sleep quality and trajectories of interleukin-6 in older adults. American Journal of Geriatric Psychiatry, 29(2), 204-208. https://pubmed.ncbi.nlm.nih.gov/32680764/
Xiao, Q., Qian, J., Evans, D. S., Redline, S., Lane, N. E., Ancoli-Israel, S., Scheer, F. A. J. L., & Stone, K. (2022). Cross-sectional and prospective associations of rest-activity rhythms with circulating inflammatory markers in older men. Journals of Gerontology: Series A, 77(1), 55-65. https://pubmed.ncbi.nlm.nih.gov/33822930/
Piber, D., Olmstead, R., Cho, J. H., & Irwin, M. R. (2023). Disturbance of sleep maintenance, but not sleep duration, activates nuclear factor-kB and signal transducer and activator of transcription family proteins in older adults: Sex differences. Sleep, 46(10), zsad130. https://pubmed.ncbi.nlm.nih.gov/37140651/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 8 references cited
