Brain fog and poor sleep often arrive together — and they share an upstream driver. Neuroinflammation, the activation of the brain’s own immune response, connects the two through a self-reinforcing loop: sleep loss activates brain immune cells, those cells release inflammatory molecules that impair cognition, and the same inflammation fragments the deep sleep stages that would otherwise resolve it. This article covers how sleep restriction triggers neuroinflammation, how that inflammation produces the cognitive impairment commonly described as brain fog, and what happens when the loop runs unchecked. For the broader picture of how inflammation disrupts sleep through multiple pathways, see Inflammatory Sleep Disruption.
How Does Sleep Loss Trigger Brain Inflammation?
The first evidence came from a controlled human experiment. Irwin et al. (2008) kept 14 healthy adults awake from 11pm to 3am for a single night — a four-hour sleep window, the kind of partial night many people experience routinely. By the next morning, NF-κB activation in their peripheral blood mononuclear cells had increased by 30% compared to baseline. NF-κB is the transcription factor that switches on inflammatory gene expression: when it activates, the downstream result is production of pro-inflammatory cytokines including IL-6 and TNF-α.
One night produces the activation. But whether that activation becomes sustained depends on how many nights follow.
Haack et al. (2007) tested what happens across 10 consecutive nights of four-hour sleep in a randomized controlled trial with 18 healthy volunteers. By the end of the restriction period, IL-6 levels were elevated compared to the eight-hour sleep condition (p < 0.05). The IL-6 elevation correlated with increased pain sensitivity (r = 0.67, p < 0.01) — demonstrating that the inflammatory response was not just measurable in blood work but was producing measurable downstream effects.
The 2026 meta-analysis by Ballesio et al. sharpened the threshold. Across 35 human experimental studies (n = 887), multiple nights of partial sleep deprivation — averaging 4.3 hours for three or more consecutive nights — produced an IL-6 effect size of 0.42 (p < 0.01) and a CRP effect size of 0.76 (p = 0.03). A single night of total sleep deprivation did not. This converges with Irwin et al.'s (2016) earlier meta-analysis of 72 studies (n > 50,000), which found the same distinction: chronic sleep disturbance — not isolated total deprivation — drove sustained elevations in CRP and IL-6 across cohort and experimental designs. The pattern is consistent: it is chronic partial restriction, not an isolated bad night, that drives the sustained peripheral inflammation.
The question is how peripheral inflammation reaches the brain. Hu et al. (2024) identified a molecular upstream trigger. In sleep-deprived rats, mitochondria inside microglial cells became damaged, leaking oxidized mitochondrial DNA (mtDNA) into the cell’s cytoplasm. This mtDNA leakage activated NF-κB within the microglia themselves — triggering the same inflammatory cascade, but now inside the brain. When the researchers suppressed mtDNA oxidation, both the mtDNA release and the downstream inflammatory markers decreased. This places mitochondrial stress as a bridge between sleep loss and microglial NF-κB activation.
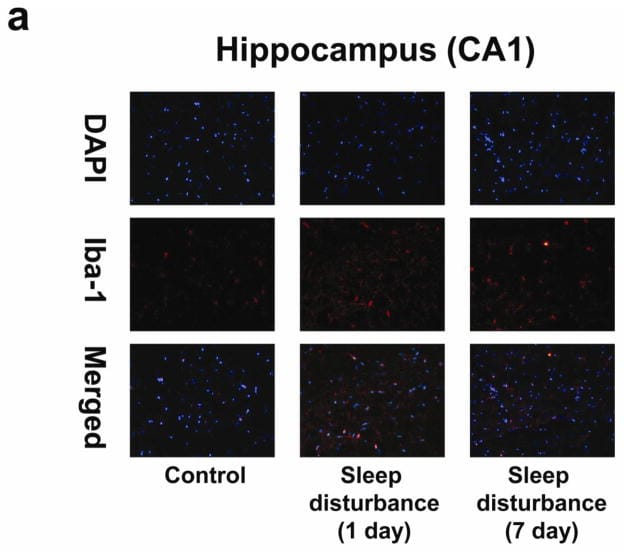
Once microglia activate, the inflammation is not diffuse — it concentrates in the brain regions that matter for cognition and sleep. Zhu et al. (2012) found that 24 hours of sleep disturbance in mice elevated IL-6 and activated microglia (measured by Iba-1 staining) in the hippocampal CA1 and CA3 regions — the areas responsible for memory encoding. The cortex showed no comparable activation. The hippocampus bore the inflammatory burden, and hippocampus-dependent contextual memory was impaired while other memory types were not.
What Is Brain Fog and Is It Caused by Inflammation?
The experience is familiar to anyone who has slept poorly for several consecutive nights: words are harder to retrieve, reading comprehension drops, and routine decisions require disproportionate effort. This is different from ordinary tiredness, which resolves with a nap or a full night of rest. Inflammation-driven cognitive impairment persists even when sleep duration is normal, because the inflammatory milieu in the brain continues to disrupt neural circuits responsible for attention and memory.
You et al. (2024) examined this relationship at population scale using NHANES data from 2011-2014 (n = 2,641; mean age 69.58 years). Participants with severe short sleep — under six hours per night — showed lower cognitive function scores across multiple domains. Independently, a higher immune-inflammation index (SII) predicted worse cognitive performance. The inflammatory burden and the sleep restriction each contributed to cognitive decline, and both were present simultaneously in short sleepers.
The same study included single-cell transcriptomics in mice. Sleep deprivation activated inflammatory and oxidative stress pathways predominantly in GABAergic neurons — the neurons responsible for inhibitory control and cognitive regulation. The enriched pathways overlapped with those seen in Alzheimer’s disease and Huntington’s disease models.
Ballesio et al. (2026) provided the quantitative human evidence for the inflammation itself. Across controlled experimental studies, chronic partial sleep deprivation produced a measurable IL-6 increase (effect size 0.42). IL-6 crosses the blood-brain barrier and acts on neurons and glia in the prefrontal cortex and hippocampus — the same regions where Zhu et al. (2012) found concentrated microglial activation after sleep disturbance. The convergence is direct: sleep restriction raises peripheral IL-6, IL-6 reaches the brain, and the brain regions with the highest IL-6 receptor density are the ones responsible for the cognitive functions that brain fog impairs.
Tiredness resolves with recovery sleep. Inflammation-driven brain fog does not — it requires the inflammatory load itself to decrease, which depends on whether the upstream causes (ongoing sleep restriction, gut inflammation, chronic stress, metabolic imbalance) are addressed.
Can Sleep Deprivation Cause Long-Term Brain Damage?
Under normal conditions, microglia serve a maintenance role. They monitor synapses, remove metabolic waste, and support the neural architecture that produces healthy sleep. But when microglial activation becomes chronic — sustained by weeks or months of poor sleep, ongoing inflammation, or chronic stress — microglia change their functional state.
Rábago-Monzón et al. (2025) reviewed how chronic stress promotes this transition. Sustained stress exposure pushes microglia into a pro-inflammatory phenotype — releasing cytokines that both fragment sleep and impair glymphatic clearance (the brain’s waste-drainage process that runs during deep sleep). The result is a compounding loop: pro-inflammatory microglia fragment sleep, fragmented sleep prevents glymphatic waste removal, accumulated waste perpetuates microglial activation, and the cycle continues.
Bellesi et al. (2017) demonstrated in mice that sleep loss increases astrocytic phagocytosis of synaptic components — astrocytes (the other major glial cell type in the brain) begin consuming parts of synapses at higher rates after sleep deprivation. In parallel, microglial phagocytic activity increases. When this process continues over extended periods, it preferentially targets the synaptic connections that generate slow-wave oscillations and sleep spindles — the electrophysiological signatures of restorative deep sleep.
This microglial over-pruning of synaptic connections creates a forward-feed sequence. Neuroinflammation fragments deep sleep. Fragmented deep sleep reduces glymphatic clearance. Reduced clearance allows inflammatory proteins, amyloid-β, and metabolic waste to accumulate. Accumulation sustains microglial activation. Activated microglia prune synaptic connections. Fewer synaptic connections reduce the brain’s capacity for slow-wave generation.
The Zhu et al. (2012) data showed that microglial activation in the hippocampus persisted at 7 days post-disturbance, even after normal sleep was restored — the brain’s immune response outlasts the sleep disruption that triggered it. But microglial activation is responsive to sustained improvements in sleep and inflammatory load. Reducing ongoing inflammation, improving sleep continuity, and allowing consolidated deep sleep to resume can begin reversing the activated microglial state.
Does Deep Sleep Repair Brain Inflammation?
During consolidated NREM sleep, interstitial space in the brain expands by approximately 60%, allowing cerebrospinal fluid to flow through brain tissue and carry waste products toward the lymphatic drainage routes. This glymphatic process is the brain’s primary clearance mechanism for inflammatory proteins and metabolic byproducts that accumulate during waking hours.
Yang et al. (2024) demonstrated that microglial function follows a circadian pattern that synchronizes with glymphatic activity. During the sleep-dominant phase, microglia move toward a homeostatic, surveillance mode — monitoring synapses and supporting waste clearance rather than releasing inflammatory cytokines. This microglial state transition is not passive. It depends on receiving the electrophysiological input from sustained slow-wave oscillations. When sleep is fragmented and slow-wave periods are shortened or interrupted, microglia do not complete the transition to their restorative state.
The bidirectional relationship between neuroinflammation and deep sleep becomes visible here. Neuroinflammation fragments deep sleep. Fragmented deep sleep prevents microglial state transition. Microglia remain in their activated, inflammatory phenotype. The glymphatic process, which depends on both consolidated slow-wave sleep and microglial cooperation, runs at reduced capacity. Inflammatory proteins that would normally be cleared accumulate. The accumulation sustains the neuroinflammation that fragmented deep sleep in the first place.
The same relationship also runs in the recovery direction. Improving deep sleep quality — through reducing inflammatory load and stabilizing sleep continuity — can initiate a reversal. As slow-wave sleep consolidates, microglia begin transitioning back to homeostatic function, glymphatic clearance improves, and the inflammatory protein load decreases. With less neuroinflammation, sleep fragments less the following night.
Neuroinflammation might be one piece of what is fragmenting your sleep — but it rarely acts alone. It might be compounding with autonomic dysregulation, metabolic factors like blood sugar instability, hormonal changes, or circadian disruption. Identifying which causes might be active in your specific pattern is a useful next step before addressing any one of them.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can Neuroinflammation Cause Sleep Problems?
The relationship is bidirectional. Sleep loss activates microglia, and activated microglia disrupt sleep. Zhu et al. (2012) showed that microglial activation concentrated in the hippocampus — a region involved in both memory encoding and sleep-state regulation. Rábago-Monzón et al. (2025) reviewed how pro-inflammatory microglial states impair glymphatic function, which depends on consolidated deep sleep to function. When microglia are chronically activated, the cytokines they release act on the same neural circuits that generate slow-wave oscillations, reducing both the depth and continuity of NREM sleep.
Is Brain Fog a Sign of Neuroinflammation?
Brain fog has multiple potential contributors: metabolic imbalance, hormonal changes, gut inflammation, and sleep deprivation itself. When neuroinflammation is involved, the pattern tends to persist even after a full night of sleep — because the inflammatory milieu in the brain continues to impair prefrontal and hippocampal function regardless of sleep duration. The You et al. (2024) NHANES data showed that inflammatory indices predicted cognitive decline independently of sleep duration, supporting this distinction.
How Do You Reduce Neuroinflammation?
Consolidated slow-wave sleep allows microglia to return to their homeostatic, non-inflammatory state and enables the glymphatic waste clearance that removes inflammatory proteins from brain tissue (Yang et al., 2024). Improving sleep continuity — reducing awakenings, increasing time in sustained deep sleep — gives the brain the conditions it needs to begin resolving the inflammation.
The other direction is addressing upstream inflammatory sources: gut permeability allows inflammatory molecules to enter circulation and cross the blood-brain barrier, chronic stress maintains elevated cortisol and NF-κB activation, and metabolic imbalance (insulin resistance, elevated blood glucose) contributes its own inflammatory load. Each of these feeds the neuroinflammatory cycle from outside the brain.
What Is Microglial Activation and How Does It Affect Sleep?
In their surveillance (homeostatic) state, microglia monitor neural activity, support synaptic maintenance, and cooperate with glymphatic drainage during deep sleep. In their activated (pro-inflammatory) state — triggered by sleep deprivation, peripheral inflammation, or accumulated metabolic waste — they release IL-6, TNF-α, and IL-1β directly onto sleep-regulatory neurons, reducing the brain’s ability to generate and sustain slow-wave oscillations. Hu et al. (2024) showed that the activation trigger in rats involves mitochondrial damage within microglia: oxidized mtDNA leaks into the cytoplasm and activates NF-κB. The activation is not an on-off switch — it is driven by accumulated mitochondrial stress that builds over consecutive nights of poor sleep.
Related Reading
- Inflammatory Sleep Disruption — the full cause overview covering all five inflammatory mechanisms
- Why Does Histamine Wake You Up at 3am? — how brain-resident mast cells and the circadian histamine peak drive early-morning waking
- Can Histamine Intolerance Cause Sleep Problems? — how DAO deficiency and mast cell activation compound histamine-driven insomnia
- Why Is Inflammation Worse at Night? — how NF-kB and the circadian clock create a pre-dawn inflammatory vulnerability window
- How Does Leaky Gut Affect Sleep? — how gut permeability and LPS translocation trigger neuroinflammation that fragments sleep
- How Does the Glymphatic System Work During Sleep? — how the brain’s waste clearance pathway depends on deep sleep
- What Is the Connection Between Chronic Inflammation and Insomnia? — the wired-but-tired cycle of cytokines, sleep loss, and 24-hour hyperarousal
- Why Do Men Sleep Worse After 50?
- What Is Autoimmune Insomnia and Why Does It Resist Standard Sleep Approaches?
- Can Inflammation Cause 3am Wakeups?
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- Can Chronic Stress Cause Insomnia Through Inflammation?
- Gut Bacteria and Insomnia: Which Microbes Affect Your Sleep (and Which Ones Help)
- Does an Anti-Inflammatory Diet Improve Sleep?
References
1. Ballesio, A., Fiori, V., & Lombardo, C. (2026). Effects of experimental sleep deprivation on peripheral inflammation: An updated meta-analysis of human studies. Journal of Sleep Research, 35, e70099. https://pubmed.ncbi.nlm.nih.gov/40474574/
2. Bellesi, M., de Vivo, L., Chini, M., Gilli, F., Tononi, G., & Cirelli, C. (2017). Sleep loss promotes astrocytic phagocytosis and microglial activation in mouse cerebral cortex. Journal of Neuroscience, 37(21), 5263-5273. https://pubmed.ncbi.nlm.nih.gov/28539349/
3. Haack, M., Sanchez, E., & Mullington, J. M. (2007). Elevated inflammatory markers in response to prolonged sleep restriction are associated with increased pain experience in healthy volunteers. Sleep, 30(9), 1145-1152. https://pubmed.ncbi.nlm.nih.gov/17910386/
4. Hu, Y., Wang, Y., Wang, Y., Zhang, Y., Wang, Z., Xu, X., Zhang, T., Zhang, T., Zhang, S., Hu, R., Shi, L., Wang, X., Li, J., Shen, H., Liu, J., Noda, M., Peng, Y., & Long, J. (2024). Sleep deprivation triggers mitochondrial DNA release in microglia to induce neural inflammation: Preventative effect of hydroxytyrosol butyrate. Antioxidants, 13(7), 833. https://pubmed.ncbi.nlm.nih.gov/39061901/
5. Irwin, M. R., Olmstead, R., & Carroll, J. E. (2016). Sleep disturbance, sleep duration, and inflammation: A systematic review and meta-analysis of cohort studies and experimental sleep deprivation. Biological Psychiatry, 80(1), 40-52. https://pubmed.ncbi.nlm.nih.gov/26140821/
6. Irwin, M. R., Wang, M., Ribeiro, D., Cho, H. J., Olmstead, R., Breen, E. C., Martinez-Maza, O., & Cole, S. (2008). Sleep loss activates cellular inflammatory signaling. Biological Psychiatry, 64(6), 538-540. https://pubmed.ncbi.nlm.nih.gov/18561896/
7. Rábago-Monzón, A. R., et al. (2025). Stress-induced sleep dysregulation: The roles of astrocytes and microglia in neurodegenerative and psychiatric disorders. Biomedicines, 13(5), 1121. https://pubmed.ncbi.nlm.nih.gov/40426947/
8. Yang, J., et al. (2024). Microglia synchronizes with the circadian rhythm of the glymphatic system and modulates glymphatic system function. IUBMB Life, 76(12), 1209-1222. https://pubmed.ncbi.nlm.nih.gov/39223969/
9. You, Y., Li, J., Zhang, Y., Li, X., Li, X., & Ma, X. (2024). Exploring the potential relationship between short sleep risks and cognitive function from the perspective of inflammatory biomarkers and cellular pathways: Insights from population-based and mice studies. CNS Neuroscience & Therapeutics, 30(6), e14783. https://pubmed.ncbi.nlm.nih.gov/38797980/
10. Zhu, B., Dong, Y., Xu, Z., Gompf, H. S., Ward, S. A. P., Xue, Z., Miao, C., Zhang, Y., Chamberlin, N. L., & Xie, Z. (2012). Sleep disturbance induces neuroinflammation and impairment of learning and memory. Neurobiology of Disease, 48(3), 348-355. https://pubmed.ncbi.nlm.nih.gov/22776332/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 10 references cited
