“Wired but exhausted” is a recurring description in insomnia communities — tired enough to collapse, but the brain will not shut down. This maps to a documented inflammatory pattern: pro-inflammatory cytokines that should peak at night to support sleep consolidation are instead peaking during the day, producing daytime fatigue while removing the nighttime cytokine pattern that consolidates sleep.
This article covers the inflammatory mechanism behind that experience — the bidirectional loop between inflammation and insomnia, the cytokine timing inversion that produces daytime exhaustion with nighttime hyperarousal, and the evidence that breaking this cycle from the insomnia side reverses the inflammatory load. For the broader overview of how inflammation disrupts sleep, see the parent article: Inflammatory Sleep Disruption.
How Does Inflammation Cause Insomnia?
The relationship between inflammation and sleep is not linear. IL-1β and TNF-α are classified as sleep-regulatory substances — at low, physiological concentrations, they promote NREM sleep duration. But when concentrations remain elevated chronically, the effect reverses. Chronic cytokine elevation fragments NREM, suppresses REM, and increases cortical arousal.
TNF-α acts locally in the cortex. When applied unilaterally to one hemisphere in animal models, it produces localized slow-wave disruption in that hemisphere only — demonstrating that inflammatory sleep disruption can be region-specific rather than a global arousal state.
The meta-analytic evidence supports this relationship in humans. Irwin et al. (2016) analyzed 72 studies with over 50,000 participants and found that chronic sleep disturbance was associated with elevated IL-6 (effect size 0.20) and CRP (effect size 0.12). These are modest individual effect sizes, but they are consistent across study designs and populations — and they represent sustained, low-grade inflammation rather than an acute immune response.
More recent work has identified one of the molecular pathways involved. Liu et al. (2026) demonstrated that sleep deprivation activates the TLR4/NF-κB pathway — the same innate immune pathway triggered by bacterial infection. This means the brain’s inflammatory response to sleep loss uses the same molecular machinery as its response to pathogens, which may explain why chronic insomnia produces immune markers that resemble chronic infection.
How Does Sleep Loss Cause Inflammation?
The reverse direction of the inflammation-insomnia relationship — sleep loss driving inflammation — is equally well-documented.
Irwin et al. (2008) conducted one of the earliest mechanistic studies. In 14 healthy adults (mean age 51.8 years), a single night of partial sleep deprivation produced a 30% increase in NF-κB activation in peripheral blood mononuclear cells compared to baseline. NF-κB is the primary inflammatory transcription factor — it controls the expression of genes encoding IL-6, TNF-α, and IL-1β. One night was enough to activate it.
The study also revealed a sex difference: the NF-κB response was statistically detectable in female participants but not in male participants. Leukocyte populations did not change, indicating that the effect was direct molecular activation of existing immune cells — not recruitment of new inflammatory cells.
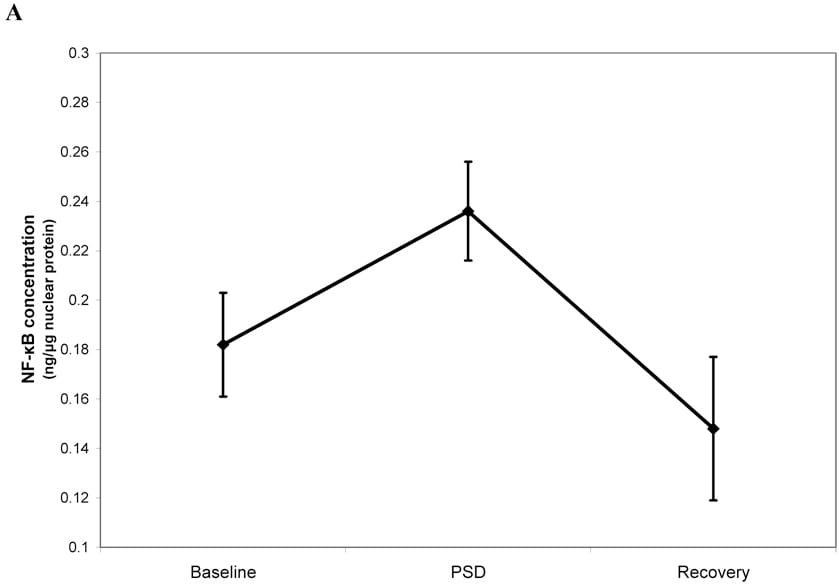
When sleep restriction extends beyond a single night, the inflammatory load accumulates. Haack et al. (2007) randomized healthy adults to either 4-hour or 8-hour sleep conditions for 10 consecutive nights. The 4-hour group showed sustained IL-6 elevation that correlated with heightened pain sensitivity (r=0.67, P<0.01). This is not an abstract biomarker finding — the elevated IL-6 from sleep restriction directly predicted increased physical pain, which itself further fragments sleep.
The speed of this cascade matters. Inflammatory activation from poor sleep does not require weeks or months. Meta-analytic evidence confirms that three or more consecutive nights of restricted sleep produce detectable increases in circulating inflammatory markers (Ballesio et al., 2026). For someone with an existing inflammatory condition, the threshold may be lower.
Why Does Inflammation Make You Tired But Unable to Sleep?
This is the finding that explains the “wired but tired” experience at the molecular level.
Vgontzas et al. (2002) compared 24-hour cytokine secretion patterns in approximately 20 chronic insomnia participants versus healthy controls. In healthy sleepers, IL-6 secretion peaked at approximately 4am and TNF-α followed a regular circadian pattern — both supporting overnight sleep consolidation. In the chronic insomnia group, the IL-6 peak had moved to approximately 7pm, and TNF-α lost its circadian rhythm, instead following a dysregulated 4-hour secretory pulse pattern throughout the day.
The distinction is not just elevated cytokines — it is temporal inversion. The participants with chronic insomnia were not producing more cytokines overall; they were producing them at the wrong time. Daytime cytokine surges drive the fatigue, cognitive impairment, and sensation of exhaustion that characterize chronic insomnia. Flattened nighttime cytokine concentrations remove the immune contribution to sleep consolidation.
The cortisol data adds another dimension. Dressle et al. (2022) conducted a meta-analysis of 20 case-control studies (449 people with insomnia, 357 healthy controls) and found that people with chronic insomnia show moderately elevated cortisol compared to healthy sleepers (standardized mean difference = 0.50). The elevation persists across all measurement windows — daytime, nighttime, and pre-bedtime — consistent with 24-hour hyperarousal rather than a localized nighttime problem. Cortisol elevation during the night, when it should be at its lowest, maintains sympathetic activation: elevated heart rate, heightened alertness, and increased arousal-related neural activity at the hours when these should be declining.
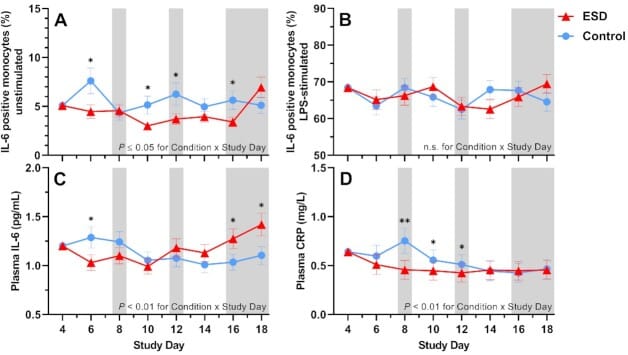
The sex-dependent dimension matters here. Besedovsky et al. (2022) used a controlled 19-day in-hospital study design to examine how prolonged sleep disturbance affects inflammation differently in women and men. In women, peripheral inflammatory markers (IL-6, CRP) decreased — potentially because elevated cortisol suppressed peripheral inflammation while central neuroinflammation may have been increasing. In men, the response was predominantly pro-inflammatory, with reduced cortisol and elevated IL-6 and monocyte cytokine production. This means the “wired but tired” phenotype may be driven by different inflammatory profiles depending on sex, and blood markers alone may not capture the full picture in women.
Can Resolving Insomnia Lower Inflammation?
This is the finding that closes the loop.
Irwin et al. (2015) randomized 123 older adults with insomnia to one of three groups: CBT-I, Tai Chi Chih (a movement-based practice), or a sleep education control. At 4 months, the CBT-I group showed reduced CRP, reduced monocyte production of pro-inflammatory cytokines, and reduced pro-inflammatory gene expression compared to the control group. Both CBT-I and Tai Chi reduced NF-κB and AP-1 transcriptional activity — the same inflammatory switches that sleep loss activates.
The durability of the effect is notable. At 16-month follow-up, the CRP reduction in the CBT-I group persisted. This was not a temporary suppression during the active phase of the study — the inflammatory reversal held after the program ended.
Savin et al. (2023) conducted a review of 15 randomized controlled trials (N=2,067 participants) examining CBT-I effects on cardiometabolic biomarkers. CRP showed the strongest improvement across studies, with three studies showing reductions versus controls. IL-6 and TNF-α effects were more variable across studies, suggesting CRP may be the inflammatory marker that responds best to insomnia resolution.
What this means for the bidirectional cycle: addressing inflammation directly is not required. Behavioral approaches that restructure sleep timing and reduce conditioned arousal — CBT-I being the best-studied — reduce the inflammatory load. Lower inflammation then improves sleep consolidation. The same cycle that amplifies in the negative direction can reverse in the positive direction.
Chronic insomnia is not just a downstream consequence of inflammation. It is an independent, modifiable driver of the inflammatory state. Breaking the insomnia side of the loop reduces the inflammatory load even when the original inflammatory source (gut inflammation, autoimmune conditions, metabolic stress) remains present.
Chronic inflammation might be one of several causes contributing to 3am waking — but it often compounds with other mechanisms. Autonomic dysregulation, hormonal changes, circadian misalignment, or gut permeability might all be active at the same time, each amplifying the others. Identifying which causes might be driving your pattern is a useful next step before deciding where to focus.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can Cytokines Cause Insomnia?
The mechanism is not indirect. TNF-α and IL-1β are classified as somnogenic cytokines — they have direct effects on hypothalamic and brainstem circuits that regulate sleep-wake transitions. Chronic elevation from conditions like autoimmune disease, persistent gut inflammation, or sustained psychological stress pushes these cytokines from their sleep-promoting role into a sleep-fragmenting one. The meta-analytic data from Irwin et al. (2016) — 72 studies, over 50,000 participants — confirms that this association is consistent across populations and study designs.
What Inflammatory Markers Are Associated With Poor Sleep?
TNF-α is also associated with poor sleep, though its measurement in standard blood panels is less standardized than CRP. Fasting insulin and ferritin are indirect markers that can indicate inflammatory states relevant to sleep — elevated fasting insulin reflects metabolic inflammation, and elevated ferritin can indicate iron-driven oxidative stress or chronic inflammatory conditions.
For someone tracking the inflammation-insomnia relationship over time, hs-CRP is the single marker with the broadest evidence base. It is inexpensive, widely available, and has the strongest evidence showing improvement when insomnia improves (Savin et al., 2023).
Does Interleukin-6 Affect Sleep?
The circadian IL-6 inversion is currently the only inflammatory marker with this temporal profile in chronic insomnia. While CRP and TNF-α are also elevated in people with chronic sleep disturbance, IL-6 is the only cytokine where the timing of production — not just the amount — has been shown to invert in chronic insomnia. This temporal inversion distinguishes inflammatory insomnia from other inflammatory states where IL-6 remains elevated but follows its normal circadian pattern.
Can Tumor Necrosis Factor Cause Sleep Problems?
In the chronic insomnia population studied by Vgontzas et al. (2002), TNF-α lost its normal circadian rhythm and instead followed an irregular 4-hour secretory pulse pattern throughout the day. This loss of TNF-α circadian patterning, combined with the IL-6 temporal inversion, contributes to the 24-hour dysregulation of immune and arousal pathways that maintains the “wired but tired” state.
Related Reading
- Inflammatory Sleep Disruption — the full cause overview covering all five inflammatory mechanisms
- Why Does Histamine Wake You Up at 3am? — how brain-resident mast cells and the circadian histamine peak drive early-morning waking
- Can Histamine Intolerance Cause Sleep Problems? — how DAO deficiency and mast cell activation compound histamine-driven insomnia
- Why Is Inflammation Worse at Night? — how NF-kB and the circadian clock create a pre-dawn inflammatory vulnerability window
- How Does Leaky Gut Affect Sleep? — how gut permeability and LPS translocation trigger neuroinflammation that fragments sleep
- Does Poor Sleep Cause Brain Inflammation? — the bidirectional loop between neuroinflammation, brain fog, and sleep loss
- How Does the Glymphatic System Work During Sleep? — how the brain’s waste clearance pathway depends on deep sleep
- Why Do Men Sleep Worse After 50?
- What Is Autoimmune Insomnia and Why Does It Resist Standard Sleep Approaches?
- Can Inflammation Cause 3am Wakeups?
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- Can Chronic Stress Cause Insomnia Through Inflammation?
- Gut Bacteria and Insomnia: Which Microbes Affect Your Sleep (and Which Ones Help)
- Does an Anti-Inflammatory Diet Improve Sleep?
References
Ballesio, A., Fiori, V., & Lombardo, C. (2026). Effects of experimental sleep deprivation on peripheral inflammation: An updated meta-analysis of human studies. Journal of Sleep Research, 35(1), e70099. https://pubmed.ncbi.nlm.nih.gov/40474574/
Besedovsky, L., Dang, R., Engert, L. C., Goldstein, M. R., Devine, J. K., Bertisch, S. M., Mullington, J. M., Simpson, N., & Haack, M. (2022). Differential effects of an experimental model of prolonged sleep disturbance on inflammation in healthy females and males. PNAS Nexus, 1(1), pgac004. https://pubmed.ncbi.nlm.nih.gov/36380854/
Dressle, R. J., Feige, B., Spiegelhalder, K., Schmucker, C., Benz, F., Mey, N. C., & Riemann, D. (2022). HPA axis activity in patients with chronic insomnia: A systematic review and meta-analysis of case-control studies. Sleep Medicine Reviews, 62, 101588. https://pubmed.ncbi.nlm.nih.gov/35091194/
Haack, M., Sanchez, E., & Mullington, J. M. (2007). Elevated inflammatory markers in response to prolonged sleep restriction are associated with increased pain experience in healthy volunteers. Sleep, 30(9), 1145-1152. https://pubmed.ncbi.nlm.nih.gov/17910386/
Irwin, M. R., Wang, M., Ribeiro, D., Cho, H. J., Olmstead, R., Breen, E. C., Martinez-Maza, O., & Cole, S. (2008). Sleep loss activates cellular inflammatory signaling. Biological Psychiatry, 64(6), 538-540. https://pubmed.ncbi.nlm.nih.gov/18561896/
Irwin, M. R., Olmstead, R., Breen, E. C., Witarama, T., Carrillo, C., Sadeghi, N., Arevalo, J. M. G., Ma, J., Nicassio, P., Bootzin, R., & Cole, S. (2015). Cognitive behavioral therapy and tai chi reverse cellular and genomic markers of inflammation in late-life insomnia: A randomized controlled trial. Biological Psychiatry, 78(10), 721-729. https://pubmed.ncbi.nlm.nih.gov/25748580/
Irwin, M. R., Olmstead, R., & Carroll, J. E. (2016). Sleep disturbance, sleep duration, and inflammation: A systematic review and meta-analysis of cohort studies and experimental sleep deprivation. Biological Psychiatry, 80(1), 40-52. https://pubmed.ncbi.nlm.nih.gov/26140821/
Liu, T., Zhang, Y., Li, X., Liu, S., Zhu, X., Ren, Y., Han, R., Liu, K., Sun, C., Ji, X., & Li, X. (2026). Sleep deprivation-induced TLR4/MyD88/NF-κB pathway dysregulation worsens cognitive impairment in Parkinson’s Disease. Behavioural Brain Research, 504, 116058. https://pubmed.ncbi.nlm.nih.gov/41577012/
Savin, K. L., Clark, T. L., Perez-Ramirez, P., Allen, T. S., Parra, M. T., & Gallo, L. C. (2023). The effect of cognitive behavioral therapy for insomnia (CBT-I) on cardiometabolic health biomarkers: A systematic review of randomized controlled trials. Behavioral Sleep Medicine, 21(6), 671-694. https://pubmed.ncbi.nlm.nih.gov/36476211/
Vgontzas, A. N., Zoumakis, M., Papanicolaou, D. A., Bixler, E. O., Prolo, P., Lin, H. M., Vela-Bueno, A., Kales, A., & Chrousos, G. P. (2002). Chronic insomnia is associated with a shift of interleukin-6 and tumor necrosis factor secretion from nighttime to daytime. Metabolism, 51(7), 887-892. https://pubmed.ncbi.nlm.nih.gov/12077736/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 10 references cited
