Men experiencing fragmented sleep after 40 often don’t connect it to hormonal changes. Testosterone declines 1-2% per year from around age 30, and sleep quality declines in parallel — but the relationship between them is bidirectional, not just age-related. Each one degrades the other.
This article covers how low testosterone affects sleep architecture, how poor sleep suppresses testosterone production, what the evidence shows about the amount of sleep needed to maintain testosterone levels, and why this feedback loop tends to worsen over time. It does not cover cortisol-testosterone interactions or GABA-related mechanisms — those are addressed in the cortisol article and in Does GABA Affect Testosterone and Sleep in Men?. Testosterone and sleep is one of several hormonal mechanisms behind sleep disruption in men; for the broader picture, see Hormonal Sleep Disruption in Men.
How Does Low Testosterone Affect Sleep Architecture in Men?
The largest polysomnographic study on this topic comes from the MrOS cohort (Barrett-Connor et al., 2008). Researchers measured serum testosterone in 1,312 community-dwelling men aged 65 and older, then assessed their sleep 3.4 years later using both 72-hour wrist actigraphy and one-night in-home polysomnography. Men with lower total testosterone had reduced sleep efficiency, woke up more often during the night, and spent less time in slow-wave sleep. They also had a higher apnea-hypopnea index and more cumulative time with oxygen saturation below 90%.
What makes this study useful is the distinction it draws between sleep duration and sleep architecture. Total sleep time did not differ by testosterone level. The differences were in sleep depth and continuity — the structural quality of sleep, not the number of hours.
There is an important caveat. These associations were attenuated after adjusting for body mass index and waist circumference. Visceral fat contains aromatase, an enzyme that converts testosterone to estradiol. Men carrying more visceral fat tend to have both lower testosterone and worse sleep, making adiposity a mediating variable in this relationship. The testosterone-sleep connection is documented, but body composition is part of the mechanism.
More recent data from a U.S. claims database (Agrawal et al., 2024) quantified the association differently. In men aged 40-70, insomnia was associated with a 1.74-fold increase in odds of testosterone deficiency after propensity score matching for age, BMI, hypertension, diabetes, and other comorbidities. Circadian rhythm disorders carried the highest odds — 2.63-fold — consistent with mechanistic data showing testosterone production requires both intact sleep architecture and appropriate circadian entrainment.
Beyond architecture, low testosterone can disrupt sleep through vasomotor instability — the same mechanism behind hot flashes in menopausal women. Gonzalez et al. (2018) documented that men on androgen deprivation therapy experienced night sweats as a primary sleep disruptor, and nocturia (waking to urinate) was also more frequent, both contributing to fragmented sleep independent of sleep stage changes.
Does Poor Sleep Lower Testosterone in Men?
The landmark study on this topic is Leproult and Van Cauter (2011). Ten young healthy men (mean age 24) spent one week sleeping 10 hours per night (rested condition), then one week sleeping 5 hours per night (restricted condition), with blood sampled every 15-30 minutes across full 24-hour periods. Daytime testosterone was 10-15% lower after the sleep-restricted week. The testosterone profiles diverged in the afternoon and evening, with the restricted group showing consistently lower values from early afternoon onward.
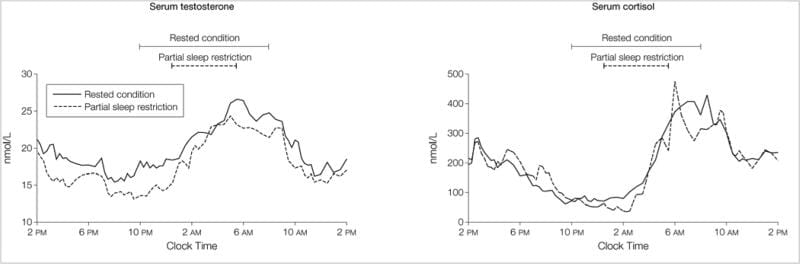
A 10-15% decline is not trivial. For context, normal age-related testosterone decline runs 1-2% per year. One week of short sleep compressed over a decade of hormonal aging into seven days.
Slow-wave sleep integrity matters independently of total sleep time. Ukraintseva et al. (2018) isolated the specific sleep stage responsible. In a randomized crossover design, 12 healthy men slept one night undisturbed and one night with acoustic tones that selectively suppressed slow-wave sleep by 54% — without reducing total sleep time or waking the men fully. Morning testosterone was lower after the slow-wave-suppressed night (p = 0.017), and 17-alpha-hydroxyprogesterone — a direct precursor in the testosterone synthesis pathway — was also reduced (p = 0.011). Cortisol did not change. The suppression was specific to testosterone production rather than a generalized stress response.
Sleep fragmentation matters through a different mechanism. Luboshitzky et al. (2001) placed 10 healthy men on a 24-hour ultrashort sleep-wake cycle designed to fragment sleep architecture without reducing total sleep time. Overall daily testosterone production was preserved, but the nocturnal testosterone rise — the surge that normally begins during the first sleep cycle — was delayed by approximately 5 hours. In the 6 men who had no REM sleep during the fragmented condition, the nocturnal testosterone spike was abolished. Only the 4 men who achieved REM sleep showed the expected rise. This links testosterone release to the appearance of REM sleep specifically.
A meta-analysis by Kim and Cho (2021) pooled 18 studies involving 252 healthy men. Total sleep deprivation (24+ hours of wakefulness) produced a consistent testosterone reduction that scaled with duration — the effect size grew from 24 hours to 40-48 hours. Partial sleep deprivation (reduced but not eliminated sleep) showed a trend in the same direction but did not reach statistical significance. This distinction matters: a single night of 5-6 hours may not acutely suppress testosterone, but chronic short sleep does.
As for recovery — Liu and Reddy (2022) note that testosterone can recover after acute sleep loss, but chronic restriction may produce a sustained downward pressure on androgen levels, particularly in aging men whose baseline production is already declining.
How Much Sleep Do Men Need to Maintain Testosterone?
Testosterone production during sleep depends on reaching and maintaining specific sleep stages. The Ukraintseva (2018) data demonstrated that slow-wave sleep drives androgen synthesis — suppress it by 54%, and morning testosterone drops even when total sleep time is unchanged. The Luboshitzky (2001) data showed that REM sleep is required for the nocturnal testosterone surge to occur. Both stages are necessary.
This has practical implications for the “how many hours” question. Sleep architecture is front-loaded: slow-wave sleep dominates the first half of the night, while REM sleep concentrates in the second half. Cutting sleep short — going to bed late or waking early — disproportionately cuts REM time, which is when the nocturnal testosterone rise occurs.
The Leproult data showed that 5 hours per night for one week was sufficient to produce a 10-15% testosterone decline. The Kim meta-analysis found that partial restriction (6-hour range) did not produce a statistically detectable acute effect, while total deprivation did. The practical threshold for sustained testosterone maintenance likely falls in the 7-hour range, though individual variation exists.
Age complicates the picture. Hernandez-Perez et al. (2024) analyzed NHANES data from 8,748 adults and found the sleep-testosterone relationship is nonlinear across the lifespan. In young men aged 20-40, short sleep (6 hours or less) was paradoxically associated with elevated testosterone (OR = 3.62) — likely reflecting acute stress-axis activation or selection effects in healthier short sleepers. In middle-aged men aged 41-64, longer sleep was associated with low testosterone (OR = 2.03), which may reflect hypersomnia secondary to low testosterone or underlying conditions driving both extended sleep and hormonal suppression.
The question is whether the sleep you are getting includes enough consolidated slow-wave sleep and REM sleep to support the androgen synthesis pathway. Fragmented 8-hour sleep may produce less testosterone than consolidated 7-hour sleep with normal architecture.
Why Does the Testosterone-Sleep Cycle Get Worse Over Time?
The testosterone-sleep feedback loop works like this: lower testosterone is associated with less slow-wave sleep and more nighttime awakenings (Barrett-Connor, 2008). Less slow-wave sleep suppresses the testosterone synthesis pathway (Ukraintseva, 2018). Lower testosterone then further degrades sleep quality. Each rotation makes both the hormonal decline and the sleep disruption worse.
Age does not cause this feedback loop, but it accelerates it. Three processes converge in midlife. First, testosterone declines 1-2% per year from around age 30 as part of normal reproductive aging. Second, slow-wave sleep declines independently with age — men lose deep sleep even when testosterone is normal. Third, visceral fat tends to increase, and adipose tissue contains aromatase, which converts testosterone to estradiol. More visceral fat means more testosterone-to-estradiol conversion, less available testosterone, and — as Barrett-Connor showed — this adiposity partially explains the testosterone-sleep association.
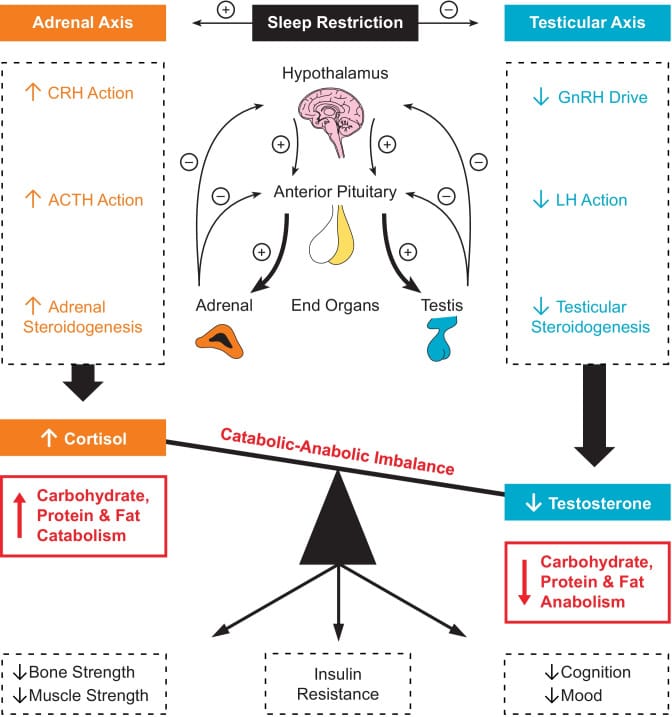
Liu and Reddy (2022) frame this as a catabolic-anabolic imbalance. Sleep restriction simultaneously suppresses testosterone (the primary anabolic hormone) and elevates afternoon cortisol (catabolic). The result tilts the hormonal environment toward catabolism — reduced protein synthesis, increased insulin resistance, lower bone density. Their review highlights a novel dual-hormone clamping approach in which normalizing both cortisol and testosterone during sleep-restricted conditions mitigated the insulin resistance that sleep loss normally produces. This demonstrates that the metabolic consequences of poor sleep are partly mediated through the testosterone-cortisol ratio.
Stress amplifies the testosterone-sleep feedback loop. Hirokawa et al. (2022) studied Japanese workers and found that elevated cortisol suppressed the protective relationship between testosterone and sleep efficiency. In men with low cortisol, higher testosterone was associated with better sleep. In men with high cortisol, that protective relationship disappeared. Chronic stress — through cortisol elevation — can override testosterone’s sleep-supporting effects.
There is also causal evidence that testosterone directly modulates sleep architecture. Morssinkhof et al. (2023) followed a transgender cohort receiving testosterone therapy and found that exogenous testosterone reshaped sleep architecture within 3 months. This moves beyond the correlational data from observational studies and supports a direct causal role for testosterone in determining sleep quality.
The practical implication: by the time a man notices worsening sleep in his 40s or 50s, the testosterone-sleep feedback loop may have been running for years. Low testosterone, declining slow-wave sleep, increasing visceral fat, and accumulated sleep debt can all be contributing simultaneously — and each one feeds the others.
Low testosterone and fragmented sleep compound each other, but they rarely act alone. Cortisol elevation, GABA receptor changes, metabolic disruption, and circadian misalignment might also be contributing to sleep disruption. Men experiencing sleep maintenance problems after 40 often have more than one mechanism at work. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Sleeping More Increase Testosterone?
The distinction between sleep duration and sleep architecture is consistent across the evidence. The Ukraintseva (2018) study showed that testosterone production depends on slow-wave sleep integrity. Luboshitzky et al. (2001) demonstrated that without REM sleep, the nocturnal testosterone rise does not occur — regardless of how many total hours are logged.
This means “sleep more” is an incomplete recommendation. If the additional sleep is fragmented — interrupted by awakenings, apneas, or environmental disruption — it may not support testosterone synthesis. The more precise goal is consolidated sleep with preserved slow-wave and REM stages.
How Do You Address Low Testosterone Sleep Problems?
Because the relationship is bidirectional, intervening on either side can create positive momentum. Improving sleep continuity — by addressing whatever is fragmenting it — gives the androgen synthesis pathway more slow-wave sleep and REM time to work with. If testosterone levels are measurably low, addressing that directly can improve sleep architecture, which in turn supports further testosterone production.
The complicating factor is that sleep disruption in men over 40 rarely has a single cause. Cortisol elevation, GABA receptor changes, blood sugar instability, and circadian misalignment can all fragment sleep independently. Each of these reduces slow-wave sleep, which reduces testosterone, which further degrades sleep. Identifying which upstream causes are active determines the starting point. For how GABA and testosterone interact to maintain sleep, see Does GABA Affect Testosterone and Sleep in Men?.
Does Sleep Apnea Affect Testosterone?
Apnea events cause micro-arousals that fragment sleep architecture and reduce the amount of time spent in slow-wave sleep and REM — the two stages required for nocturnal testosterone production. Barrett-Connor et al. (2008) found that lower testosterone was associated with both a higher apnea-hypopnea index and more cumulative time with oxygen desaturation below 90%, suggesting that both the sleep fragmentation and the intermittent hypoxia contribute to testosterone suppression.
Liu and Reddy (2022) note that obstructive sleep apnea correlates with reduced testosterone independent of both age and body weight. High-dose exogenous testosterone may worsen sleep apnea, though physiological replacement dosing has not been as consistently implicated — a consideration for men with both conditions.
Does Low Testosterone Cause Night Sweats?
Gonzalez et al. (2018) documented this in men undergoing androgen deprivation therapy for prostate cancer. The abrupt withdrawal of testosterone produced vasomotor effects — including night sweats and hot flashes — that became a primary cause of sleep disruption. While androgen deprivation therapy represents an extreme reduction, the mechanism is the same one that acts at a lower intensity in men with naturally declining testosterone. Vasomotor instability does not require testosterone to reach zero — it can begin as levels drop below individual thresholds.
Night sweats from low testosterone tend to cluster in the first half of the night and can cause full awakenings that are difficult to recover from, compounding the sleep architecture disruption that low testosterone produces through other pathways.
Related Reading
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, growth hormone, and GABA interact to fragment sleep in men
- Can Low GABA Cause Waking Up at 3am? — The GABAergic mechanism behind 3am waking
- What Are the Signs of Low GABA at Night? — How to recognize impaired GABAergic function
- Do GABA Supplements Help You Stay Asleep Through the Night? — Trial evidence for oral GABA and the blood-brain barrier question
- Does GABA Affect Testosterone and Sleep in Men? — How testosterone-derived neurosteroids modulate GABA-A receptors
- How Do You Increase GABA Levels Naturally for Better Sleep? — Exercise, yoga, fermented foods, and gut health for GABA support
- Can a Cortisol Spike Wake You Up at 3am? — Cortisol-testosterone axis and nocturnal waking
- Does Growth Hormone Decline Affect Your Sleep After 40? — GH-SWS coupling and age-related decline
- Does Andropause Cause Insomnia? What Men Over 50 Need to Know — Compound hormonal decline in aging men
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep? — How visceral fat suppresses testosterone through aromatase and disrupts sleep
- Can Ultra-Processed Food Lower Testosterone and Disrupt Sleep? — How endocrine-disrupting chemicals in processed food suppress testosterone and degrade sleep
- Can Inflammation Suppress Testosterone and Disrupt Your Sleep? — How inflammatory cytokines suppress testosterone production at the brain and testicular level
- Does Testosterone Replacement Therapy Affect Sleep? — How TRT affects sleep quality, its interaction with sleep apnea, and the role of body weight
References
Agrawal, P., Singh, S. M., Able, C., Delijani, K., Javier, J., Bhasin, S., & Seftel, A. D. (2024). Sleep disorders are associated with testosterone deficiency and erectile dysfunction — a U.S. claims database analysis. International Journal of Impotence Research, 36, 315-321. https://pubmed.ncbi.nlm.nih.gov/36473958/
Barrett-Connor, E., Dam, T., Stone, K., Harrison, S. L., Redline, S., & Orwoll, E. (2008). The association of testosterone levels with overall sleep quality, sleep architecture, and sleep-disordered breathing. Journal of Clinical Endocrinology & Metabolism, 93(7), 2602-2609. https://pubmed.ncbi.nlm.nih.gov/18413429/
Gonzalez, B. D., et al. (2018). Sleep disturbance in men receiving androgen deprivation therapy for prostate cancer: The role of hot flashes and nocturia. Cancer, 124(3), 499-506. https://pubmed.ncbi.nlm.nih.gov/29072790/
Hernandez-Perez, J. G., Zamudio-Haas, S., Torres-Flores, D., Lopez-Ramos, O., & Gutierrez-Ospina, G. (2024). Association of sleep duration and quality with serum testosterone concentrations among men and women: NHANES 2011-2016. Andrology, 12(3), 456-467. https://pubmed.ncbi.nlm.nih.gov/37452666/
Hirokawa, K., Sakamoto, T., Kasahara, S., & Murata, C. (2022). Associations of testosterone and cortisol concentrations with sleep quality in Japanese male workers. Comprehensive Psychoneuroendocrinology, 12, 100159. https://pubmed.ncbi.nlm.nih.gov/36148025/
Kim, S. D., & Cho, K. S. (2021). Effect of partial and total sleep deprivation on serum testosterone in healthy males: a systematic review and meta-analysis. Sleep Medicine Reviews, 58, 101429. https://pubmed.ncbi.nlm.nih.gov/34801825/
Leproult, R., & Van Cauter, E. (2011). Effect of 1 week of sleep restriction on testosterone levels in young healthy men. JAMA, 305(21), 2173-2174. https://pubmed.ncbi.nlm.nih.gov/21632481/
Liu, P. Y., & Reddy, R. T. (2022). Sleep, testosterone and cortisol balance, and ageing men. Reviews in Endocrine and Metabolic Disorders, 23, 1323-1339. https://pubmed.ncbi.nlm.nih.gov/36152143/
Luboshitzky, R., Zabari, Z., Shen-Orr, Z., Herer, P., & Lavie, P. (2001). Disruption of the nocturnal testosterone rhythm by sleep fragmentation in normal men. Journal of Clinical Endocrinology & Metabolism, 86(3), 1134-1139. https://pubmed.ncbi.nlm.nih.gov/11238497/
Morssinkhof, M. W. L., et al. (2023). Influence of sex hormone use on sleep architecture in a transgender cohort. Sleep, 46(12), zsad271. https://pubmed.ncbi.nlm.nih.gov/37715990/
Ukraintseva, Y. V., Liaukovich, K. M., Saltykov, K. A., Belov, D. A., & Nizhnik, A. N. (2018). Selective slow-wave sleep suppression affects testosterone and 17α-hydroxyprogesterone secretion. Sleep Medicine, 48, 117-122. https://pubmed.ncbi.nlm.nih.gov/29894840/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 11 references cited
