Three orexin receptor antagonists are now approved for insomnia: suvorexant (Belsomra), lemborexant (Dayvigo), and daridorexant (Quviviq). They represent a mechanistically different category from zolpidem (Ambien), eszopiclone (Lunesta), and benzodiazepines. Most people who take them — or consider taking them — do not understand why these drugs feel different from the sedative-hypnotics that preceded them, or why one DORA might work when another does not.
This article covers how DORAs work at the receptor level, how they compare to GABA-targeting medications on sleep architecture, and how the three approved DORAs differ from each other. It does not cover what orexin is or how the circadian clock gates orexin activity — see What Is Orexin and Why Does It Keep You Awake When Your Body Clock Says Sleep?. For lifestyle-based approaches to modulating orexin, see Can You Lower Orexin Naturally? What Controls Your Brain’s Wake Drive.
Medication decisions belong with a physician. This article explains the receptor-level mechanisms and the trial evidence behind the options. For the broader circadian picture, see the parent pillar on Circadian Rhythm Disruption and Sleep.
How Do Dual Orexin Receptor Antagonists Work Differently From Ambien at the Receptor Level?
Zolpidem binds the alpha-1 subunit of the GABA-A receptor — a chloride ion channel found on neurons throughout the brain. When zolpidem binds, the channel opens more readily in response to GABA (gamma-aminobutyric acid, the brain’s primary inhibitory neurotransmitter), increasing chloride ion flow into the neuron and hyperpolarizing it. The result is rapid, widespread neuronal suppression. This is why zolpidem induces sedation within 15–30 minutes, but it is also why it produces amnesia, parasomnias (sleepwalking, sleep-eating), and impaired cognition — GABA-A receptors are distributed across cortical, hippocampal, and brainstem regions, and zolpidem suppresses activity in all of them.
Benzodiazepines work through a GABA-A mechanism but bind multiple subunits (alpha-1, alpha-2, alpha-3, alpha-5), producing an even broader range of effects: anxiolysis, muscle relaxation, amnesia, and sedation. Benzodiazepines also suppress both slow-wave sleep (SWS) and REM sleep — they change not only whether you sleep, but the composition of your sleep.
DORAs take a different approach. Suvorexant, lemborexant, and daridorexant are competitive antagonists at the OX1R and OX2R receptors — the two receptor subtypes that orexin-A and orexin-B bind to maintain wakefulness. By blocking these receptors, DORAs reduce the excitatory input that orexin neurons deliver to the brain’s arousal centers: the locus coeruleus, the tuberomammillary nucleus, the dorsal raphe, and the basal forebrain cholinergic neurons. The effect is specific to the wake-promoting circuit rather than a broad suppression of neural activity.
When orexin receptors are blocked, the brain’s endogenous sleep-promoting neurons — the ventrolateral preoptic area (VLPO) neurons that release GABA and galanin — can gain dominance over the arousal centers without pharmacological assistance. The sleep that follows uses the brain’s own machinery. With zolpidem, GABA-A activation forces neuronal suppression regardless of what the endogenous sleep circuitry is doing. This is why people taking DORAs often describe their sleep as feeling more natural.
De Crescenzo et al. (2022) conducted the largest network meta-analysis of insomnia pharmacotherapy to date, covering 154 randomized controlled trials and 44,089 adults. The analysis compared benzodiazepines, Z-drugs, DORAs, melatonin receptor agonists, and other drug classes across efficacy and tolerability endpoints. DORAs showed efficacy for both sleep onset and sleep maintenance, while benzodiazepines and Z-drugs had higher rates of discontinuation due to adverse events — consistent with their broader receptor engagement and wider side effect profiles.
How Do Dual Orexin Receptor Antagonists Affect Sleep Architecture Compared to Z-Drugs?
The most direct head-to-head comparison comes from the SUNRISE 1 trial. Moline et al. (2021) analyzed sleep architecture data from 1,006 adults aged 55 and older with insomnia randomized to lemborexant 5 mg, lemborexant 10 mg, zolpidem extended-release 6.25 mg, or placebo for 30 nights with polysomnographic (PSG) recording. The primary finding: lemborexant at both doses increased REM sleep duration and decreased REM latency (the time from first falling asleep to the first REM period) compared to both placebo and zolpidem. Zolpidem did not improve REM latency at all. Lemborexant also produced greater increases in total sleep time compared to zolpidem.
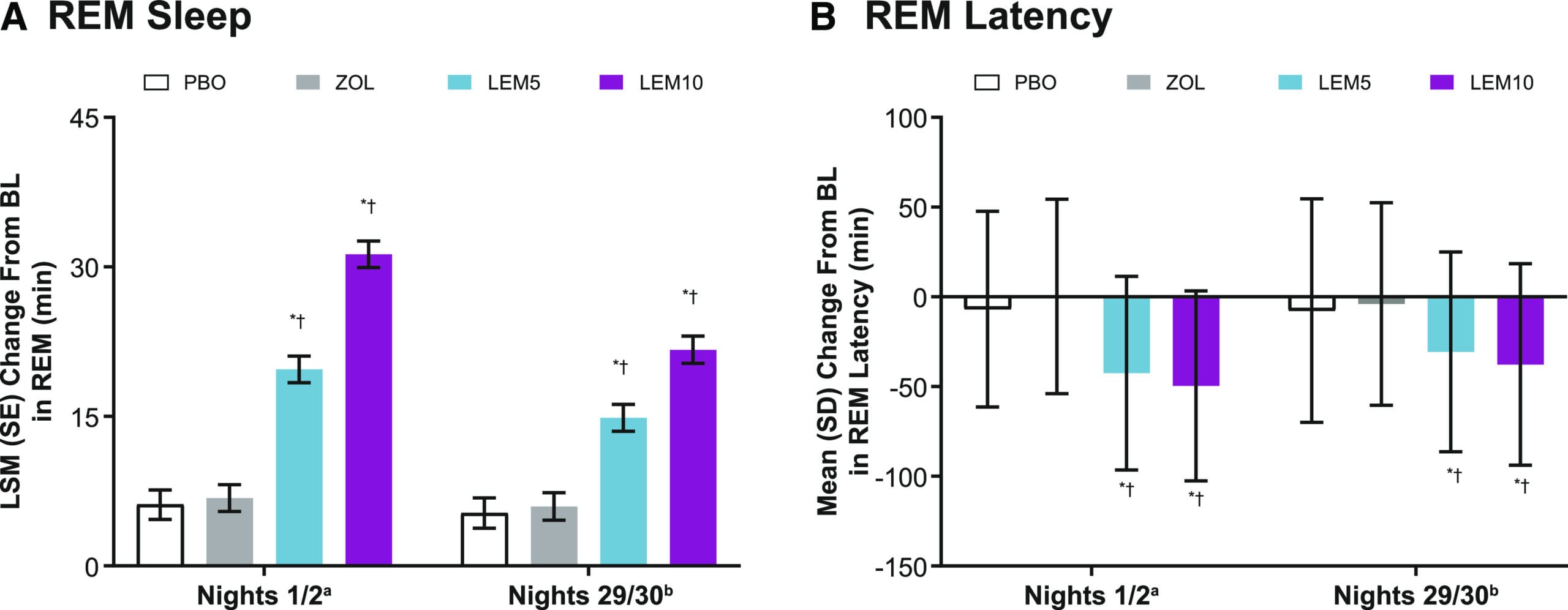
This REM-enhancing effect is the opposite of what GABA-targeting drugs do. Benzodiazepines suppress both REM and SWS. Zolpidem is less disruptive than benzodiazepines but still does not enhance REM sleep. DORAs, by reducing the orexin-mediated wake drive, allow the brain to cycle into REM more readily and stay in REM longer — because orexin normally suppresses REM-promoting neurons in the sublaterodorsal tegmental nucleus.
Snyder et al. (2016) pooled polysomnographic data from 1,482 people in suvorexant phase 3 trials. Suvorexant increased REM sleep by up to 3.9% of total sleep time. SWS proportions were preserved — suvorexant did not reduce deep sleep. Small spectral power changes (reductions in higher-frequency activity during NREM) appeared on the first night but were transient, normalizing by subsequent recordings.
Di Marco et al. (2024) analyzed pooled PSG data from two phase 3 trials of daridorexant 50 mg. At month 3, daridorexant increased REM sleep duration by 16.21 minutes compared to placebo. The analysis also showed reduced relative beta power during wakefulness and N1 sleep — beta activity (13–30 Hz) is a well-established EEG marker of cortical hyperarousal, and its reduction suggests that daridorexant addressed the hyperarousal component of insomnia rather than sedating the brain. Sleep spindle density, spindle amplitude, and slow-wave activity — markers of normal NREM microarchitecture — were not affected by daridorexant. The drug increased sleep time and reduced arousal without degrading sleep quality at the microstructural level.
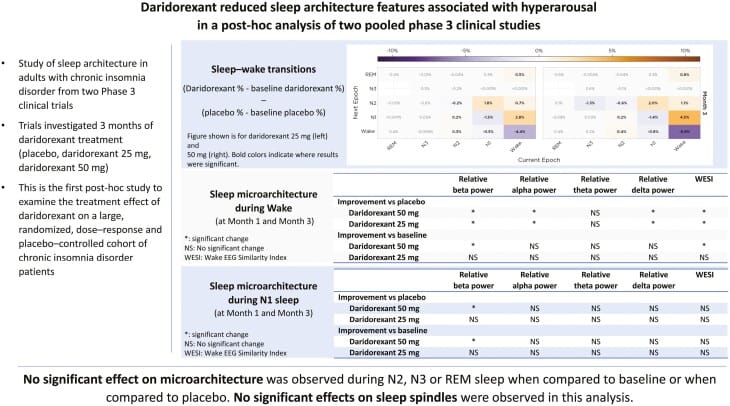
The pattern across all three DORAs is consistent: more REM sleep, faster entry into REM, preserved deep sleep, and reduced markers of hyperarousal. This is a fundamentally different pharmacological fingerprint from GABA-A agonists.
How Do the Three Dual Orexin Receptor Antagonists Differ From Each Other?
Kishi et al. (2025) published a network meta-analysis of 8 randomized controlled trials encompassing 5,198 adults (mean age 56.33 years, 67.84% female) comparing all three DORAs at their approved doses. At month 1, lemborexant 10 mg produced the greatest improvement in subjective sleep onset latency — how long people felt it took them to fall asleep. Daridorexant 50 mg produced the greatest improvement in subjective total sleep time — how long people felt they slept. All three DORAs outperformed placebo across both endpoints.
These outcome differences trace back to pharmacological differences.
Receptor selectivity. All three drugs block both OX1R and OX2R, but with different binding ratios. Lemborexant has approximately 8-fold stronger affinity for OX2R compared to OX1R. Suvorexant and daridorexant bind both receptor subtypes with more balanced affinity. This matters because OX2R is more involved in NREM-to-REM transitions and in the maintenance of consolidated wakefulness, while OX1R plays a stronger role in wake-to-NREM transitions and in the regulation of emotional and reward-related arousal. Lemborexant’s stronger OX2R preference may contribute to its faster effect on sleep onset.
Half-life. Daridorexant has a half-life of approximately 8 hours — it was designed to have enough duration to maintain sleep through the night while washing out by morning. Suvorexant has a half-life of approximately 12 hours. Lemborexant has the longest and most variable half-life at 17–55 hours, meaning it accumulates with repeated dosing and washes out more slowly. Half-life affects next-morning residual effects: a longer half-life increases the probability of morning drowsiness, while a shorter half-life may reduce coverage in the final hours of sleep.
Dosing adjustments. Suvorexant has specific age-adjusted dosing: 15 mg is the recommended starting dose for adults 65 and older, compared to 10 mg or 20 mg in younger adults. This reflects age-related changes in drug metabolism and the higher sensitivity of older adults to orexin receptor blockade.
What the trial data do not resolve. The Kishi et al. meta-analysis shows population-level differences in efficacy endpoints, but individual responses vary based on metabolism, the specific nature of the person’s insomnia (sleep onset vs. sleep maintenance vs. both), and concurrent medications. The receptor selectivity and half-life differences provide pharmacological rationale for why one DORA might work better than another for a given individual, but predicting which one will work best for a specific person requires a physician’s judgment.
This article does not recommend specific medications. It explains the mechanistic differences so readers can have a more informed conversation with their physician.
How Do Dual Orexin Receptor Antagonists Compare on Dependency and Side Effects?
Abuse potential is assessed in a specific way: recreational drug users — people with histories of sedative misuse — receive the study drug and rate how much they “like” it on a visual analog scale (VAS). These scores determine a drug’s DEA scheduling classification.
Ufer et al. (2022) tested daridorexant in 63 recreational sedative users. At the approved 50 mg dose, daridorexant produced lower drug-liking VAS scores than both suvorexant 150 mg and zolpidem 30 mg (supratherapeutic comparator doses used per standard abuse potential study design). At supratherapeutic doses (100–150 mg, two to three times the maximum approved dose), the scores converged — all drugs produced comparable liking at high doses. This is a standard finding across sedative classes: at multiples of the therapeutic dose, most sedating compounds produce euphoria.
Landry et al. (2022) tested lemborexant in 32 recreational sedative users. Lemborexant drug-liking at the approved 10 mg dose was not statistically different from zolpidem 30 mg or suvorexant 40 mg. All three DORAs and zolpidem are classified as Schedule IV controlled substances by the DEA.
An important distinction: abuse potential scores in recreational users measure one specific dimension — euphoria and drug-liking in people who are seeking those effects. This does not translate directly to the side-effect experience of people taking these medications for insomnia at standard doses. The side effects that matter in daily use come from the receptor mechanisms themselves.
DORA side effects trace back to REM enhancement. Because DORAs allow more REM sleep and faster REM entry, the most commonly reported side effects are vivid dreams and, less commonly, transient sleep paralysis or hypnagogic hallucinations (visual or auditory experiences at the boundary between waking and sleep). These effects are consequences of increased REM expression — the same mechanism that makes DORAs beneficial for sleep architecture.
Z-drug side effects trace back to GABA-A suppression. Zolpidem’s most concerning adverse effects — amnesia, complex sleep behaviors (sleepwalking, sleep-driving, sleep-eating with no memory of the event), and next-morning cognitive impairment — result from broad GABA-A receptor activation in the hippocampus (memory consolidation) and prefrontal cortex (executive function). These are not idiosyncratic reactions; they are predictable consequences of the drug’s mechanism acting on brain regions where GABA-A receptors are dense.
De Crescenzo et al. (2022), in their 154-trial network meta-analysis, found that benzodiazepines and Z-drugs had higher discontinuation rates due to adverse events compared to DORAs. This population-level finding is consistent with the narrower receptor target of DORAs producing fewer off-target effects.
Medication choice is one component of sleep management. Whether orexin overactivity, melatonin decline, cortisol timing, metabolic factors, or inflammatory causes might be contributing to your sleep disruption affects which approaches are likely to help. Identifying the contributing causes is a useful first step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Is Ambien a Dual Orexin Receptor Antagonist?
Z-drugs were introduced in the 1990s as alternatives to benzodiazepines. Targeting a single GABA-A subunit (alpha-1) reduced some benzodiazepine side effects — less muscle relaxation, less anxiolysis — but the amnesia and parasomnia risks persisted because the alpha-1 subunit is still widely expressed in memory and executive function regions.
DORAs represent a conceptual departure: instead of imposing sedation by amplifying inhibition, they reduce the wake-promoting input and allow the brain’s own sleep machinery to take over.
Why Do Dual Orexin Receptor Antagonists Cause Vivid Dreams?
This effect is the pharmacological opposite of what happens with Z-drugs and benzodiazepines. GABA-A agonists suppress REM sleep, reducing dream content during use. When people stop taking them, REM sleep rebounds — often producing intense, disturbing nightmares as the brain compensates for the period of REM suppression. With DORAs, vivid dreams occur during use (because REM is enhanced from the start) and there is no REM rebound on discontinuation because REM was never suppressed.
Can You Switch From Ambien to a Dual Orexin Receptor Antagonist?
Zolpidem produces dependence through GABA-A receptor adaptation — the brain downregulates its own GABA sensitivity in response to chronic exogenous stimulation. When zolpidem is stopped, the reduced endogenous GABA sensitivity can produce rebound insomnia and, in some cases, anxiety and agitation. DORAs do not act on the GABA pathway, so they do not address GABA withdrawal. A person switching from zolpidem to a DORA may experience rebound insomnia from GABA withdrawal even while the DORA reduces orexin-mediated wake drive. The two mechanisms are independent, and the transition requires medical oversight.
Related Reading:
- Circadian Rhythm Disruption and Sleep — The full cause reference page covering all five circadian mechanisms
- What Is Orexin and Why Does It Keep You Awake When Your Body Clock Says Sleep?
- How Does Orexin Change With Age? Why Sleep Fragments More After 50
- How Melatonin Suppresses Orexin: The Push-Pull That Decides Whether You Sleep or Stay Awake
- How Stress Hijacks Your Orexin: Why “Wired but Tired” Is a Real Brain State
- Can You Lower Orexin Naturally? What Controls Your Brain’s Wake Drive
References
De Crescenzo, F., D’Alò, G. L., Ostinelli, E. G., Ciabattini, M., Di Franco, V., Watanabe, N., … & Cipriani, A. (2022). Comparative effects of pharmacological interventions for the acute and long-term management of insomnia disorder in adults: A systematic review and network meta-analysis. The Lancet, 400(10347), 170–184. https://pubmed.ncbi.nlm.nih.gov/35843245/
Di Marco, T., Djonlagic, I., Dauvilliers, Y., Sadeghi, K., Little, D., Datta, A. N., … & Scammell, T. E. (2024). Effect of daridorexant on sleep architecture in patients with chronic insomnia disorder: A pooled post hoc analysis of two randomized phase 3 clinical studies. SLEEP, 47(11), zsae098. https://pubmed.ncbi.nlm.nih.gov/38644625/
Kishi, T., Ikuta, T., Citrome, L., Sakuma, K., Hatano, M., Hamanaka, S., Nishii, Y., & Iwata, N. (2025). Comparative efficacy and safety of daridorexant, lemborexant, and suvorexant for insomnia: A systematic review and network meta-analysis. Translational Psychiatry, 15(1), 211. https://pubmed.ncbi.nlm.nih.gov/40555730/
Landry, I., Hall, N., Aluri, J., Filippov, G., Reyderman, L., Setnik, B., Henningfield, J., & Moline, M. (2022). Abuse potential of lemborexant, a dual orexin receptor antagonist, compared with zolpidem and suvorexant in recreational sedative users. Journal of Clinical Psychopharmacology, 42(4), 365–373. https://pubmed.ncbi.nlm.nih.gov/35749758/
Moline, M., Zammit, G., Cheng, J. Y., Perdomo, C., Kumar, D., & Mayleben, D. (2021). Comparison of the effect of lemborexant with placebo and zolpidem tartrate extended release on sleep architecture in older adults with insomnia disorder. Journal of Clinical Sleep Medicine, 17(6), 1167–1174. https://pubmed.ncbi.nlm.nih.gov/33590823/
Snyder, E., Ma, J., Svetnik, V., Connor, K. M., Lines, C., Michelson, D., & Herring, W. J. (2016). Effects of suvorexant on sleep architecture and power spectral profile in patients with insomnia: Analysis of pooled phase 3 data. Sleep Medicine, 19, 93–100. https://pubmed.ncbi.nlm.nih.gov/27198953/
Ufer, M., Kelsh, D., Schoedel, K. A., & Dingemanse, J. (2022). Abuse potential assessment of the new dual orexin receptor antagonist daridorexant in recreational sedative drug users as compared to suvorexant and zolpidem. SLEEP, 45(3), zsab224. https://pubmed.ncbi.nlm.nih.gov/34480579/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
