The discovery of the glymphatic system in 2012 changed the understanding of why sleep is biologically necessary — the brain physically cleans itself during deep sleep. Since then, human studies have confirmed that sleep deprivation acutely increases waste protein levels in cerebrospinal fluid. This article covers how the glymphatic system works, which sleep stage activates it, what happens when clearance fails, and what the emerging human evidence shows about sleep-dependent waste removal. It does not cover the full inflammatory sleep disruption picture — that is in the parent article. The glymphatic system is one mechanism within a broader set of inflammatory causes. For the full overview of how inflammation fragments sleep, see Inflammatory Sleep Disruption.
What Is the Glymphatic System and Why Does It Need Sleep?
The term “glymphatic” combines “glial” and “lymphatic” — a glial-cell-dependent drainage pathway that performs a lymphatic-like function inside the brain, where no conventional lymphatic vessels exist.
Iliff et al. (2012) mapped the pathway: cerebrospinal fluid (CSF) enters brain tissue along perivascular spaces surrounding penetrating arteries, flows through the interstitial space, and exits along paravenous drainage routes. The movement depends on aquaporin-4 (AQP4) water channels concentrated on astrocytic endfeet — the extensions of astrocyte cells that wrap around blood vessels. In mice lacking AQP4, interstitial solute clearance dropped by approximately 70% (Iliff et al., 2012).
Xie et al. (2013) demonstrated that the glymphatic system is approximately twice as active during sleep compared to wakefulness. Using in vivo two-photon imaging in mice, they found that the interstitial space expanded by roughly 60% during sleep — astrocytes physically shrank, widening the channels for fluid flow. Amyloid-beta was cleared twice as fast during sleep as during waking.
The reason sleep is required is neurochemical. During wakefulness, norepinephrine — a neurotransmitter involved in arousal — keeps astrocytes in an expanded state that narrows the interstitial channels. When norepinephrine drops during NREM sleep, astrocytes contract, the interstitial space opens, and CSF can flow through the tissue to carry waste out. CSF enters along arteries, mixes with interstitial fluid to pick up metabolic waste (amyloid-beta, tau, lactate), and drains out along veins. The entire process depends on the interstitial space being open — and that opening requires the low-norepinephrine, low-arousal state of deep sleep.
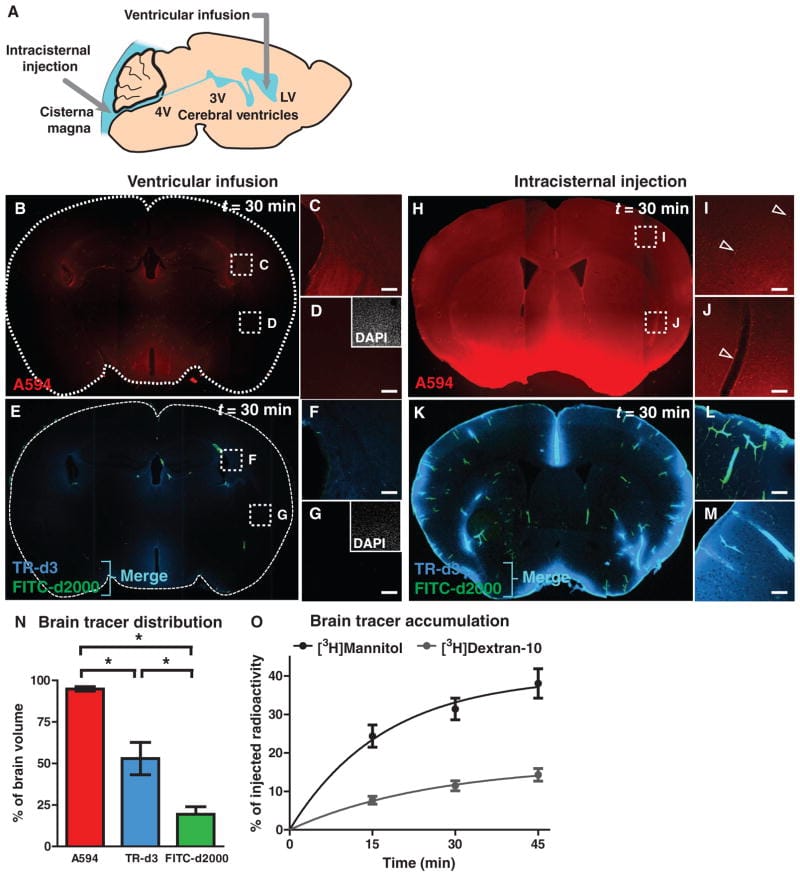
What Stage of Sleep Is Best for Glymphatic Clearance?
The connection between slow-wave sleep and glymphatic clearance is mechanical. The large delta waves (0.5-4 Hz) characteristic of NREM stage 3 produce coordinated fluctuations in cerebral blood volume. These fluctuations create pressure gradients that physically push CSF through the interstitial space.
Hauglund et al. (2025), published in Cell, identified the mechanical driver. Infraslow norepinephrine fluctuations originating from the locus coeruleus — a brainstem nucleus involved in arousal regulation — drive rhythmic arterial constriction and dilation during NREM sleep. This vasomotion acts as a pump: as arteries rhythmically expand and contract, CSF is pushed into and through the perivascular spaces. Using optogenetic stimulation of the locus coeruleus in mice, the researchers produced coordinated vasomotion and CSF dynamics — confirming that these oscillations drive fluid movement. Synchronized oscillations in norepinephrine, cerebral blood volume, and CSF were the strongest predictors of glymphatic clearance efficiency.
One finding from the Hauglund study carries relevance for people taking sleep medications. In the mouse model, zolpidem — a widely prescribed sleep aid — suppressed norepinephrine oscillations and impaired glymphatic flow. The drug induced sleep, but the type of sleep it produced lacked the vasomotor dynamics needed for waste clearance. Whether this translates to humans at standard doses is not yet confirmed, but the mechanism raises a question about whether medication-induced sleep supports the same degree of brain waste removal as unmedicated slow-wave sleep.
Human evidence supports the NREM-clearance link. Ju et al. (2017) experimentally disrupted slow-wave activity in 17 healthy adults using auditory tones delivered during NREM — without reducing total sleep time. The disruption of slow-wave activity correlated with increased CSF amyloid-beta40 levels (r = 0.610, P = 0.009). The effect was selective for slow-wave disruption and selective for amyloid-beta — total sleep efficiency, tau, and other CSF proteins did not change. This demonstrated in humans that the quality of NREM slow-wave sleep — not sleep duration — predicts amyloid-beta clearance.
A 2025 randomized crossover trial added further human evidence. Lyckenvik et al. (2025) measured CSF amyloid-beta and tau in 12 healthy adults across three conditions: a night of sleep, total sleep deprivation, and an afternoon control. CSF amyloid-beta and tau were consistently lower after sleep compared to deprivation. The clearance was selective — neurofilament light chain and glial fibrillary acidic protein did not change, indicating protein-selective clearance rather than bulk dilution.
What Happens When the Glymphatic System Does Not Work Properly?
Holth et al. (2019) measured CSF tau in healthy adults using lumbar catheter sampling during sleep and during sleep deprivation. After a single night without sleep, CSF tau increased by 51.5%. In the mouse arm of the same study, interstitial fluid tau increased approximately 90% during normal wakefulness compared to sleep, and approximately 100% during sleep deprivation. Chronic sleep deprivation in transgenic mice accelerated the spreading of tau pathology — sustained sleep loss promoted the propagation of tau aggregation across brain regions.
Aging compounds the problem. Kress et al. (2014) found that glymphatic clearance of amyloid-beta was impaired by 40% in old mice compared to young mice. The underlying changes included a 27% reduction in arterial wall pulsatility and widespread mislocalization of AQP4 water channels — AQP4 becomes redistributed away from astrocytic endfeet, where it facilitates fluid exchange, across the broader astrocyte surface, reducing its effectiveness.
Nedergaard and Goldman (2020), in a Science review, framed impaired glymphatic clearance as “a final common pathway” across multiple dementias — Alzheimer’s, Parkinson’s, and vascular dementia. Impaired glymphatic flow leads to stagnation of extracellular proteins, promoting misfolding and seeding of amyloid-beta, tau, and alpha-synuclein. The review also noted that cardiovascular disease — by reducing arterial pulsatility — indirectly impairs glymphatic function, connecting vascular health to brain waste clearance.
The forward-feeding loop is the central concern. Poor sleep reduces glymphatic clearance. Reduced clearance allows amyloid-beta and tau to accumulate. Accumulated amyloid-beta activates microglia — the brain’s resident immune cells — which release pro-inflammatory cytokines including TNF-alpha and IL-6. Those cytokines fragment sleep architecture, reducing slow-wave sleep. Less slow-wave sleep means less clearance, more accumulation, more inflammation, and progressively worse sleep.
This is not a theoretical concern reserved for older adults. Holth et al. (2019) demonstrated the tau increase after a single night of sleep deprivation in healthy younger adults. Even short periods of poor sleep engage the acute end of this accumulation — whether the full feedback loop develops depends on duration and whether sleep quality recovers.
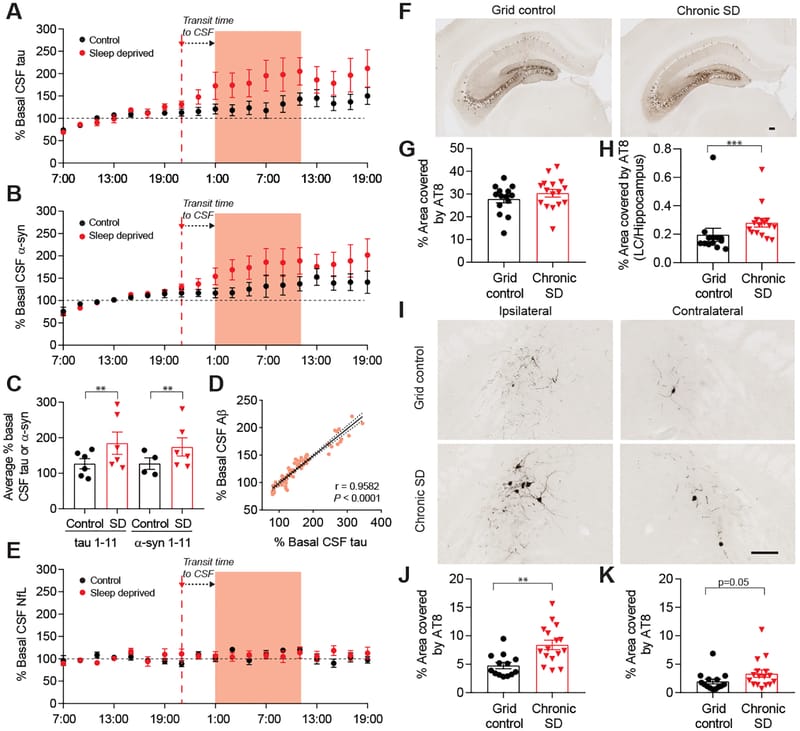
Can You Improve Glymphatic Function?
The research points to two categories of modifiable factors:
Factors that support glymphatic clearance:
- Slow-wave sleep quality. This is the primary determinant. Factors that increase time in NREM stage 3 and delta oscillation amplitude support the vasomotor dynamics that drive CSF flow. Reducing sleep fragmentation — from inflammation, cortisol dysregulation, histamine excess, or environmental disruption — has a downstream effect on glymphatic efficiency.
- Sleep position. Animal studies suggest lateral (side) sleeping may facilitate glymphatic transport compared to supine or prone positions, through gravity-assisted drainage via cervical lymphatic routes. Human evidence on this is limited, and no controlled human trials have confirmed the effect.
- Exercise. In animal models, regular physical activity is associated with improved glymphatic clearance, through increased arterial pulsatility, reduced neuroinflammation, and improved cardiovascular health. Human data on exercise and glymphatic function specifically is still emerging.
Factors that impair glymphatic clearance:
- Alcohol. Even moderate alcohol consumption disrupts sleep architecture, reducing time in slow-wave sleep and fragmenting sleep continuity. Both effects reduce the window during which glymphatic clearance can run.
- Sleep fragmentation from any cause. Each arousal from deep sleep interrupts the low-norepinephrine state the interstitial space needs to remain expanded. Frequent micro-arousals — whether from sleep apnea, pain, environmental sound, or inflammatory activation — reduce the cumulative time the glymphatic pathway has to move fluid.
- Aging-related AQP4 changes. The mislocalization of AQP4 away from astrocytic endfeet (Kress et al., 2014) is age-dependent and not yet directly modifiable — but vascular health, neuroinflammation, and sleep quality all influence the process.
Glymphatic health is downstream of sleep quality. Addressing whatever is fragmenting or shallowing deep sleep is the direct route to supporting brain waste clearance. For many people, that means identifying inflammatory causes of sleep disruption — which is the focus of the broader Inflammatory Sleep Disruption overview.
Impaired glymphatic clearance and neuroinflammation might be one piece of what is disrupting your sleep — but they often compound with other causes. Glymphatic impairment may overlap with histamine-driven waking, circadian dysregulation, or hormonal disruption, each of which might be independently fragmenting deep sleep or amplifying the inflammatory loop. Identifying which causes might be active in your pattern is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Deep Sleep Detox the Brain?
The term “detox” in this context refers to the physical flushing of metabolic byproducts — amyloid-beta, tau, lactate — through CSF movement along the glymphatic pathway. Xie et al. (2013) demonstrated that this clearance runs approximately twice as fast during sleep compared to wakefulness, with the 60% expansion of interstitial space providing the physical channels for fluid transport.
The process is not detoxification in the liver-enzyme sense. CSF physically carries waste out of brain tissue and into the cervical lymphatic drainage for processing. There is no chemical breakdown of waste molecules within the brain itself during this process.
Can Poor Sleep Cause Alzheimer’s?
Nedergaard and Goldman (2020) described impaired glymphatic clearance as “a final common pathway” leading to dementia — one mechanism through which multiple risk factors (poor sleep, cardiovascular disease, aging) converge on the same outcome. The evidence includes: 51.5% increased CSF tau after one night of sleep deprivation (Holth et al., 2019), 40% reduced amyloid-beta clearance in aging brains (Kress et al., 2014), and increased CSF amyloid-beta from slow-wave disruption alone in healthy adults (Ju et al., 2017).
Alzheimer’s is a multifactorial condition. Impaired glymphatic clearance from poor sleep is one contributor — but one with a mechanistic chain now supported by both animal and human evidence.
What Waste Products Does the Brain Flush During Sleep?
These are not foreign substances. Amyloid-beta is a byproduct of amyloid precursor protein processing, tau is a microtubule-associated protein released into interstitial fluid during neuronal activity, and lactate is a metabolic byproduct of glucose utilization. They accumulate during wakefulness as a normal consequence of neural activity (Holth et al., 2019).
The problem arises when clearance does not keep pace with production. Lyckenvik et al. (2025) demonstrated that clearance during sleep is protein-selective: amyloid-beta and tau are cleared, while neurofilament light chain and glial fibrillary acidic protein are not affected — indicating that the glymphatic system preferentially clears certain metabolic waste rather than bulk-flushing everything.
How Does Amyloid Beta Relate to Sleep?
During waking hours, neural activity produces amyloid-beta at a rate roughly proportional to the level of that activity. During NREM slow-wave sleep, the glymphatic system clears amyloid-beta at approximately twice the waking rate (Xie et al., 2013). When sleep is insufficient or fragmented, amyloid-beta accumulates in the interstitial space — and Ju et al. (2017) showed that disrupting slow-wave activity for a single night, without changing total sleep time, increased CSF amyloid-beta in healthy adults.
The bidirectional component: accumulated amyloid-beta activates microglia and cytokine release, which fragment subsequent sleep, reducing the clearance window further. Each incomplete clearance cycle leaves a larger residual amyloid-beta burden for the next night.
Does Norepinephrine Drive Glymphatic Flow During Sleep?
Hauglund et al. (2025) found that during NREM sleep, norepinephrine does not drop to zero — it oscillates at infraslow frequencies. These oscillations, originating from the locus coeruleus, drive rhythmic constriction and dilation of cerebral arteries. That vasomotion creates the pressure differentials that push CSF through perivascular spaces.
The requirement is not the absence of norepinephrine but rather its oscillation at the frequency and amplitude that drive vasomotion — conditions that occur during consolidated NREM slow-wave sleep. When arousal disrupts this oscillation pattern, the vasomotor pump loses its rhythm and CSF flow decreases. This is why zolpidem impaired glymphatic flow in the Hauglund study: the drug suppressed the norepinephrine oscillations driving the clearance pump.
Related Reading
- Inflammatory Sleep Disruption — the full cause overview covering all five inflammatory mechanisms
- Why Does Histamine Wake You Up at 3am? — how brain-resident mast cells and the circadian histamine peak drive early-morning waking
- Can Histamine Intolerance Cause Sleep Problems? — how DAO deficiency and mast cell activation compound histamine-driven insomnia
- Why Is Inflammation Worse at Night? — how NF-kB and the circadian clock create a pre-dawn inflammatory vulnerability window
- How Does Leaky Gut Affect Sleep? — how gut permeability and LPS translocation trigger neuroinflammation that fragments sleep
- Does Poor Sleep Cause Brain Inflammation? — the bidirectional loop between neuroinflammation, brain fog, and sleep loss
- What Is the Connection Between Chronic Inflammation and Insomnia? — the wired-but-tired cycle of cytokines, sleep loss, and 24-hour hyperarousal
- Why Do Men Sleep Worse After 50?
- What Is Autoimmune Insomnia and Why Does It Resist Standard Sleep Approaches?
- Can Inflammation Cause 3am Wakeups?
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- Can Chronic Stress Cause Insomnia Through Inflammation?
- Gut Bacteria and Insomnia: Which Microbes Affect Your Sleep (and Which Ones Help)
- Does an Anti-Inflammatory Diet Improve Sleep?
References
1. Iliff, J. J., Wang, M., Liao, Y., Plogg, B. A., Peng, W., Gundersen, G. A., Benveniste, H., Vates, G. E., Deane, R., Goldman, S. A., Nagelhus, E. A., & Nedergaard, M. (2012). A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid beta. Science Translational Medicine, 4(147), 147ra111. https://pubmed.ncbi.nlm.nih.gov/22896675/
2. Xie, L., Kang, H., Xu, Q., Chen, M. J., Liao, Y., Thiyagarajan, M., O’Donnell, J., Christensen, D. J., Nicholson, C., Iliff, J. J., Takano, T., Deane, R., & Nedergaard, M. (2013). Sleep drives metabolite clearance from the adult brain. Science, 342(6156), 373-377. https://pubmed.ncbi.nlm.nih.gov/24136970/
3. Kress, B. T., Iliff, J. J., Xia, M., Wang, M., Wei, H. S., Zeppenfeld, D., Xie, L., Kang, H., Xu, Q., Liew, J. A., Plog, B. A., Ding, F., Deane, R., & Nedergaard, M. (2014). Impairment of paravascular clearance pathways in the aging brain. Annals of Neurology, 76(6), 845-861. https://pubmed.ncbi.nlm.nih.gov/25204284/
4. Ju, Y.-E. S., Ooms, S. J., Sutphen, C., Macauley, S. L., Zangrilli, M. A., Jerome, G., Fagan, A. M., Mignot, E., Zempel, J. M., Claassen, J. A. H. R., & Holtzman, D. M. (2017). Slow wave sleep disruption increases cerebrospinal fluid amyloid-beta levels. Brain, 140(8), 2104-2111. https://pubmed.ncbi.nlm.nih.gov/28899014/
5. Holth, J. K., Fritschi, S. K., Wang, C., Pedersen, N. P., Cirrito, J. R., Mahan, T. E., Finn, M. B., Manis, M., Geerling, J. C., Fuller, P. M., Lucey, B. P., & Holtzman, D. M. (2019). The sleep-wake cycle regulates brain interstitial fluid tau in mice and CSF tau in humans. Science, 363(6429), 880-884. https://pubmed.ncbi.nlm.nih.gov/30679382/
6. Hauglund, N. L., Andersen, M., Tokarska, K., Radovanovic, T., Kjaerby, C., Sorensen, F. L., Bojarowska, Z., Untiet, V., Ballestero, S. B., Kolmos, M. G., Weikop, P., Hirase, H., & Nedergaard, M. (2025). Norepinephrine-mediated slow vasomotion drives glymphatic clearance during sleep. Cell, 188, 606-622.e17. https://pubmed.ncbi.nlm.nih.gov/39788123/
7. Lyckenvik, T., Olsson, M., Forsberg, M., Wasling, P., Zetterberg, H., Hedner, J., & Hanse, E. (2025). Sleep reduces CSF concentrations of beta-amyloid and tau: A randomized crossover study in healthy adults. Fluids and Barriers of the CNS, 22(1), 84. https://pubmed.ncbi.nlm.nih.gov/40830882/
8. Nedergaard, M., & Goldman, S. A. (2020). Glymphatic failure as a final common pathway to dementia. Science, 370(6512), 50-56. https://pubmed.ncbi.nlm.nih.gov/33004510/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 8 references cited
