The gut-sleep connection is one of the more underappreciated intersections in health research — and the mechanism linking them runs through inflammation.
When the intestinal lining becomes permeable, bacterial endotoxins reach the bloodstream. From there, they activate the same inflammatory cascade — NF-kB, IL-6, TNF-alpha — that fragments sleep architecture. And the relationship feeds itself: poor sleep degrades the gut microbiome within days, which increases permeability further.
This article covers four pieces of that pathway: how gut-derived inflammation reaches the brain, how short-chain fatty acids regulate sleep, what sleep deprivation does to gut bacteria, and why the timing of gut-driven inflammation aligns with early morning waking. For the broader inflammatory sleep picture — including cortisol, histamine, and neuroinflammation — the full framework is in Inflammatory Sleep Disruption.
How Does Gut Inflammation Reach the Brain?
The first route is bloodstream translocation. When intestinal permeability increases, LPS — fragments of gram-negative bacterial cell walls — enter the bloodstream. LPS binds to TLR4 receptors on circulating immune cells and on microglia in the brain. That binding activates NF-kB, which drives production of IL-6, TNF-alpha, and other pro-inflammatory cytokines. These are the same cytokines that fragment sleep architecture when elevated at night.
A 2023 meta-analysis of eight studies (n=897) found that the severity of sleep-disordered breathing tracked with gut barrier biomarkers — LPS, LPS-binding protein, and zonulin levels all correlated with the apnea-hypopnea index (Mashaqi et al., 2023). The relationship between compromised gut barriers and disrupted sleep held across multiple study designs.
The second route is the vagus nerve. In a mouse model, Zhang et al. (2021) found that sleep deprivation after LPS exposure elevated plasma IL-6 and TNF-alpha while reducing anti-inflammatory IL-10. Subdiaphragmatic vagotomy — severing the vagal connection between gut and brain — abolished this amplification of inflammatory cytokines. This demonstrated that the vagus nerve is the relay carrying gut-derived inflammatory information to the brain.
The third route involves short-chain fatty acid depletion. When dysbiosis reduces populations of butyrate-producing bacteria, the brain loses a compound that supports GABA synthesis — GABA being the primary inhibitory neurotransmitter involved in sleep initiation and maintenance. This third pathway connects to the next question.
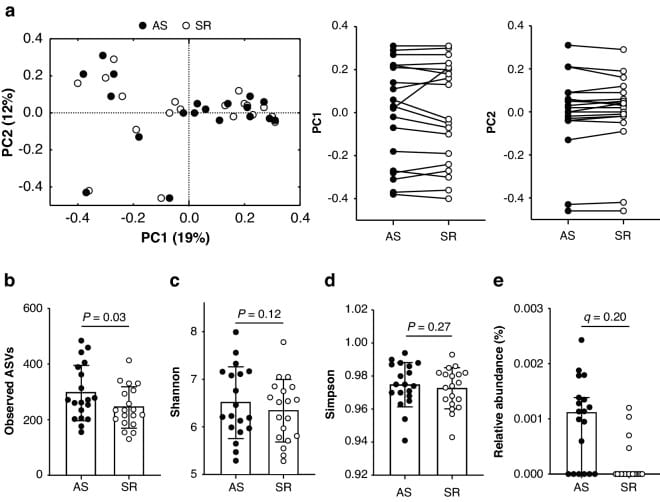
Karl et al. (2023) ran a crossover design with 19 healthy men: three nights of two hours of sleep versus three nights of seven to nine hours, with controlled diet throughout. The sleep-restricted condition reduced gut microbiota community richness by approximately 21%.
How Do Short-Chain Fatty Acids Affect Sleep?
Butyrate is produced in the colon by bacterial fermentation of dietary fiber. It supports intestinal barrier integrity, feeds colonocytes (the cells lining the colon), and — through a less obvious pathway — promotes non-REM sleep.
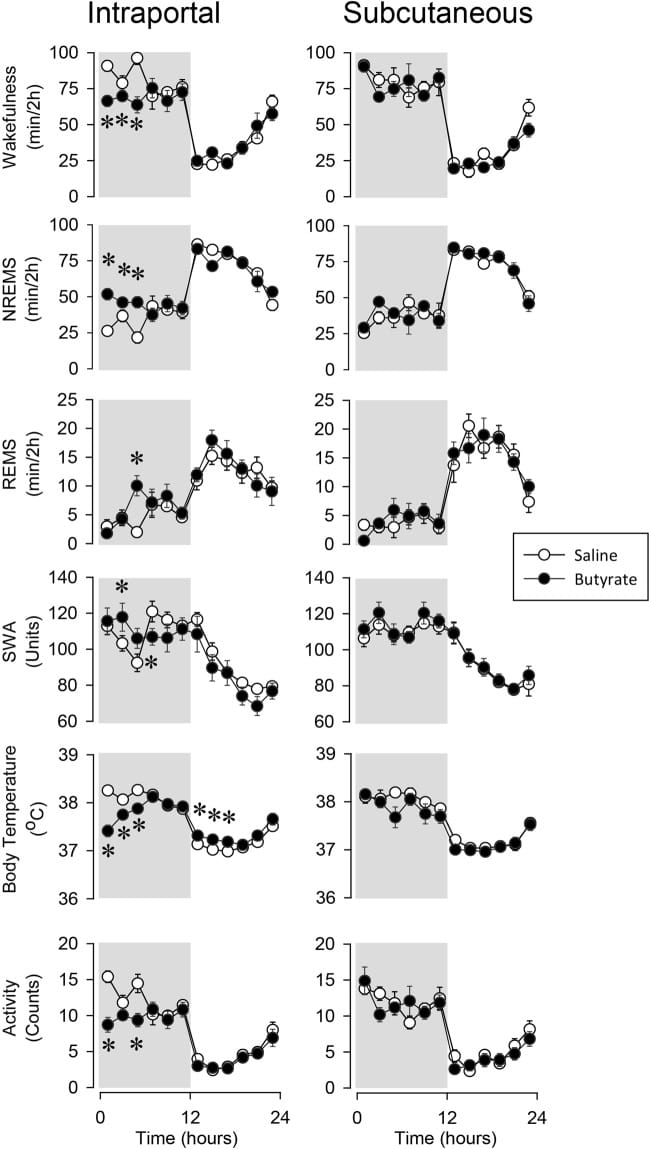
Szentirmai et al. (2019) demonstrated this in mice and rats. Oral tributyrin (a butyrate pro-drug) produced a roughly 50% increase in non-REM sleep duration in mice over four hours. Intraportal butyrate injection in rats increased non-REM sleep by 70% in the first six hours. The finding that distinguishes this from a generalized effect: subcutaneous or intraperitoneal butyrate delivery had no sleep effect. The sleep-promoting action was hepatoportal-specific — meaning butyrate acts through receptors in the portal vein and liver, not through the general bloodstream.
This matters because it identifies a precise mechanism. Butyrate from gut bacteria reaches the portal vein, activates sensory receptors there, and promotes non-REM sleep through that route.
Human data supports the connection. Magzal et al. (2021) measured fecal short-chain fatty acid concentrations in 59 older adults with insomnia and correlated them with actigraphy-measured sleep. Short sleepers (six hours or fewer) had higher fecal SCFA concentrations than normal sleepers. The authors interpreted this as impaired epithelial SCFA absorption — meaning the gut lining was not taking up these compounds efficiently, reducing the amount reaching the portal circulation and the brain. BMI, total sleep time, and sleep onset latency together explained 40.7% of fecal SCFA variance in their regression model.
The SCFA-sleep connection also runs through GABA. Wang et al. (2025) found consistent depletion of butyrate-producing genera (Faecalibacterium and Lachnospira) across insomnia populations in their review of 15 case-control studies. When these bacteria are depleted — as they are in the insomnia microbiome profiles described by Wang et al. — both the hepatoportal sleep-promotion pathway and GABA precursor production are weakened.
Does Sleep Deprivation Harm Gut Bacteria?
Karl et al. (2023) ran a crossover design with 19 healthy men: three nights of two hours of sleep versus three nights of seven to nine hours, with controlled diet throughout. The sleep-restricted condition reduced gut microbiota community richness by approximately 21%. Shannon and Simpson diversity indices did not differ, meaning it was species count that took the hit — the community became less varied while overall evenness held.
The study also measured intestinal permeability using a dual-sugar lactulose/mannitol absorption test and found no change in barrier function after three nights. The authors noted that effects may accumulate with longer restriction — acute severe restriction may degrade the microbiome before measurable barrier changes emerge. This suggests a temporal sequence: dysbiosis first, then barrier compromise.
Karl et al. (2023) measured species-level richness, not which genera were lost. Wang et al. (2025) supplies the genus-level data from insomnia populations: reviewing 15 case-control studies across 1,321 subjects, eight of fifteen studies showed reduced alpha diversity in insomnia versus controls. Eleven showed altered beta diversity. The depleted taxa were consistent across geographically diverse cohorts from Asia, Europe, and North America: Faecalibacterium and Lachnospira — both butyrate-producing, barrier-protective genera — were depleted in insomnia. Blautia, Streptococcus, and Clostridium were enriched. Insomnia severity correlated negatively with Faecalibacterium and Lachnospira abundance.
This creates a self-reinforcing loop. Poor sleep depletes butyrate-producing bacteria. Fewer butyrate-producing bacteria means less short-chain fatty acid reaching the portal circulation, weakening the sleep-promoting hepatoportal pathway. Weakened sleep-promotion leads to worse sleep. And worse sleep depletes the microbiome further. Breaking this loop requires addressing both the sleep disruption and the gut environment.
Can Gut Inflammation Cause Early Morning Waking?
NF-kB activity is not constant — it follows a circadian rhythm with peak activation in the early morning hours. In people with intact gut barriers, this peak is managed without waking. But in people with increased intestinal permeability, LPS-driven NF-kB activation adds to the baseline circadian peak.
Szentirmai et al. (2024) demonstrated that low-dose intraportal LPS — at levels consistent with gut translocation, not a major infection — increased non-REM sleep by approximately 40% and prolonged fever in rats without raising circulating LPS levels in the rest of the body. The sleep and fever responses were blocked by indomethacin (a prostaglandin synthesis inhibitor), demonstrating that prostaglandin E2 mediates the downstream effects. This means even small amounts of LPS from a permeable gut, reaching the portal circulation, can alter sleep architecture through local prostaglandin production.
In the early morning hours, several factors converge. Cortisol begins its natural pre-dawn rise. Histamine — a waking neurotransmitter — increases. NF-kB activity peaks. In someone with gut-derived LPS reaching the portal circulation, prostaglandin-mediated inflammation adds another activating input during this window. The result is a convergence of waking-promoting factors between roughly 3 and 5 a.m. — which is the waking pattern many people with gut permeability issues describe.
The bidirectional nature of the gut-sleep relationship means this early waking itself further damages the microbiome. Each night of fragmented sleep reduces microbial richness, which reduces barrier integrity, which increases LPS translocation the following night. Without addressing the gut environment alongside sleep architecture, the LPS-dysbiosis-waking loop sustains itself.
Gut-driven inflammation might be one of several causes contributing to 3am waking — but it rarely acts alone. Leaky gut and LPS translocation may compound with hormonal causes (cortisol dysregulation, estrogen shifts), autonomic dysregulation, or circadian misalignment, all of which might be active at the same time. Identifying which of these causes might be driving your pattern is a useful next step before targeting any one mechanism.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can Gut Inflammation Cause Insomnia?
The pathway runs from the gut lining to the brain through the three routes described above: bloodstream LPS binding to TLR4 receptors, vagal nerve transmission of inflammatory information, and loss of butyrate-mediated sleep promotion. Each route independently disrupts sleep; when two or three are active at the same time — which is common in chronic gut permeability — insomnia risk compounds.
Does Gut Dysbiosis Cause Insomnia?
The consistent depletion of butyrate-producing genera in insomnia populations is not incidental. These bacteria maintain intestinal barrier integrity and produce the short-chain fatty acids that promote non-REM sleep through the hepatoportal pathway. Their loss creates both increased permeability (more LPS reaching the bloodstream) and reduced sleep-promoting metabolite production — a dual deficit that drives insomnia from two directions.
Does Leaky Gut Cause Brain Fog and Poor Sleep?
LPS-activated microglia produce pro-inflammatory cytokines in the brain that impair both daytime cognitive performance and nighttime sleep consolidation. The two conditions co-occur frequently in people with intestinal permeability because they originate from the same inflammatory pathway.
Can Healing Your Gut Help You Sleep Better?
Because the gut-sleep relationship feeds itself in both directions, entry points for improvement exist on both sides. Restoring butyrate-producing bacteria — through dietary fiber, fermented foods, or targeted prebiotic supplementation — can increase SCFA production and strengthen the intestinal barrier. Improving sleep duration and continuity protects microbiome richness. Neither side needs to be fully resolved before the other begins to respond. Which entry point — gut-focused or sleep-focused — makes sense depends on an individual’s pattern.
Related Reading
- Inflammatory Sleep Disruption — the full cause overview covering all five inflammatory mechanisms
- Why Does Histamine Wake You Up at 3am? — how brain-resident mast cells and the circadian histamine peak drive early-morning waking
- Can Histamine Intolerance Cause Sleep Problems? — how DAO deficiency and mast cell activation compound histamine-driven insomnia
- Why Is Inflammation Worse at Night? — how NF-kB and the circadian clock create a pre-dawn inflammatory vulnerability window
- Does Poor Sleep Cause Brain Inflammation? — the bidirectional loop between neuroinflammation, brain fog, and sleep loss
- How Does the Glymphatic System Work During Sleep? — how the brain’s waste clearance pathway depends on deep sleep
- What Is the Connection Between Chronic Inflammation and Insomnia? — the wired-but-tired cycle of cytokines, sleep loss, and 24-hour hyperarousal
- Why Do Men Sleep Worse After 50?
- What Is Autoimmune Insomnia and Why Does It Resist Standard Sleep Approaches?
- Can Inflammation Cause 3am Wakeups?
- Why Does Inflammation Make You Exhausted But Unable to Sleep?
- Can Chronic Stress Cause Insomnia Through Inflammation?
- Gut Bacteria and Insomnia: Which Microbes Affect Your Sleep (and Which Ones Help)
- Does an Anti-Inflammatory Diet Improve Sleep?
References
1. Karl, J. P., Whitney, C. C., Wilson, M. A., Fagnant, H. S., Radcliffe, P. N., Chakraborty, N., Campbell, R., Hoke, A., Gautam, A., Hammamieh, R., & Smith, T. J. (2023). Severe, short-term sleep restriction reduces gut microbiota community richness but does not alter intestinal permeability in healthy young men. Scientific Reports, 13, 213. https://pubmed.ncbi.nlm.nih.gov/36604516/
2. Magzal, F., Even, C., Haimov, I., Agmon, M., Asraf, K., Shochat, T., & Tamir, S. (2021). Associations between fecal short-chain fatty acids and sleep continuity in older adults with insomnia symptoms. Scientific Reports, 11, 4052. https://pubmed.ncbi.nlm.nih.gov/33603001/
3. Mashaqi, S., Rangan, P., Saleh, A. A., Abraham, I., Gozal, D., Quan, S. F., & Parthasarathy, S. (2023). Biomarkers of gut barrier dysfunction in obstructive sleep apnea: A systematic review and meta-analysis. Sleep Medicine Reviews, 69, 101774. https://pubmed.ncbi.nlm.nih.gov/37028145/
4. Szentirmai, E., Millican, N. S., Massie, A. R., & Kapas, L. (2019). Butyrate, a metabolite of intestinal bacteria, enhances sleep. Scientific Reports, 9, 7035. https://pubmed.ncbi.nlm.nih.gov/31065013/
5. Szentirmai, E., Buckley, K., Massie, A. R., & Kapas, L. (2024). Lipopolysaccharide-mediated effects of the microbiota on sleep and body temperature. Scientific Reports, 14, 27378. https://pubmed.ncbi.nlm.nih.gov/39521828/
6. Wang, Y., Xie, S., Chen, S., Li, C., Chan, Y. L., Chan, N. Y., Wing, Y. K., Chan, F. K. L., Su, Q., & Ng, S. C. (2025). The role of gut microbiota in insomnia: A systematic review of case-control studies. Life, 15(7), 1086. https://pubmed.ncbi.nlm.nih.gov/40724588/
7. Zhang, Y., Xie, B., Chen, X., Zhang, J., & Yuan, S. (2021). A key role of gut microbiota-vagus nerve/spleen axis in sleep deprivation-mediated aggravation of systemic inflammation after LPS administration. Life Sciences, 265, 118736. https://pubmed.ncbi.nlm.nih.gov/33176177/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
