People who have been told they have fatty liver — or who suspect they do — tend to focus on diet, weight, and alcohol. Sleep rarely enters the conversation. Yet people with NAFLD show measurable sleep disruption that correlates with how advanced their liver disease is, not with their weight alone. When fatty liver disrupts sleep, it also disrupts overnight repair processes — growth hormone secretion, glucose clearance, and inflammatory reset — accelerating the progression of both conditions.
This article covers how liver disease disrupts circadian regulation and sleep architecture, how short or poor sleep drives fat accumulation in the liver, and the downstream metabolic burden — including a tripling of type 2 diabetes likelihood when both are present. It does not cover hepatic encephalopathy-related sleep disruption in advanced cirrhosis. For the full metabolic picture, see Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep.
How Does Fatty Liver Disrupt Sleep Architecture?
Bernsmeier et al. (2015) compared 46 adults with NAFLD to 22 healthy controls using wrist actigraphy, the Pittsburgh Sleep Quality Index (PSQI), and the Epworth Sleepiness Scale. The NAFLD group took an average of 26.9 minutes to fall asleep, compared to 9.8 minutes in controls (p=0.0176). Total sleep time was 6.3 hours versus 7.2 hours (p=0.0149). PSQI scores averaged 8.2 in the NAFLD group versus 4.7 in controls (p=0.0074) — well above the cutoff of 5 that marks poor sleep quality.
The more informative finding was what the daytime sleepiness scores correlated with. Epworth Sleepiness Scale scores tracked liver enzyme elevation (ALAT: r=0.44; ASAT: r=0.46), insulin resistance measured by HOMA-IR (r=0.50), and histological fibrosis stage (r=0.40) — all reaching statistical significance. These correlations held across all NAFLD stages. The sleep disruption was not confined to advanced disease.
Meal timing was also pushed toward nighttime hours in the NAFLD group (p=0.001) — suggesting concurrent circadian misalignment of feeding behavior alongside sleep-wake disruption.
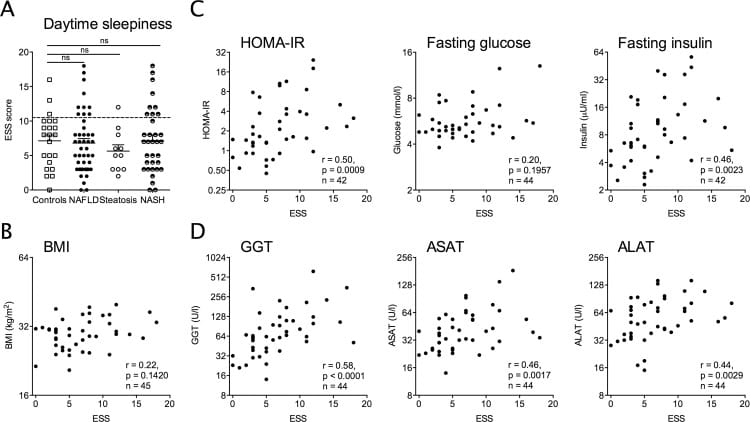
Bu et al. (2024) reviewed the mechanistic pathways from liver to sleep and identified several NAFLD-related routes: altered melatonin and cortisol secretion, low-grade inflammation reaching the brain, gut-liver axis imbalance, and metabolic dysregulation that disrupts circadian entrainment from within the liver outward. The Bernsmeier data showed that sleep parameters correlated with metabolic severity markers — enzyme levels, HOMA-IR, fibrosis stage — rather than with BMI. Fatty liver impairs sleep through liver-related mechanisms, independent of obesity.
Does Poor Sleep Cause Fatty Liver?
Wijarnpreecha et al. (2016) pooled data from six studies in a meta-analysis and found a risk ratio of 1.19 (95% CI 1.04-1.36) for NAFLD among short sleepers, with I²=0% heterogeneity — exceptional consistency across different populations.
Um et al. (2021) extended this with a Korean cohort of 143,306 adults followed over approximately four years, identifying 27,817 incident NAFLD cases. Compared to 7-hour sleepers, those sleeping five hours or less had a hazard ratio of 1.19 (95% CI 1.14-1.23) for incident NAFLD. Six-hour sleepers: HR=1.07 (95% CI 1.04-1.10). The hazard rose monotonically as sleep dropped below seven hours. The association between sleep duration and fibrosis markers differed by sleep quality (interaction p=0.047), suggesting sleep quality modifies the duration-fibrosis relationship.
Sun et al. (2023) provided the strongest evidence for causal direction using a bidirectional genetic instrumental-variable design — a method that uses genetic variants as instruments to test whether the association between two conditions reflects causation rather than shared confounders. Across seven sleep traits and four NAFLD indicators, six causal links emerged: insomnia increased NAFLD odds (OR=2.25), elevated ALT (OR=2.79), and raised liver fat percentage (OR=1.31). Snoring increased liver fat (OR=1.15) and elevated ALT (OR=1.27). Daytime drowsiness increased liver fat (OR=1.14). The primary causal flow runs from sleep traits to NAFLD indicators — not the reverse.
Poor sleep disrupts circadian regulation of hepatic lipid processing, impairs overnight insulin sensitivity, elevates cortisol, and promotes ectopic fat deposition in the liver.
Does Having Both Fatty Liver and Poor Sleep Triple Diabetes Odds?
Zhang et al. (2022) analyzed 365,339 UK Biobank participants without baseline type 2 diabetes over a median 11-year follow-up, during which 8,774 developed type 2 diabetes. The population-attributable fractions were evenly balanced: 33.8% of type 2 diabetes events were attributable to NAFLD, and 33.5% to poor sleep. Optimal sleep (7-8 hours per night) was associated with 20% lower type 2 diabetes incidence. Absence of insomnia: 18% lower incidence. Absence of daytime sleepiness: 31% lower incidence.
The combined effect exceeded what adding the individual estimates together would predict. Participants with both NAFLD and a poor sleep pattern had RR=3.17 for type 2 diabetes. The NAFLD-insomnia interaction reached P=0.003, meaning insomnia does not just add to NAFLD’s metabolic burden — insomnia amplifies it. When NAFLD impairs hepatic insulin sensitivity and poor sleep simultaneously impairs peripheral insulin sensitivity and elevates cortisol, the glucose regulatory load exceeds the threshold that pancreatic beta cells can compensate for.
Addressing insomnia in someone with NAFLD is not just a quality-of-life measure — it is a diabetes prevention strategy.
Many people have more than one cause contributing to their sleep disruption. Fatty liver-driven waking may compound with autonomic, hormonal, inflammatory, or circadian factors. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Why Do People With Advanced Fatty Liver Also Have Sleep Apnea?
Trzepizur et al. (2026) enrolled 97 adults with suspected NAFLD who underwent both in-laboratory polysomnography and liver biopsy — gold-standard measurements for sleep apnea and fibrosis. Advanced fibrosis (F≥3) was present in 40 of the 97 (41%). Moderate-to-severe OSA (AHI≥15 events per hour) was found in 63 (65%). The overlap: 82% of those with advanced fibrosis had moderate-to-severe OSA, compared to 52% of those with F0-2 fibrosis.
After multivariable adjustment, moderate-to-severe OSA remained independently associated with advanced fibrosis (OR=3.48, 95% CI 1.03-11.83). Nocturnal hypoxia metrics tracked fibrosis severity independently of other metabolic factors. OSA was not associated with hepatic steatosis (fat accumulation) or NASH (inflammatory liver disease) — its effect was specific to fibrotic progression. OSA accelerates the scarring process in a liver that already has fat.
Wang et al. (2024) mapped the molecular cascade. Repeated oxygen drops during apnea episodes activate hypoxia-inducible factors (HIFs) — transcription factors that respond to low oxygen. In the liver, HIF activation promotes insulin resistance, impairs fatty acid oxidation (beta-oxidation), upregulates new fat production (lipogenesis), triggers hepatocyte death, and activates inflammatory cascades. The hepatic effect of OSA is independent of age, sex, BMI, and abdominal obesity. CPAP (continuous positive airway pressure) therapy has been investigated for hepatic improvement, but evidence remains inconclusive — the largest randomized trial found no improvement in hepatic steatosis or fibrosis after six months of CPAP.
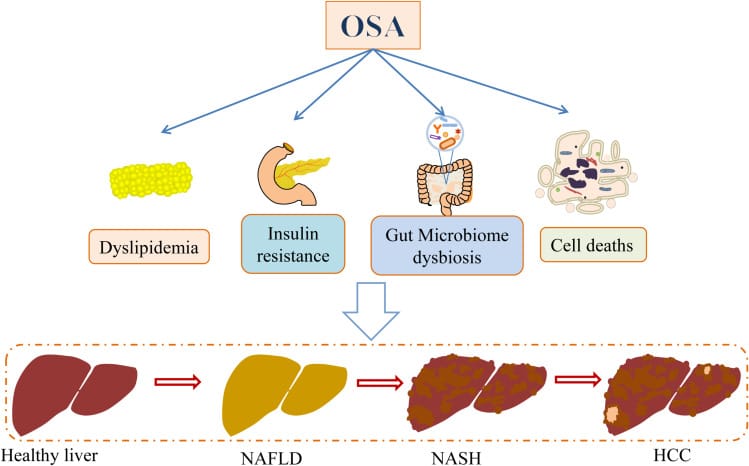
Gachon et al. (2025) placed the OSA-liver relationship within a broader circadian framework. They describe a self-reinforcing loop in MASLD: circadian disruption promotes hepatic steatosis, and liver pathology then generates oxidative stress and pro-inflammatory cytokines that propagate outward to disrupt the suprachiasmatic nucleus (SCN) — the brain’s master clock. OSA sits at the center of this loop as both a product and an amplifier of circadian injury. The OSA-fibrosis relationship is not a one-time hit but a progressive cycle: more liver disease, more sleep apnea, deeper circadian disruption, further liver progression.
People with NAFLD are not routinely evaluated for OSA. Given that 82% of advanced fibrosis cases in the Trzepizur cohort had moderate-to-severe OSA, the overlap is too large to leave to chance.
Frequently Asked Questions
Can Fatty Liver Cause Insomnia?
The mechanisms include altered melatonin secretion, low-grade inflammation originating from the liver, and disrupted cortisol rhythms — all of which can delay sleep onset and fragment sleep architecture (Bu et al., 2024). Bernsmeier et al. (2015) documented sleep onset latency nearly three times longer in the NAFLD group than in controls (26.9 vs 9.8 minutes), and PSQI scores averaging 8.2 — well above the threshold of 5 that marks poor sleep quality.
Whether addressing the underlying liver disease can improve insomnia remains an open question. Busebee et al. (2024) reviewed sleep disturbances across chronic liver diseases, though the evidence for whether addressing liver disease restores normal sleep remains limited.
Does Fatty Liver Affect Sleep Quality Specifically, or Just Sleep Duration?
Sleep onset latency was also nearly three times longer in the NAFLD group (26.9 vs 9.8 minutes), per Bernsmeier et al. (2015). The combination of harder-to-initiate sleep, less total sleep, and lower quality sleep compounds the overnight metabolic disruption compared to any single parameter alone.
Um et al. (2021) showed at scale that short sleep duration predicted incident NAFLD after adjusting for metabolic confounders, with a dose-response gradient below seven hours. The dose-response gradient for duration, and the interaction between duration and quality in predicting fibrosis markers (p=0.047), suggest that sleep features may contribute to fatty liver development through interacting pathways.
Does Addressing Sleep Apnea Help Fatty Liver?
The mechanism works in reverse: when intermittent hypoxia is eliminated with CPAP, HIF activation decreases, lipogenesis normalizes, and hepatic inflammatory load reduces. Wang et al. (2024) reviewed the evidence for CPAP improving liver histology and found the mechanistic rationale well-established, though the body of outcome data remains inconclusive.
Trzepizur et al. (2026) found that the OSA-fibrosis association was specific to fibrotic progression rather than to fat accumulation or inflammation per se. This specificity matters: it suggests that managing OSA may be the highest-impact step for slowing or preventing fibrosis advancement, which is the stage of NAFLD progression that is closely linked to long-term liver outcomes.
Is the Fatty Liver-Sleep Connection Just Because Both Are Caused by Obesity?
Obesity amplifies both fatty liver and poor sleep. But the fatty liver-sleep relationship persists in studies that control for it. The genetic evidence from Sun et al. (2023) provides the strongest argument: insomnia genetically predisposes to NAFLD (OR=2.25) through a pathway independent of shared lifestyle factors. Because the genetic instruments are assigned at conception, they are not confounded by later-life behaviors, diet, or body composition.
The Trzepizur et al. (2026) OSA-fibrosis association held after multivariable adjustment for sex, age, diabetes, and obesity (OR=3.48). Bernsmeier et al. (2015) showed that daytime sleepiness correlated with ALAT, ASAT, HOMA-IR, and fibrosis stage — liver-related markers — not with body weight. The consistency across genetic, biopsy-verified, and actigraphy-based studies makes it difficult to attribute the fatty liver-sleep relationship to obesity alone.
Related Reading
- Metabolic Sleep Disruption: The Full Picture
- Can a Blood Sugar Drop Wake You Up at 3am?
- Why Do You Wake Up at 3am Hungry?
- Why Does Cortisol Spike at 3am and Wake You Up?
- Can a Calorie Deficit Cause Insomnia?
- Adrenal Fatigue and 3am Waking
- Insulin Resistance and Insomnia
- Prediabetes and Sleep Problems
- CGM, Nocturnal Glucose, and Sleep
- Intermittent Fasting, Blood Sugar, and Sleep
- Keto and 3am Waking
- Visceral Fat, Growth Hormone, and Sleep
- Dawn Phenomenon Without Diabetes
- Gut Dysbiosis, Blood Sugar, and Sleep
- Late-Night Eating, Blood Sugar, and Sleep
References
1. Bernsmeier, C., Weisskopf, D. M., Pflueger, M. O., Mosimann, J., Campana, B., Terracciano, L., Beglinger, C., Heim, M. H., & Cajochen, C. (2015). Sleep Disruption and Daytime Sleepiness Correlating with Disease Severity and Insulin Resistance in Non-Alcoholic Fatty Liver Disease: A Comparison with Healthy Controls. PloS one, 10(11), e0143293. https://pubmed.ncbi.nlm.nih.gov/26576055/
2. Wijarnpreecha, K., Thongprayoon, C., Panjawatanan, P., & Ungprasert, P. (2016). Short sleep duration and risk of nonalcoholic fatty liver disease: A systematic review and meta-analysis. Journal of gastroenterology and hepatology, 31(11), 1802-1807. https://pubmed.ncbi.nlm.nih.gov/27029776/
3. Um, Y. J., Chang, Y., Jung, H. S., Cho, I. Y., Shin, J. H., Shin, H., Wild, S. H., Byrne, C. D., & Ryu, S. (2021). Sleep Duration, Sleep Quality, and the Development of Nonalcoholic Fatty Liver Disease: A Cohort Study. Clinical and translational gastroenterology, 12(10), e00417. https://pubmed.ncbi.nlm.nih.gov/34665792/
4. Zhang, H., Wang, Y., Chen, C., Wang, B., Chen, J., Tan, X., Xia, F., Zhang, J., Lu, Y., & Wang, N. (2022). Non-alcoholic fatty liver disease, sleep behaviors, and incident type 2 diabetes. Journal of gastroenterology and hepatology, 37(8), 1633-1640. https://pubmed.ncbi.nlm.nih.gov/35499342/
5. Sun, Z., Ji, J., Zuo, L., Hu, Y., Wang, K., Xu, T., Wang, Q., & Cheng, F. (2023). Causal relationship between nonalcoholic fatty liver disease and different sleep traits: a bidirectional Mendelian randomized study. Frontiers in endocrinology, 14, 1159258. https://pubmed.ncbi.nlm.nih.gov/37334291/
6. Bu, L. F., Xiong, C. Y., Zhong, J. Y., Xiong, Y., Li, D. M., Hong, F. F., & Yang, S. L. (2024). Non-alcoholic fatty liver disease and sleep disorders. World journal of hepatology, 16(3), 304-315. https://pubmed.ncbi.nlm.nih.gov/38577533/
7. Busebee, B., Watt, K. D., Dupuy-McCauley, K., & DuBrock, H. (2024). Sleep disturbances in chronic liver disease. Liver transplantation, 30(10), 1058-1071. https://pubmed.ncbi.nlm.nih.gov/38535627/
8. Wang, L., Liu, H., Zhou, L., Zheng, P., Li, H., Zhang, H., & Liu, W. (2024). Association of Obstructive Sleep Apnea with Nonalcoholic Fatty Liver Disease: Evidence, Mechanism, and Treatment. Nature and science of sleep, 16, 917-933. https://pubmed.ncbi.nlm.nih.gov/39006248/
9. Gachon, F., Bugianesi, E., Castelnuovo, G., Oster, H., Pendergast, J. S., & Montagnese, S. (2025). Potential bidirectional communication between the liver and the central circadian clock in MASLD. npj metabolic health and disease, 3(1), 15. https://pubmed.ncbi.nlm.nih.gov/40225783/
10. Trzepizur, W., Boursier, J., Poilane, J., Bureau, C., Sedkaoui, K., Pontier, S., Costentin, C., Pépin, J. L., Schnee, M., Bizieux, A., Guillaume, M., Hamel, J. F., & Gagnadoux, F. (2026). Association between obstructive sleep apnea and liver fibrosis in patients with suspected nonalcoholic fatty liver disease. Sleep, 49(2). https://pubmed.ncbi.nlm.nih.gov/40884485/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 10 references cited
