Consumer CGMs — Levels, Veri, Lingo, Dexcom Stelo — have made continuous glucose data available to people without diabetes for the first time. People are wearing them, seeing nocturnal dips and spikes, and searching for what those patterns mean for sleep.
CGM data can separate blood-sugar-driven waking from cortisol-driven waking — two causes that look identical from the surface but require different responses. Having a continuous overnight glucose trace turns a vague 3am wakeup into a data point with a visible metabolic event preceding it.
This article covers what CGMs detect overnight, what common nocturnal glucose patterns mean, and the limits of what a CGM can and cannot tell you about sleep disruption. For the full metabolic cause overview, see Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep.
Metabolic disruption is one of several causes of sleep fragmentation — autonomic, inflammatory, hormonal, and circadian factors can produce similar patterns, and more than one may be active at once.
What Does a Continuous Glucose Monitor Record While You Sleep?
A CGM sensor sits just beneath the skin and measures glucose in interstitial fluid rather than blood directly. This interstitial reading lags behind blood glucose by roughly 5 to 15 minutes, which matters for interpreting sharp spikes and drops — the CGM trace may show a blunted version of what happened in the bloodstream.
During a normal night, the CGM trace follows a predictable pattern. Glucose declines gradually during the first half of the night as the body transitions from burning glucose to burning fat. Between roughly 3am and 5am, cortisol and growth hormone begin rising in preparation for waking, and the liver releases stored glucose into the bloodstream. This pre-dawn rise — the dawn phenomenon — is a normal physiological event.
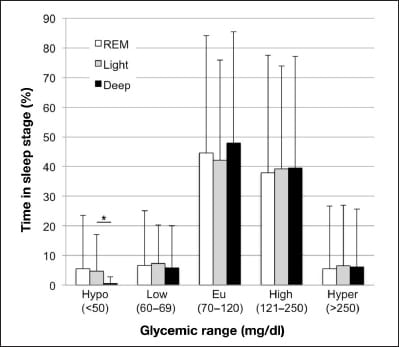
The sleep stage you are in shapes the glucose trace. Feupe et al. (2013) simultaneously recorded CGM and sleep-stage data across 39 subject-nights and found that glucose remained in the normal range 48% of the time during deep sleep compared to 42.1% during light sleep. Hypoglycemia occurred less often during deep sleep than during light sleep (p = .02). Numerically, REM sleep showed the highest hypoglycemia burden at 5.5% of the time versus 0.5% during deep sleep, though this difference did not reach statistical significance.
A consistent problem with CGM overnight analysis is the time window used. Trawley et al. (2024) compared glucose metrics from a fixed midnight-to-6am window against metrics from objectively measured sleep periods across 356 nights. The fixed window missed a median of 57 minutes of actual sleep per night. Five hypoglycemic episodes fell outside the midnight-to-6am window and would have been missed. And 96% of participants showed discrepancies of 10 percentage points or more between the two methods.
The practical takeaway: a CGM produces overnight glucose data, but that data is only as useful as the sleep timing it is measured against. Without knowing when you fell asleep and when you woke up, a CGM overnight summary may be measuring the wrong window.
What Nocturnal Glucose Patterns Does a Continuous Glucose Monitor Reveal?
Pattern 1: The nocturnal dip. Glucose drops below approximately 70 mg/dL (3.9 mmol/L), usually in the first half of the night or around 2 to 4am. The frequency of these events has been underestimated for decades. Kulzer et al. (2024) document that CGM revealed a prior underestimation of nocturnal hypoglycemic events compared to fingerstick monitoring — a burden that was invisible because people were asleep when it happened.
Gardner et al. (2023) tracked 27 adults wearing CGM and providing daily sleep quality ratings. Nights with glucose below 3.9 mmol/L produced worse sleep quality scores. Both the frequency of dips and the total duration below threshold independently predicted worse sleep. Frequent mild dips were more disruptive than rare but deeper drops — the recurrence of small events mattered more than the severity of a single episode.
Pattern 2: The dawn phenomenon rise. Blood sugar climbs during the last 1 to 2 hours of sleep as cortisol, growth hormone, and catecholamines direct the liver to release stored glucose. In someone with insulin resistance, the insulin response falls short and morning glucose ends up higher than bedtime glucose. This is distinct from the Somogyi effect, which is a rebound rise following a preceding nocturnal dip. CGM can differentiate the two because it captures the full trajectory — a dawn rise without a preceding dip is the dawn phenomenon; a rise that follows a low is Somogyi.
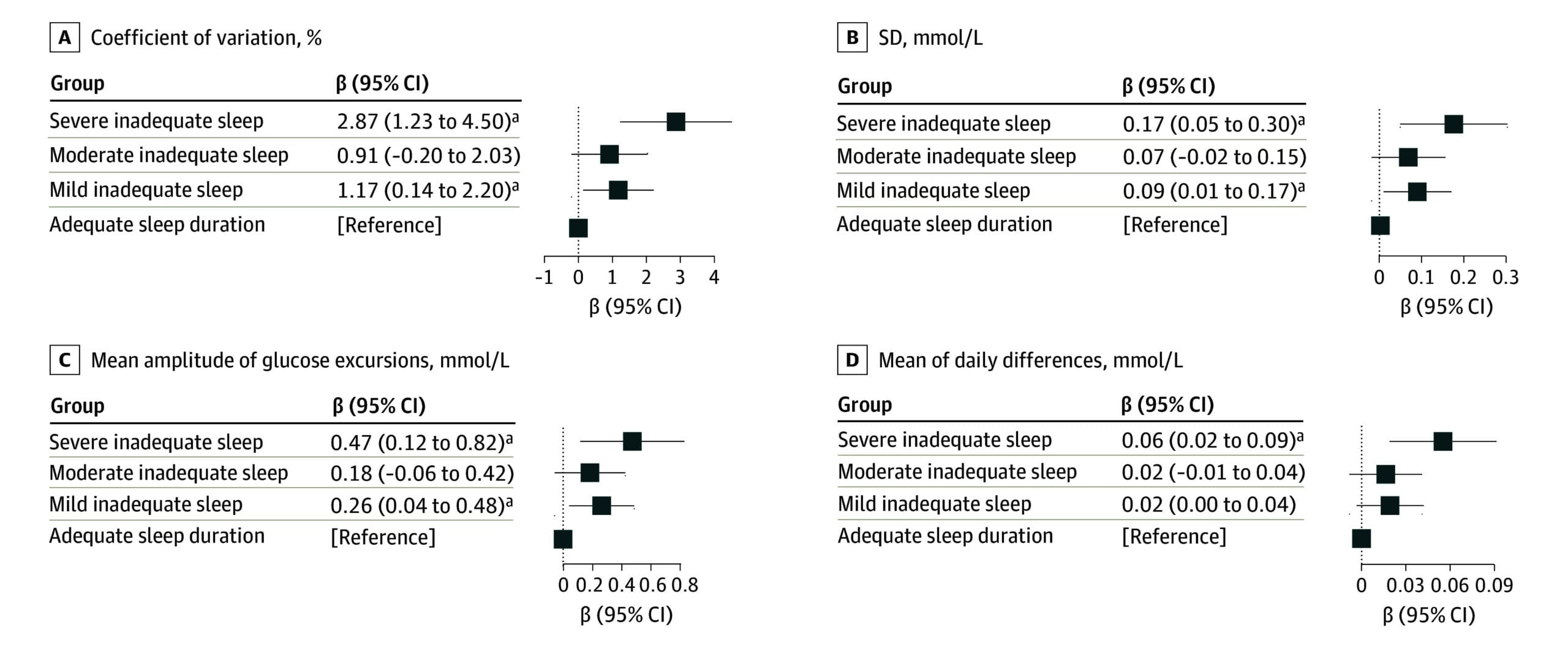
Pattern 3: High glycemic variability. Large glucose swings throughout the night — not a single dip or a single rise, but repeated oscillations. Shen et al. (2025) studied 1,156 adults and found that persistent inadequate sleep increased CGM-derived coefficient of variation (a measure of glucose swing magnitude) by 2.87%. Late sleep onset independently added another 1.18% increase in glycemic variability. These findings suggest that poor sleep may destabilize glucose regulation. Whether unstable glucose in turn fragments sleep — creating a self-reinforcing cycle — is plausible but has not been demonstrated in the same study.
Can a Nocturnal Glucose Drop on Your Continuous Glucose Monitor Explain a 3am Wakeup?
The body’s glucose-recovery response works in stages. As glucose drops, glucagon is released first, directing the liver to release stored glucose. If the drop continues, epinephrine enters the bloodstream, raising heart rate and activating the brain’s arousal centers. Cortisol and growth hormone follow to sustain glucose recovery over the next one to two hours. Epinephrine and cortisol are also arousal hormones — the same hormonal response that raises your glucose level is what pulls you out of sleep.
But the awakening response is not reliable.
Jennum et al. (2015) studied 26 adults with type 2 diabetes across two polysomnography nights: one with normal glucose and one with experimentally induced hypoglycemia held at 2.7 to 2.8 mmol/L (approximately 49 to 50 mg/dL). During hours 4 through 8 of sleep, awakening rates decreased by 27% on the hypoglycemic night compared to the normal-glucose night. Counterregulatory hormones — adrenaline, growth hormone, and cortisol — rose during hypoglycemia, showing the body detected the low glucose. But the hormonal response did not reach the threshold required to trigger waking. The hormones rose. The person stayed asleep.
This finding matters for CGM interpretation. A glucose dip that was not followed by waking may indicate a more concerning pattern than one that did wake you — the dip you slept through means your body mounted a stress hormone response that was insufficient to produce waking, and glucose may have fallen further before recovery began.
The CGM captures the glucose event but not the hormonal response. Two identical-looking dips may produce different arousal outcomes depending on the individual’s counterregulatory sensitivity. The CGM shows that glucose dropped to 65 mg/dL at 3am and recovered by 4am. It cannot tell you whether that recovery was driven by a mild glucagon response or a full adrenaline surge that woke you at 3:15am.
Sleep apnea complicates interpretation further. Gouveri et al. (2025) reviewed CGM evidence across populations with and without diabetes and found that obstructive sleep apnea severity is associated with increased glycemic variability regardless of diabetes status. Intermittent oxygen drops during apnea events trigger sympathetic activation and glucose counterregulatory responses. This means some CGM-detected nocturnal glucose instability reflects breathing events rather than dietary or metabolic causes. A person seeing nighttime glucose swings on their CGM and optimizing diet with no improvement may be looking at sleep-disordered breathing as the source.
What Can a Continuous Glucose Monitor Not Tell You About Sleep Disruption?
The first limitation is the measurement itself. CGM reads interstitial glucose, which lags behind blood glucose by 5 to 15 minutes. A glucose nadir that the body registered at 3:00am may appear on the CGM at 3:10am — which matters when trying to match a glucose event to the moment you woke up.
The second limitation is sleep stage. Feupe et al. (2013) demonstrated that sleep stage shapes glucose behavior — numerically, REM showed the highest hypoglycemia burden (5.5% of time below threshold versus 0.5% in deep sleep), though this difference was not statistically significant. But a CGM cannot identify which sleep stage you were in when a dip occurred. A glucose drop during REM has different physiological implications than the same drop during deep sleep, and the CGM trace looks the same for both.
The third limitation is sleep timing. Trawley et al. (2024) showed that without objective sleep timing data, even the glucose metrics are inaccurate — 96% of participants were mischaracterized by standard overnight windows. Any overnight summary generated by a CGM app uses assumed or user-entered sleep times, not measured ones.
The fourth limitation is competing causes. Gouveri et al. (2025) demonstrated that sleep apnea produces CGM-detectable glucose instability in non-diabetic individuals. Intermittent oxygen deprivation during apnea events triggers the same counterregulatory hormones that a glucose dip would, producing similar CGM-visible swings from a different root cause.
CGM data is useful when paired with a sleep tracker or structured sleep log to align glucose events with actual sleep timing and waking episodes. Without that pairing, the CGM shows what your glucose did overnight — but not what it means for your sleep.
A CGM trace showing a 3am glucose dip may be one piece of a multi-cause pattern. Blood sugar instability, cortisol timing, sleep architecture, and breathing disruption can overlap — and the combination determines what will help.
Find out which causes might be driving your 3am wakeups –>
Frequently Asked Questions
What Should Your Blood Sugar Be When You Wake Up?
The CGM value at 6am is not the same as a laboratory fasting glucose measurement. The CGM reading at waking reflects the tail end of the dawn phenomenon — the pre-dawn cortisol and growth hormone surge that releases glucose from the liver. This means the CGM wake-time value is typically the highest glucose reading of the night, not a metabolic baseline.
When you fell asleep also matters. Shen et al. (2025) found that late sleep onset independently elevated glycemic variability, meaning morning glucose is partly determined by what time sleep began. A person falling asleep at 1am versus 10:30pm may show different wake-time glucose values with the same diet and metabolic health.
Why Is Morning Blood Sugar Higher Than Your Bedtime Reading?
CGM makes this pattern visible to non-diabetic wearers for the first time. Many people seeing a morning rise assume something went wrong overnight. Typically, the dawn rise is normal. The concern arises when the rise is exaggerated (glucose climbing above 120 to 130 mg/dL before waking) or when a preceding dip triggers the rebound.
CGM differentiates these scenarios because it captures the full overnight trajectory. A steady decline followed by a gradual rise starting around 4 to 5am is the dawn phenomenon. A drop into the 60s or low 70s followed by a sharp rebound to 110 or higher is the Somogyi effect — involving counterregulatory hormone activation and more likely to fragment sleep (Kulzer et al., 2024).
Does Sleep Apnea Affect What a Continuous Glucose Monitor Shows Overnight?
This is an underrecognized differentiator for consumer CGM users. Someone seeing nighttime glucose swings may adjust meal timing and cut evening carbohydrates with no improvement — because the source may be sleep-disordered breathing, not diet.
CPAP use may improve blood glucose levels and glycemic variability in individuals with both type 2 diabetes and obstructive sleep apnea, though the evidence remains inconsistent across studies and effect sizes vary (Gouveri et al., 2025). When CGM-detected nocturnal glucose instability persists despite dietary changes, consider a sleep apnea evaluation.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep
- Can a Blood Sugar Drop Wake You Up at 3am?
- Why Do You Wake Up Hungry at 3am?
- Cortisol Spike at 3am: What It Means
- Can a Calorie Deficit Cause Insomnia?
- Adrenal Fatigue and 3am Waking: What Is Actually Happening?
- Insulin Resistance and Insomnia
- Prediabetes and Sleep Problems
- Why Does Intermittent Fasting Disrupt Your Sleep?
- Keto and 3am Waking
- Visceral Fat, Growth Hormone, and Sleep
- Dawn Phenomenon in Non-Diabetic People
- Fatty Liver and Sleep Problems
- Gut Dysbiosis, Blood Sugar, and Sleep
- Late-Night Eating, Blood Sugar, and Sleep
References
Feupe, S. F., Frias, P. F., Mednick, S. C., McDevitt, E. A., & Heintzman, N. D. (2013). Nocturnal continuous glucose and sleep stage data in adults with type 1 diabetes in real-world conditions. Journal of Diabetes Science and Technology, 7(5), 1337-1345. https://pubmed.ncbi.nlm.nih.gov/24124962/
Gardner, D., Tan, H. C., Lim, G. H., Zin Oo, M., Xin, X., & Rama Chandran, S. (2023). Relationship between CGM-derived nocturnal hypoglycemia and subjective sleep quality in people with type 1 diabetes. Scientific Reports, 13(1), 20887. https://pubmed.ncbi.nlm.nih.gov/38017001/
Gouveri, E., Drakopanagiotakis, F., Panou, T., Papazoglou, D., Steiropoulos, P., & Papanas, N. (2025). Continuous glucose monitoring among people with and without diabetes mellitus and sleep apnoea. Diabetes Therapy, 16(6), 1533-1555. https://pubmed.ncbi.nlm.nih.gov/40465146/
Jennum, P., Stender-Petersen, K., Rabol, R., Jorgensen, N. R., Chu, P. L., & Madsbad, S. (2015). The impact of nocturnal hypoglycemia on sleep in subjects with type 2 diabetes. Diabetes Care, 38(11), 2151-2157. https://pubmed.ncbi.nlm.nih.gov/26407587/
Kulzer, B., Freckmann, G., Ziegler, R., Schnell, O., Glatzer, T., & Heinemann, L. (2024). Nocturnal hypoglycemia in the era of continuous glucose monitoring. Journal of Diabetes Science and Technology, 18(5), 1052-1060. https://pubmed.ncbi.nlm.nih.gov/39158988/
Shen, L., Li, B. Y., Gou, W., Liang, X., Zhong, H., Xiao, C., Shi, R., Miao, Z., Yan, Y., Fu, Y., Chen, Y. M., & Zheng, J. S. (2025). Trajectories of sleep duration, sleep onset timing, and continuous glucose monitoring in adults. JAMA Network Open, 8(3), e250114. https://pubmed.ncbi.nlm.nih.gov/40042843/
Trawley, S., Kubilay, E., Colman, P. G., Lee, M. H., O’Neal, D. N., Sundararajan, V., Vogrin, S., & McAuley, S. A. (2024). What difference does sleep make? Continuous glucose monitoring metrics during fixed-overnight time versus sleep periods among older adults with type 1 diabetes. Journal of Sleep Research, 33(4), e14106. https://pubmed.ncbi.nlm.nih.gov/38050705/
Written by Kat Fu, M.S., M.S. — Last reviewed: May 2026 — 7 references cited
