Late-night eating is one of the more common behavioral patterns that disrupts metabolic sleep. The mechanism is not “digestion keeping you awake.” It is a documented hormonal sequence: eating late coincides with peak melatonin secretion, which suppresses the insulin response, keeps blood glucose elevated into sleep, and sets up a reactive crash in the early morning hours that the body resolves through cortisol — a hormone that ends deep sleep.
Repeated blood sugar crashes during sleep accelerate insulin resistance over time. Insulin resistance is associated with a higher likelihood of type 2 diabetes, cardiovascular disease, and cognitive decline. The research on meal timing and glucose tolerance is now robust enough that it has implications beyond one night of disrupted sleep — when the pattern repeats nightly, it becomes a chronic metabolic stress. A common genetic variant in the melatonin receptor gene (MTNR1B) amplifies this effect in a large portion of the population, making some individuals more vulnerable to late-eating-driven glucose disruption than others.
This article covers the specific insulin-glucose mechanism connecting late eating to nocturnal blood sugar crashes and 3am waking. It does not cover the circadian clock-resetting effects of meal timing. For the full overview of how metabolic impairment fragments sleep, see Metabolic Sleep Disruption. For the blood sugar crash mechanism itself — independent of meal timing — see Can a Blood Sugar Drop Wake You Up at 3am?.
Why Does Eating Late Raise Blood Sugar During Sleep?
The body’s ability to process carbohydrates is not constant across the day. Insulin sensitivity and pancreatic beta-cell responsiveness peak during morning hours and steadily drop through the afternoon and evening. This is an endogenous circadian rhythm — it occurs independent of physical activity levels or how much food has already been consumed that day.
Gu et al. (2020) demonstrated this in a randomized crossover trial with 20 healthy volunteers (mean age 26, mean BMI 23.2). Each participant’s dinner was served at 6 PM on one visit and at 10 PM on another (with a smaller snack at the alternate time), with sleep held constant between 11 PM and 7 AM both times. Late dinner moved the postprandial period directly into the sleep window. The result was elevated nocturnal glucose during sleep and delayed triglyceride clearance — fat metabolism was also suppressed, as the body diverted resources to carbohydrate processing that it could not efficiently complete at that hour (Gu et al., 2020).
Free fatty acid oxidation — the body’s normal fat-burning function during sleep — was reduced with late eating. The overnight metabolic program, which in a fasted state prioritizes fat oxidation, was displaced by carbohydrate metabolism the body was metabolically unequipped to handle quickly. Plasma cortisol was also elevated after the late dinner relative to the early dinner condition, an early indicator that stress-response pathways are engaged by nighttime eating (Gu et al., 2020).
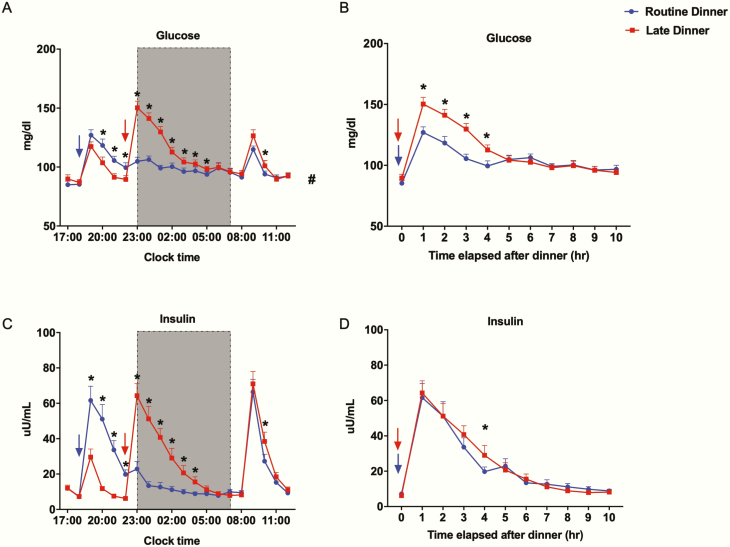
A separate controlled laboratory study by Chellappa, Qian et al. (2021) reinforced this finding. In a 14-day simulated night-work experiment, participants assigned to nighttime meals (n=10) showed a 19.4% increase in their 3-hour postprandial glucose profile compared to baseline (p=0.002). Participants assigned to daytime-only meals (n=9) showed no measurable glucose change — even though both groups had the same disrupted sleep schedule (Chellappa et al., 2021). Eating at the wrong circadian time was sufficient to impair glucose tolerance, independent of sleep deprivation.
The carbohydrate content of a late meal still matters. Timing is the primary variable: the same carbohydrates eaten four hours earlier produce less nocturnal glucose elevation. Timing functions as a variable independent of dietary composition.
How Does Melatonin Block Insulin and Keep Glucose Elevated?
Melatonin is released by the pineal gland beginning one to two hours before habitual sleep time. Its primary role is signaling darkness and initiating sleep physiology. It also has a well-established role at the pancreas: binding to MT2 receptors on beta cells and suppressing insulin release. This dual function means that a meal eaten during peak melatonin encounters a pancreas that is hormonally instructed to release less insulin. (For more on the relationship between melatonin and glucose regulation, see Melatonin and Blood Sugar.)
Garaulet et al. (2022) tested this directly in the largest human trial to date on the melatonin-meal timing interaction. In a randomized crossover design with 845 participants, each person underwent two oral glucose tolerance tests — one simulating an early dinner (4 hours before sleep) and one simulating a late dinner (1 hour before sleep). Late dinner timing produced serum melatonin levels 3.5-fold higher than the early dinner condition. That elevated melatonin was associated with 6.7% lower insulin AUC and 8.3% higher glucose AUC across the full sample — the body released less insulin and processed glucose more slowly during the late eating condition (Garaulet et al., 2022).
The effect was amplified in individuals carrying the MTNR1B G allele, a common genetic polymorphism in the melatonin receptor gene. MTNR1B G-allele carriers showed a stronger genotype-by-timing interaction for glucose AUC (p=0.009), corrected insulin response (p=0.022), and disposition index (p=0.018). These individuals have greater beta-cell inhibition from melatonin — their pancreas responds more strongly to melatonin’s instruction to reduce insulin output (Garaulet et al., 2022).

Lopez-Minguez, Bandin et al. (2018) established the genotype specificity in an earlier crossover study with 40 participants — 20 MTNR1B GG carriers and 20 CC non-carriers. Late dinner impaired glucose tolerance in GG carriers (glucose AUC 292.2 vs 270.9 mmol/L x h, p=0.006) but not in CC non-carriers (277.3 vs 268.2 mmol/L x h, p=0.122). The genotype-timing interaction reached p=0.045, showing that genetic background determines how severely melatonin interferes with insulin secretion during late eating (Lopez-Minguez et al., 2018).
Diaz-Rizzolo et al. (2024) added further support in a prediabetes population (n=26): late eaters showed higher OGTT glucose at 30 minutes (p=0.028) and 60 minutes (p=0.036) after adjustment for body weight, fat mass, total energy intake, and diet composition. Meal timing was an independent driver of glucose intolerance, not a proxy for poor diet quality (Diaz-Rizzolo et al., 2024).
The mechanism in sequence: late meal arrives, melatonin is already elevated, melatonin binds MT2 receptors on beta cells, insulin secretion is suppressed, postprandial glucose stays elevated longer into sleep than it would after an early meal.
What Is the Reactive Blood Sugar Crash and How Does It Wake You Up?
After melatonin suppresses the initial insulin response and glucose stays elevated longer than normal, the pancreas does not cleanly halt insulin release when glucose is still circulating. Insulin secretion eventually catches up or overshoots — producing a delayed reactive insulin peak that drives glucose below the normal fasting range. This is a reactive blood sugar crash.
When blood glucose falls below the brain’s threshold, the hypothalamus activates the counterregulatory response: epinephrine and cortisol are released to drive gluconeogenesis and restore glucose levels. Both are activating hormones. Cortisol is the body’s primary arousal hormone — it is the same hormone that rises sharply at natural morning wake time. Epinephrine increases heart rate and triggers the fight-or-flight state. The combination ends deep sleep.
Corrado et al. (2025) demonstrated this relationship in a cohort of 189 adults with type 1 diabetes using continuous glucose monitors. Severe hypoglycemia (time below 54 mg/dL) was the main predictor of lower sleep quality — blood sugar crashes produce waking rather than deeper sleep because the brain’s counterregulatory response overrides sleep architecture. Dinner timing was the main predictor of HbA1c, and severe hypoglycemia was the main predictor of lower sleep quality — suggesting that late eating and blood sugar instability contribute to sleep disruption through related but distinct pathways. Even in participants using automated insulin delivery, the timing effect persisted (Corrado et al., 2025).
Reutrakul and Van Cauter (2014) reviewed experimental evidence showing that sleep insufficiency substantially reduces insulin sensitivity, establishing a compounding cycle: late eating disrupts sleep, and disrupted sleep further impairs glucose regulation the following night. Each night of disrupted sleep can make the next night’s glucose response less stable.
The 3am specificity is not coincidental. Cortisol follows its own circadian rhythm, with a trough around 2-3am and its first rise beginning around 3-4am. When a reactive blood sugar crash from a late meal coincides with this early cortisol rise, the hormonal environment is already transitioning toward waking. The crash accelerates and amplifies the transition out of deep sleep.
This is not insomnia in the conventional sense. There is no racing mind or anxiety at onset. The hallmark is waking with sudden alertness, sometimes with a fast heart rate or a feeling of hunger. The body’s counterregulatory response has restored glucose, but it has also ended sleep in the process.
What Does the Research Show About Eating Earlier and Sleep Quality?
Vahlhaus et al. (2025) quantified the relationship between meal timing and insulin sensitivity using a twin study design (n=92). Later circadian caloric midpoint — the clock time by which 50% of daily calories are consumed — was independently associated with insulin sensitivity (ISI Stumvoll: beta=0.304, p=5.9×10^-4) and insulin resistance (HOMA-IR: beta=-0.258, p=0.011) after adjustment for sex, age, energy intake, and sleep duration. The effect was measurable at the population level (Vahlhaus et al., 2025).
Vahlhaus et al. also found that meal timing preferences showed high heritability in the twin design. This is not purely a behavioral habit — it reflects genetic influence on eating chronotype. Some individuals have a stronger biological pull toward late eating, which has implications for how difficult late-eating correction is for different people (Vahlhaus et al., 2025).
Rovira-Llopis et al. (2024) pooled data from 18 studies (n=1,169) in a meta-analysis of time-restricted eating and glycemic outcomes. Time-restricted eating reduced HbA1c (Hedges’ g=-0.27, p=0.01) and fasting insulin (Hedges’ g=-0.40, p=0.01) compared to controls. Early eating windows uniquely reduced fasting glucose (g=-0.38, p<0.01), while late eating windows negated the glycemic benefit — the window timing mattered as much as having a window at all (Rovira-Llopis et al., 2024).
Maqsood et al. (2026) reviewed the accumulated experimental evidence on circadian-aligned time-restricted eating and glucose regulation, finding that TRE enhances insulin sensitivity and glucose metabolism through alignment of food intake with circadian metabolic rhythms. These mechanisms are consistent with the melatonin-insulin pathway described above — when meals land during peak beta-cell responsiveness rather than during peak melatonin, insulin secretion matches the glucose load (Maqsood et al., 2026).
On the practical question of how many hours before bed to stop eating: the Garaulet et al. (2022) experimental conditions used 4 hours before sleep as the “early” condition, producing 3.5-fold lower melatonin overlap than eating 1 hour before sleep. This is not a universal prescription, but it establishes that a 3-4 hour gap between the last meal and sleep is sufficient to reduce the overlap between postprandial glucose processing and peak melatonin.
Late eating is not categorically harmful for every person. The metabolic cost of late eating is measurable, documented, and highest in MTNR1B risk allele carriers. Individual glucose response monitoring via continuous glucose monitoring (CGM) provides the best direct evidence of how late eating affects any specific person.
Many people have more than one cause contributing to their sleep disruption. Meal-timing-driven blood sugar crashes may compound with autonomic, hormonal, inflammatory, or circadian factors. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Which Foods Are Likely to Cause Nighttime Waking?
High-glycemic carbohydrates — refined grains, sugary foods, sweetened beverages — produce a rapid glucose spike that collides with melatonin-suppressed insulin secretion when eaten late. The larger the glucose load, the greater the potential for a delayed insulin overshoot and reactive crash during the early morning hours.
High-fat meals have a different but overlapping effect. Gu et al. (2020) showed that late eating suppressed free fatty acid oxidation overnight and delayed triglyceride clearance, meaning the body’s normal fat-burning function during sleep was displaced by ongoing digestive metabolism. Cortisol was also elevated after the late dinner, engaging stress-response pathways during what should be a restorative period.
Alcohol raises blood sugar initially, then causes a rebound drop in the second half of the night through a separate metabolic pathway. For the general blood sugar crash mechanism independent of meal timing, see Can a Blood Sugar Drop Wake You Up at 3am?.
Does Sugar Cause Nighttime Waking?
The effect is pronounced in individuals carrying the MTNR1B genetic variant. Garaulet et al. (2022) showed that MTNR1B G-allele carriers had a stronger glucose-impairing response to late eating, and Lopez-Minguez, Bandin et al. (2018) found that late dinner impaired glucose tolerance in GG carriers (p=0.006) while CC non-carriers showed no statistically detectable impairment (p=0.122).
The waking pattern from sugar-driven reactive hypoglycemia is distinct from generalized insomnia. It characteristically occurs in the second half of the night — between 2am and 4am — with sudden alertness rather than difficulty falling asleep. The wake-up is abrupt because the counterregulatory hormone surge (epinephrine, cortisol) activates the brain rapidly.
How Can You Reduce Nighttime Blood Sugar Drops?
Rovira-Llopis et al. (2024) showed that early time-restricted eating windows reduced fasting glucose (g=-0.38, p<0.01), while late eating windows produced no measurable glycemic improvement despite the same eating window duration. The circadian position of the eating window — not just its length — determined the metabolic benefit.
Vahlhaus et al. (2025) found that late eating tendencies have high heritability. Some people will find the change to earlier eating harder than others due to genetic influence on eating chronotype — this reflects genetic influence, not effort. Individual glucose response monitoring via CGM provides the best evidence of whether a specific timing change is working for a given person. For more on the hunger response that follows nocturnal blood sugar crashes, see Why Do You Wake Up at 3am Hungry?.
What Should You Eat to Prevent Nighttime Blood Sugar Drops?
Diaz-Rizzolo et al. (2024) showed that late eating was associated with lower glucose tolerance independently of diet quality, body weight, and caloric intake in a prediabetes population. The timing variable remained statistically robust after adjusting for every dietary confounder measured. This does not mean food composition is irrelevant — it means timing is an independent factor that works on top of dietary choices.
The combination that produces the worst glucose profiles is high glycemic load plus late timing during peak melatonin. A lower-glycemic last meal — foods with more fiber, protein, and fat relative to refined carbohydrates — produces a smaller and slower glucose rise, reducing the magnitude of any delayed insulin overshoot. Moving that lower-glycemic meal earlier in the evening addresses both variables.
Related Reading
- Metabolic Sleep Disruption
- Can a Blood Sugar Drop Wake You Up at 3am?
- Why Do You Wake Up Hungry at 3am?
- Does Cortisol Wake You Up at 3am?
- Can a Calorie Deficit Cause Insomnia?
- Adrenal Fatigue and 3am Waking
- Insulin Resistance and Insomnia
- Prediabetes and Sleep Problems
- CGM, Nocturnal Glucose, and Sleep
- Intermittent Fasting, Blood Sugar, and Sleep
- Keto and 3am Waking
- Visceral Fat, Growth Hormone, and Sleep
- The Dawn Phenomenon in Non-Diabetics
- Fatty Liver and Sleep Problems
- Gut Dysbiosis, Blood Sugar, and Sleep
References
1. Garaulet, M., Lopez-Minguez, J., Dashti, H. S., Vetter, C., Hernandez-Martinez, A. M., Perez-Ayala, M., Baraza, J. C., Wang, W., Florez, J. C., Scheer, F. A. J. L., & Saxena, R. (2022). Interplay of Dinner Timing and MTNR1B Type 2 Diabetes Risk Variant on Glucose Tolerance and Insulin Secretion: A Randomized Crossover Trial. Diabetes Care, 45(3), 512-519. https://pubmed.ncbi.nlm.nih.gov/35015083/
2. Chellappa, S. L., Qian, J., Vujovic, N., Morris, C. J., Nedeltcheva, A., Nguyen, H., Rahman, N., Heng, S. W., Kelly, L., Kerlin-Monteiro, K., Srivastav, S., Wang, W., Aeschbach, D., Czeisler, C. A., Shea, S. A., Adler, G. K., Garaulet, M., & Scheer, F. A. J. L. (2021). Daytime eating prevents internal circadian misalignment and glucose intolerance in night work. Science Advances, 7(49), eabg9910. https://pubmed.ncbi.nlm.nih.gov/34860550/
3. Gu, C., Brereton, N., Schweitzer, A., Cotter, M., Duan, D., Borsheim, E., Wolfe, R. R., Pham, L. V., Polotsky, V. Y., & Jun, J. C. (2020). Metabolic Effects of Late Dinner in Healthy Volunteers-A Randomized Crossover Clinical Trial. The Journal of Clinical Endocrinology and Metabolism, 105(8), 2789-2802. https://pubmed.ncbi.nlm.nih.gov/32525525/
4. Lopez-Minguez, J., Saxena, R., Bandin, C., Scheer, F. A., & Garaulet, M. (2018). Late dinner impairs glucose tolerance in MTNR1B risk allele carriers: A randomized, cross-over study. Clinical Nutrition, 37(4), 1133-1140. https://pubmed.ncbi.nlm.nih.gov/28455106/
5. Reutrakul, S., & Van Cauter, E. (2014). Interactions between sleep, circadian function, and glucose metabolism: implications for risk and severity of diabetes. Annals of the New York Academy of Sciences, 1311, 151-173. https://pubmed.ncbi.nlm.nih.gov/24628249/
6. Vahlhaus, J., Peters, B., Hornemann, S., Ost, A. C., Kruse, M., Busjahn, A., Pfeiffer, A. F. H., & Pivovarova-Ramich, O. (2025). Later eating timing in relation to an individual internal clock is associated with lower insulin sensitivity and affected by genetic factors. EBioMedicine, 116, 105737. https://pubmed.ncbi.nlm.nih.gov/40305967/
7. Corrado, A., Scida, G., Abuqwider, J., Annuzzi, E., Giosue, A., Pisano, F., Annuzzi, G., & Bozzetto, L. (2025). Interplay among sleep quality, dinner timing, and blood glucose control in users of advanced technologies: A study in a cohort of adults with type 1 diabetes. Diabetes Research and Clinical Practice, 221, 112034. https://pubmed.ncbi.nlm.nih.gov/39929339/
8. Diaz-Rizzolo, D. A., Santos Baez, L. S., Popp, C. J., Borhan, R., Sordi-Guth, A., Manoogian, E. N. C., Panda, S., Cheng, B., & Laferrere, B. (2024). Late eating is associated with poor glucose tolerance, independent of body weight, fat mass, energy intake and diet composition in prediabetes or early onset type 2 diabetes. Nutrition & Diabetes, 14(1), 90. https://pubmed.ncbi.nlm.nih.gov/39455572/
9. Rovira-Llopis, S., Luna-Marco, C., Perea-Galera, L., Banuls, C., Morillas, C., & Victor, V. M. (2024). Circadian alignment of food intake and glycaemic control by time-restricted eating: A systematic review and meta-analysis. Reviews in Endocrine & Metabolic Disorders, 25(2), 325-337. https://pubmed.ncbi.nlm.nih.gov/37993559/
10. Maqsood, S., Amjad, S., Ahmed, F., & Ahmad, M. F. (2026). Time-restricted eating and circadian rhythms: A new frontier in diabetes and obesity management. Primary Care Diabetes, 20(1), 1-12. https://pubmed.ncbi.nlm.nih.gov/41276433/
Written by Kat Fu, M.S., M.S. — Last reviewed: May 2026 — 10 references cited
