Energy intake and sleep quality are linked through hormonal pathways that regulate stress, arousal, and glucose metabolism. Caloric restriction intended to improve health can undermine sleep through these same pathways — driving insulin resistance, increased appetite, and weight gain that counteract the deficit. This article covers the cortisol and orexin mechanisms by which energy deficits disrupt sleep, the role of carbohydrate restriction in reducing melatonin precursors, the evidence on intermittent fasting and sleep, and sleep extension as a reversal strategy. For a broader view of how blood sugar, cortisol, and hormonal changes interact to fragment sleep, see the full article: Metabolic Sleep Disruption: How Blood Sugar, Cortisol, and Hormones Fragment Your Sleep.
How Does Eating Less Affect Cortisol and Sleep?
In a controlled experiment, Tomiyama et al. (2010) assigned 121 women to one of four conditions: caloric restriction at 1,200 kcal/day, calorie monitoring, both, or neither. Restricting caloric intake increased total cortisol output, measured by diurnal salivary cortisol collected over two days — and this increase occurred independently of perceived stress. The women who restricted calories had elevated cortisol whether or not they reported feeling stressed. Monitoring calories, on the other hand, increased perceived stress without raising cortisol. The two dieting behaviors carry separate physiological and psychological costs.
Cortisol elevation depends on the depth of the deficit. A 2016 meta-analysis by Nakamura et al. pooled data from 13 studies (357 participants) and found that fasting-level restriction produced a strong cortisol-elevating effect, while moderate low-calorie diets did not produce reliable cortisol increases on their own. Meta-regression also showed a negative association between cortisol elevation and the duration of restriction — HPA axis activation peaks in the first days of a deficit and attenuates as restriction is sustained over weeks. This time-dependency helps explain conflicting findings across studies: measurements taken early in a restriction period tend to capture cortisol elevations, while longer-term studies in moderate restriction often do not.
Why does elevated cortisol matter for sleep? Dressle et al. (2022) conducted a meta-analysis of 20 case-control studies comparing cortisol output in 449 medication-free people with chronic insomnia versus 357 good sleepers. People with chronic insomnia showed a standardized mean difference (SMD) of 0.50 in cortisol levels (95% CI: 0.21-0.80) — a moderate but consistent elevation. When restricted to blood plasma measurements, the effect size increased to SMD 0.67. The findings are consistent with a 24-hour hyperarousal model: sustained HPA activation drives increased nocturnal cortisol that opposes the normal inhibitory effect of sleep on the stress axis. For someone in a caloric deficit — which itself acutely elevates cortisol — the additive HPA load of concurrent poor sleep compounds the neuroendocrine burden.
Cortisol elevation from caloric restriction and cortisol elevation from insomnia feed into each other. A caloric deficit raises cortisol, elevated cortisol fragments sleep, and fragmented sleep raises cortisol further the next evening. Breaking the cortisol-insomnia feedback loop requires addressing energy balance, sleep duration, or both.
How Does Caloric Restriction Disrupt Sleep Architecture?
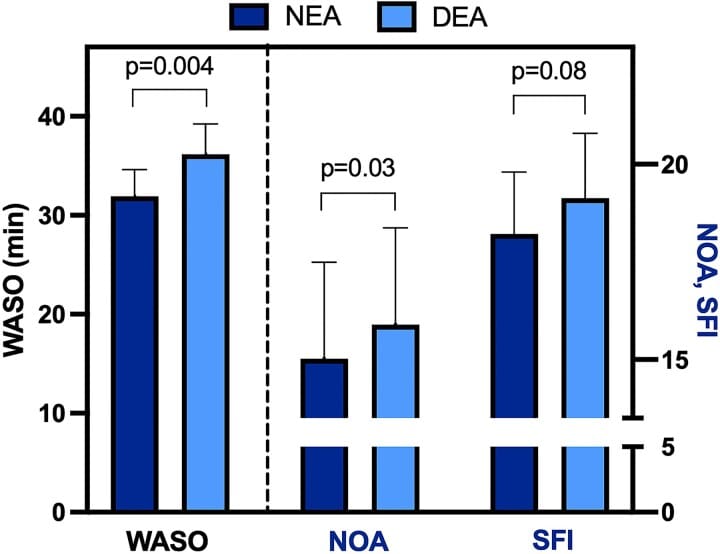
Kim et al. (2025) conducted a controlled crossover study of 38 lean women (mean age 23.6 years), with 17 completing both conditions — neutral energy availability and restricted energy availability — during the early follicular phase across two menstrual cycles, yielding 918 total nights of actigraphy data. Dietary restriction increased wake after sleep onset (WASO) by 4.3 minutes (p = .005), increased the number of nocturnal awakenings (p = .03), and decreased sleep efficiency (p = .01). Even moderate caloric restriction produced measurable objective sleep disruption in healthy, lean women.
Two findings from Kim et al. are relevant here. First, sleep disruption was amplified by menstrual cycle phase: WASO was 9.9 minutes higher in the late luteal phase versus the early follicular phase (p = .008), and awakenings increased by 3.2 events (p = .0006). The hormonal environment of the late luteal phase — higher progesterone, fluctuating estrogen — compounds the sleep-disrupting effects of energy deficit. Second, orexin-A showed a positive association with WASO (p = .02), implicating this wake-promoting neuropeptide as a mediator of restriction-induced sleep fragmentation. Orexin-A is upregulated during energy deficit, and its role is to maintain wakefulness when food is scarce — an adaptive response that becomes counterproductive when the deficit is deliberate.
Rogers et al. (2024) reviewed the broader metabolic consequences of this sleep disruption. Inadequate sleep elevates cortisol through HPA axis disinhibition, and elevated cortisol amplifies hunger, promotes preferential fat storage, and suppresses satiety — creating a physiological environment that counteracts caloric restriction. The review also identified exercise as having measurable capacity to attenuate the adverse metabolic effects of sleep disruption, potentially by improving HPA axis regulation and increasing insulin sensitivity independently of sleep duration.
Carbohydrate restriction introduces a separate mechanism. Reduced carbohydrate intake lowers insulin’s role in clearing competing amino acids from the bloodstream, which reduces tryptophan’s relative availability for transport across the blood-brain barrier. Tryptophan is the amino acid precursor to serotonin, which is in turn the precursor to melatonin. This is a plausible mechanism for melatonin reduction during low-carb dieting, though it is less directly studied in humans than the cortisol pathway.
Most people have 2-3 causes working together, not just one. Calorie-deficit-driven cortisol elevation is one of several metabolic mechanisms that fragment sleep. The 3AM Decoder maps which causes are active in your case — so you can address the right ones.
Can Extending Sleep Reduce the Metabolic Cost of Dieting?
Tasali et al. (2022) enrolled 80 overweight adults (mean age 29.8 years; 51.3% male) who habitually slept fewer than 6.5 hours per night and assigned them to a two-week sleep extension targeting 8.5 hours in bed or a control condition maintaining habitual sleep. The sleep extension group increased sleep duration by approximately 1.2 hours per night (95% CI: 1.0-1.4 hours; p < .001), and energy intake decreased by a mean of 270 kcal per day compared to controls (95% CI: -393 to -147 kcal/d; p < .001), measured objectively via doubly labeled water. Total energy expenditure did not differ between groups, meaning the reduced intake produced a net negative energy balance — mirroring the caloric reduction of a moderate diet, achieved through sleep normalization rather than food restriction.
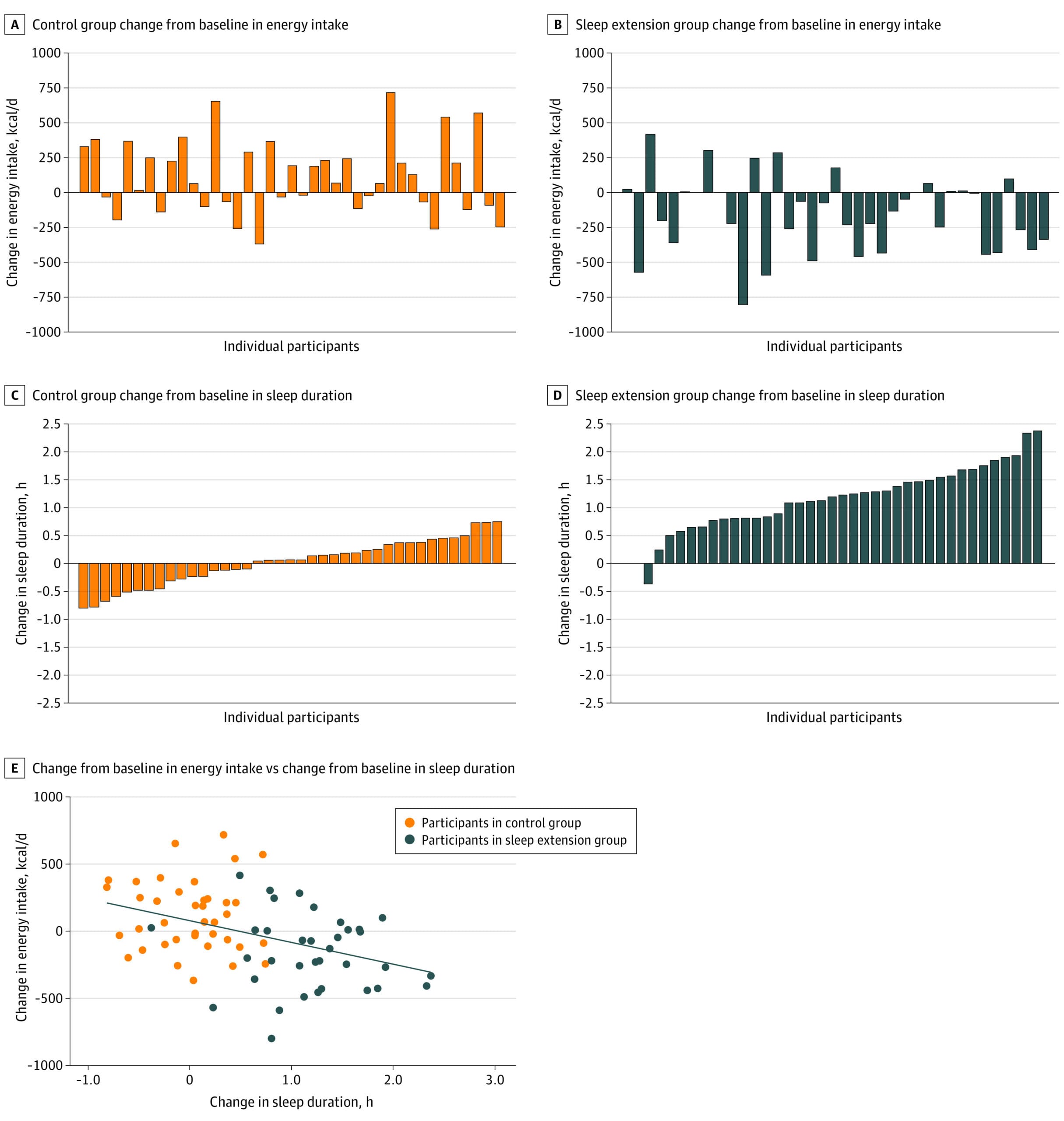
Sleep duration was inversely correlated with energy intake across individuals (r = -0.41; 95% CI: -0.59 to -0.20; p < .001), suggesting a dose-response relationship: greater sleep recovery produced greater spontaneous reduction in caloric intake.
For someone in a caloric deficit who is also sleeping poorly, this has direct implications. Rather than increasing the deficit or adjusting macronutrients — both of which can raise cortisol and fragment sleep further — extending sleep duration may produce an equivalent caloric outcome through a different mechanism: reduced voluntary intake driven by improved appetite regulation. Duraccio et al. (2024) reviewed the emerging evidence for sleep extension as a cardiometabolic strategy and noted that larger increases in sleep duration were associated with improvements in insulin sensitivity (measured through fasting markers and HOMA-IR), and that adolescents extending sleep also increased consumption of foods with a low glycemic index — suggesting sleep extension improves not only how much but what people choose to eat.
Longer sleep allows a deeper cortisol nadir overnight, which reduces HPA activation, which reduces nocturnal awakenings, which supports longer sleep the next night. For someone whose deficit-driven cortisol elevation is fragmenting sleep, prioritizing sleep duration may be more effective than modifying the diet.
Does Intermittent Fasting Cause Sleep Problems?
Nakamura et al. (2016) found that fasting — defined as complete or near-complete energy deprivation — produced a strong cortisol-elevating effect, while moderate caloric restriction did not. For intermittent fasting specifically, this distinction matters: schedules that produce extended periods without food (16:8, 20:4, alternate-day fasting) may acutely activate the HPA axis more than evenly distributed moderate restriction across the day.
Eating window timing adds a variable. An eating window that ends early in the evening may deplete hepatic glycogen before the second half of the night, when cortisol begins its natural pre-dawn rise. If glycogen stores are low at that point, the body initiates gluconeogenesis — a cortisol-dependent process — earlier and more aggressively, which can trigger awakenings.
Bohlman et al. (2024) reviewed 6 controlled trials (548 enrolled participants, 430 completers) examining time-restricted eating and sleep. The results were mixed: one study found that skipping dinner in a time-restricted eating schedule reduced evening cortisol, while others found reductions in sleep efficiency of 2-2.7%. The review noted that time-restricted eating altered the expression of six circadian rhythm genes. The evidence suggests that adaptation may occur — acute cortisol elevation attenuates over weeks, consistent with the Nakamura meta-analysis — but the first days to weeks of an intermittent fasting schedule carry the highest sleep disruption risk.
Does Dieting Affect Melatonin Production?
Tryptophan crosses the blood-brain barrier via a transporter it shares with other large neutral amino acids (leucine, isoleucine, valine, tyrosine, phenylalanine). Carbohydrate intake triggers insulin release, and insulin clears those competing amino acids into muscle tissue, increasing the ratio of tryptophan to its competitors and increasing brain tryptophan uptake. When carbohydrate intake drops, this insulin-mediated clearance is reduced, and tryptophan competes less effectively for transport.
Once in the brain, tryptophan is converted to serotonin, and serotonin is converted to melatonin in the pineal gland. Reduced tryptophan availability can lower serotonin production, which in turn may reduce melatonin synthesis. Rogers et al. (2024) discussed this mechanistic chain in the context of how sleep disruption drives insulin resistance and cortisol elevation — noting that cortisol mediates hunger amplification and that the relationship between sleep, metabolism, and appetite hormones is bidirectional.
This mechanism is plausible and consistent with the known biochemistry, but direct human studies measuring melatonin output during carbohydrate-restricted diets are limited. The cortisol pathway described in the sections above has stronger and more direct evidence linking caloric deficit to sleep disruption.
Why Can’t I Sleep When I’m in a Calorie Deficit?
Cortisol drives the first mechanism. As Tomiyama et al. (2010) demonstrated, caloric restriction at 1,200 kcal/day raised cortisol output independently of psychological stress. Elevated cortisol delays the evening cortisol nadir that normally facilitates sleep onset, increases nocturnal HPA axis activity, and produces more frequent awakenings — the same pattern Dressle et al. (2022) documented in chronic insomnia.
Orexin-A drives the second mechanism. Kim et al. (2025) found that orexin-A levels were positively associated with wake after sleep onset during caloric restriction in lean women. Orexin-A is a neuropeptide that promotes and maintains wakefulness; its upregulation during energy deficit is an adaptive response — keeping the organism alert to find food — but it works against sleep when the deficit is intentional.
These two pathways — cortisol-mediated arousal and orexin-driven wakefulness — produce lighter, more fragmented sleep with more awakenings. A person in a deficit may fall asleep adequately but wake repeatedly through the night, spend more time in lighter sleep stages, and feel unrefreshed in the morning. The sleep disruption then compounds the metabolic cost of the deficit: fragmented sleep itself raises cortisol, increases hunger hormones, promotes insulin resistance, and can redirect weight loss away from fat and toward lean mass.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the full metabolic cause overview
- Can a Blood Sugar Drop Wake You Up at 3am? — how nocturnal blood sugar drops trigger the hormonal cascade that wakes you up
- Why Do You Wake Up Hungry at 3am? — how leptin, ghrelin, and blood sugar interact during sleep
- Why Does Cortisol Spike at 3am and Wake You Up? — the cortisol-sleep feedback loop and HPA axis timing
- Is Waking Up at 3am a Sign of Adrenal Fatigue? — the real HPA axis mechanism behind what people call adrenal fatigue
- Does Insulin Resistance Affect Sleep Quality? — the bidirectional relationship between insulin sensitivity and deep sleep
- Can Prediabetes Cause Sleep Problems? — how prediabetes changes sleep architecture and accelerates metabolic decline
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 8 references cited
References
Bohlman, C., McLaren, C., Ezzati, A., Vial, P., Ibrahim, D., & Anton, S. D. (2024). The effects of time-restricted eating on sleep in adults: A systematic review of randomized controlled trials. Frontiers in Nutrition, 11, 1419811. https://pubmed.ncbi.nlm.nih.gov/39144285/
Dressle, R. J., Feige, B., Spiegelhalder, K., Schmucker, C., Benz, F., Mey, N. C., & Riemann, D. (2022). HPA axis activity in patients with chronic insomnia: A systematic review and meta-analysis of case-control studies. Sleep Medicine Reviews, 62, 101588. https://pubmed.ncbi.nlm.nih.gov/35091194/
Duraccio, K. M., Kamhout, S., Baron, K. G., Reutrakul, S., & Depner, C. M. (2024). Sleep extension and cardiometabolic health: What it is, possible mechanisms and real-world applications. The Journal of Physiology, 602(23), 6571-6586. https://pubmed.ncbi.nlm.nih.gov/38268197/
Kim, A. E., Ku, B., Li, J., Bodurka, J., & Bhatt, D. L. (2025). Caloric restriction, the menstrual cycle, and sleep in women without obesity. The Journal of Clinical Endocrinology and Metabolism, 110(11), 3108-3117. https://pubmed.ncbi.nlm.nih.gov/40042851/
Nakamura, Y., Walker, B. R., & Ikuta, T. (2016). Systematic review and meta-analysis reveals acutely elevated plasma cortisol following fasting but not less severe calorie restriction. Stress, 19(2), 151-157. https://pubmed.ncbi.nlm.nih.gov/26586092/
Rogers, E. M., Banks, N. F., & Jenkins, N. D. M. (2024). The effects of sleep disruption on metabolism, hunger, and satiety, and the influence of psychosocial stress and exercise: A narrative review. Diabetes/Metabolism Research and Reviews, 40(2), e3667. https://pubmed.ncbi.nlm.nih.gov/37269143/
Tasali, E., Wroblewski, K., Kahn, E., Kilkus, J., & Schoeller, D. A. (2022). Effect of sleep extension on objectively assessed energy intake among adults with overweight in real-life settings: A randomized clinical trial. JAMA Internal Medicine, 182(4), 365-374. https://pubmed.ncbi.nlm.nih.gov/35129580/
Tomiyama, A. J., Mann, T., Vinas, D., Hunger, J. M., DeJager, J., & Taylor, S. E. (2010). Low calorie dieting increases cortisol. Psychosomatic Medicine, 72(4), 357-364. https://pubmed.ncbi.nlm.nih.gov/20368473/
