Cortisol’s role in sleep is more than “stress hormone disrupts rest.” Cortisol follows a precise 24-hour rhythm that is necessary for healthy sleep-wake transitions. The 3am waking pattern reflects a timing disruption in that rhythm, rooted in HPA axis regulation. Understanding this matters for long-term brain health: chronic HPA axis dysregulation is associated with hippocampal volume loss and accelerated cognitive decline. This article covers the cortisol-sleep relationship — why cortisol rises at 3am, what the cortisol awakening response is, and how sleep architecture and cortisol regulate each other. For the broader metabolic context, see Metabolic Sleep Disruption: The Full Picture.
What Is the Normal Cortisol Rhythm and Why Does It Rise Before You Wake?
The 24-hour cortisol curve is one of the strongest circadian rhythms in the human body. Cortisol concentration drops to its lowest point (its nadir) around midnight to 2am, stays low during the first half of the night, then begins a gradual rise from approximately 2-3am onward. By the time of waking, cortisol is near its daily peak.
This pre-waking rise is not pathological. It is orchestrated by the suprachiasmatic nucleus (SCN) — the brain’s central circadian clock. The SCN controls pre-awakening adrenal sensitivity to adrenocorticotropic hormone (ACTH) through an extra-pituitary pathway, meaning the morning cortisol surge requires circadian “release” from overnight inhibition rather than a stress-based trigger (Clow et al., 2010).
The cortisol awakening response (CAR) is the rapid increase in cortisol that occurs in the 30 minutes after waking. In healthy adults, salivary cortisol typically increases by 38-75% during this window, peaking at approximately 30 minutes post-arousal (Elder et al., 2014). The CAR is linked to the resolution of sleep inertia — that groggy, slow-to-start feeling on waking — and functions as an energy-mobilization event at the sleep-to-wake transition.
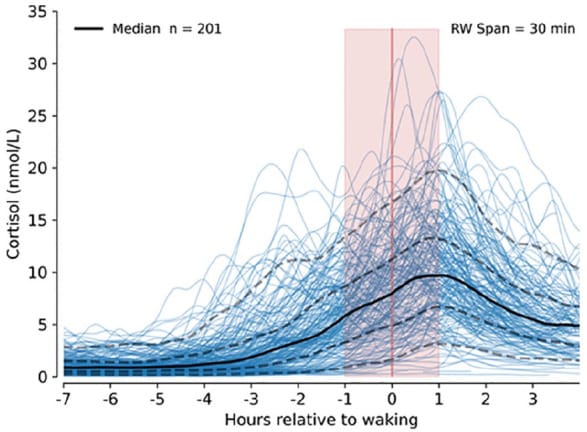
A 2025 study using continuous subcutaneous microdialysis in 201 healthy volunteers challenged the assumption that the CAR is a distinct post-waking event. Velazquez Sanchez and Dalley found that, at the population level, the rate of cortisol increase in the hour after waking did not differ from the rate during the hour before waking. Sleep duration modulated the timing of peak cortisol: long sleepers (mean 548 minutes) showed maximal cortisol release approximately 97 minutes before waking, while short sleepers (mean 369 minutes) showed peak increases approximately 12 minutes after waking. Circadian-misaligned sleepers peaked 68 minutes before waking, while circadian-aligned sleepers peaked 12 minutes post-waking (Velazquez Sanchez & Dalley, 2025). This suggests the pre-waking cortisol rise may be a continuation of circadian rhythmicity whose timing relative to waking depends on sleep duration and circadian alignment.
For someone waking at 3am, the practical implication is this: if circadian alignment or sleep duration is altered, the cortisol peak can arrive hours earlier than it should.
How Does Sleep Disruption Drive Cortisol Higher and How Does Elevated Cortisol Fragment Sleep?
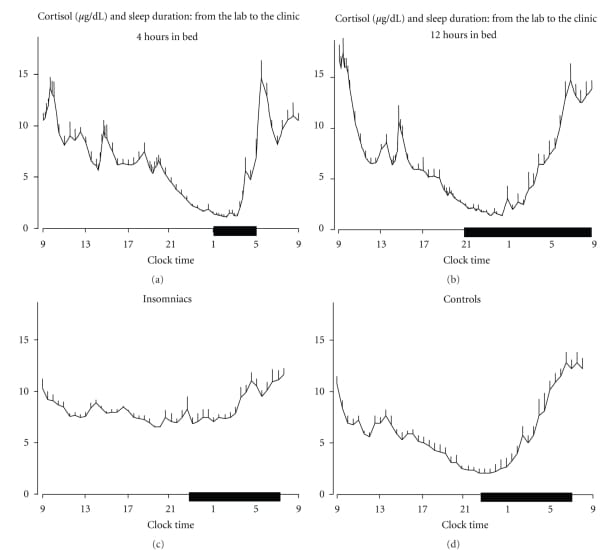
The relationship between cortisol and sleep runs in both directions. Balbo, Leproult, and Van Cauter documented this bidirectional feedback in a 2010 review: sleep onset exerts a measurable inhibitory effect on cortisol secretion, while awakenings and sleep offset actively stimulate cortisol release. Elevated HPA axis activity then feeds back to suppress slow-wave sleep and impair sleep continuity (Balbo et al., 2010).
Sleep fragmentation — frequent brief arousals, even without full waking — independently elevates plasma cortisol in otherwise healthy individuals. The fragmentation itself, rather than the reduction in total sleep time, is enough to activate the HPA axis. In people with chronic insomnia, both ACTH and cortisol are elevated across the full 24-hour period, consistent with a state of central nervous hyperarousal that overrides the inhibitory influence sleep normally exerts on cortisol (Balbo et al., 2010).
The bidirectional nature of this relationship means the question “is cortisol causing the waking, or is the waking causing the cortisol?” does not have a one-directional answer. Both are happening. Fragmented sleep elevates cortisol, and elevated cortisol makes subsequent sleep more fragmented — particularly by suppressing slow-wave (N3) sleep, which is the stage with the strongest cortisol-inhibiting effect.
A 2024 polysomnography study in 35 menopausal women by Sahola et al. added an important nuance. Objective sleep disruption — measured as lower sleep efficiency, reduced slow-wave sleep percentage, higher wake after sleep onset, and fewer REM sleep periods — was associated with elevated cortisol levels. But self-reported insomnia and sleepiness showed no association with cortisol. The dissociation held after adjusting for age, BMI, vasomotor experiences, and depression scores (Sahola et al., 2024).
The implication: cortisol elevation can be driven by sleep architecture disruption that the person does not perceive as poor sleep. Someone sleeping six hours with frequent micro-arousals and reduced slow-wave sleep may report “sleeping fine” while carrying elevated cortisol across the full day. The subjective experience and the physiological state do not have to match.
How Do You Know If Cortisol Is Driving Your 3am Wakeups?
The experience of a cortisol-driven 3am waking is recognizable. People describe eyes snapping open — not groggy, but fully alert. Racing thoughts begin without a conscious trigger. A feeling of unease rises without an identifiable cause. Heart rate may be elevated. Falling back to sleep is difficult because the arousal state is neurochemical, not psychological. This contrasts with blood sugar-driven waking, which tends to come with hunger, sweating, or a sense of low energy rather than sharp alertness.
In a study of 34 people with chronic insomnia (mean age 45.3 years), Passos et al. (2023) found insomnia severity correlated with morning cortisol (r=0.37, p=0.03). Insomnia severity also correlated with depression (r=0.44, p<0.01) and tension-related unease (r=0.39, p=0.02). Polysomnography data from the same study showed that N3 (slow-wave) sleep had a protective association — it negatively correlated with both tension-related unease (r=-0.36, p=0.04) and fatigue (r=-0.37, p=0.04). When slow-wave sleep is reduced, the buffer against cortisol-driven arousal is weakened.
A 2025 study by Lee et al. in 71 healthy adults (aged 25-37) found that the relationship between stress and morning cortisol depends on the type of stress burden. Greater exposure to major life stressors over the preceding five years predicted elevated morning cortisol (p=0.014). But higher psychopathological burden predicted lower morning cortisol (p=0.016) — consistent with HPA axis blunting, a pattern documented in burnout and prolonged stress exposure. Perceived stress did not independently predict morning cortisol. The combined model explained 68.9% of the variance in morning cortisol across participants (Lee et al., 2025).
This produces two distinct patterns. Acute or accumulated life stress tends to drive cortisol upward — and this is the pattern more commonly associated with 3am waking, racing thoughts, and difficulty returning to sleep. Chronic psychological burden, by contrast, tends to produce a blunted cortisol response, where the HPA axis output is suppressed rather than elevated — this produces a different set of sleep disruption patterns, often characterized more by fatigue than hyperarousal.
For measuring cortisol, the standard approach is morning salivary cortisol at the moment of waking, plus a second sample 30 minutes later. This two-sample method captures the cortisol awakening response and can distinguish between an elevated, normal, or blunted CAR (Elder et al., 2014).
Cortisol dysregulation frequently co-occurs with blood sugar instability and metabolic inflammation. The 3am waking may involve more than one mechanism — cortisol is often one contributor among several.
The 3AM Decoder can help you identify which metabolic, hormonal, and neurological factors may be driving your 3am waking pattern. Based on your answers, it maps the combination of contributors — including cortisol timing — and explains what they mean for your sleep architecture.
Is Waking Up at 3am a Sign of Adrenal Fatigue?
The label “adrenal fatigue” implies the adrenal glands are exhausted from overwork. What the research describes is more precise: the HPA axis can be in a state of hyperactivation (producing too much cortisol) or hypoactivation (producing too little). These are different states with different causes and different downstream effects.
Lee et al. (2025) demonstrated this distinction directly. In 71 healthy adults, greater life stressor burden predicted elevated morning cortisol, while higher psychopathological burden predicted lower morning cortisol. The two patterns are opposite ends of HPA axis regulation, and lumping them under one label obscures what is happening.
If cortisol is elevated (the hyperactivation pattern), 3am waking with racing thoughts and a sense of unease is characteristic. If cortisol is blunted (the hypoactivation pattern), the sleep disruption tends to present differently — more as fatigue, difficulty waking, and unrefreshing sleep rather than the sharp nocturnal arousal associated with high cortisol.
Can You Lower Cortisol at Night to Sleep Better?
The bidirectional relationship between cortisol and sleep means that improving one can improve the other. Balbo et al. (2010) documented that sleep onset inhibits cortisol and that slow-wave sleep exerts a particularly strong suppressive effect on HPA axis output. Increasing slow-wave sleep and improving sleep continuity reduces cortisol — and lower cortisol, in turn, supports deeper and less fragmented sleep.
Sahola et al. (2024) reinforced this with an important finding: objective sleep quality (measured by polysomnography) predicted cortisol levels, while self-reported sleep quality did not. This means that addressing the factors that disrupt sleep architecture — even when the person does not perceive their sleep as poor — is more relevant than cortisol-lowering strategies applied in isolation.
Investigating the root cause of the sleep architecture disruption matters more than targeting cortisol directly. The cortisol elevation is often downstream of something else: circadian misalignment, blood sugar instability, chronic inflammation, or hormonal changes. Addressing the upstream cause is what allows the cortisol rhythm to normalize.
Why Do You Wake Up at 3am with Anxiety?
The cortisol rise that begins in the second half of the night is a circadian event — it occurs whether or not there is anything to be anxious about. When this rise happens too early or too steeply, it activates the sympathetic nervous branch: heart rate increases, alertness heightens, and the subjective experience is an anxious, activated state — even though the trigger is hormonal rather than psychological.
Clow et al. (2010) described this as part of the SCN-driven pre-awakening process: upon gaining consciousness, a “flip-flop” switch of regional brain activation simultaneously initiates HPA axis stimulation and restores adrenal sensitivity to ACTH. If the circadian timing of this switch is advanced — as it can be with short sleep duration or circadian misalignment — the arousal occurs at 3am instead of 6am.
Passos et al. (2023) found that insomnia severity correlates with both morning cortisol (r=0.37) and tension-related unease (r=0.39). N3 (slow-wave) sleep negatively correlated with tension-related unease (r=-0.36), meaning that people with more slow-wave sleep had lower scores on that measure. The protective role of slow-wave sleep against cortisol-driven arousal is consistent across studies: when N3 sleep is reduced, the inhibitory effect that deep sleep exerts on the HPA axis is diminished, and cortisol-driven waking becomes more likely.
The nocturnal unease is not “in your head.” It is a neurochemical state produced by the intersection of cortisol timing, sympathetic activation, and insufficient slow-wave sleep.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the full metabolic cause overview
- Can a Blood Sugar Drop Wake You Up at 3am? — how nocturnal blood sugar drops trigger the hormonal cascade that wakes you up
- Why Do You Wake Up Hungry at 3am? — how leptin, ghrelin, and blood sugar interact during sleep
- Can a Calorie Deficit Cause Insomnia? — how energy restriction elevates cortisol and fragments sleep
- Is Waking Up at 3am a Sign of Adrenal Fatigue? — the real HPA axis mechanism behind what people call adrenal fatigue
- Does Insulin Resistance Affect Sleep Quality? — the bidirectional relationship between insulin sensitivity and deep sleep
- Can Prediabetes Cause Sleep Problems? — how prediabetes changes sleep architecture and accelerates metabolic decline
References
1. Clow, A., Hucklebridge, F., Stalder, T., Evans, P., & Thorn, L. (2010). The cortisol awakening response: more than a measure of HPA axis function. Neuroscience & Biobehavioral Reviews, 35(1), 97-103. https://pubmed.ncbi.nlm.nih.gov/20026350/
2. Balbo, M., Leproult, R., & Van Cauter, E. (2010). Impact of sleep and its disturbances on hypothalamo-pituitary-adrenal axis activity. International Journal of Endocrinology, 2010, 759234. https://pubmed.ncbi.nlm.nih.gov/20628523/
3. Elder, G. J., Wetherell, M. A., Barclay, N. L., & Ellis, J. G. (2014). The cortisol awakening response — applications and implications for sleep medicine. Sleep Medicine Reviews, 18(3), 215-224. https://pubmed.ncbi.nlm.nih.gov/23835138/
4. Passos, G. S., Youngstedt, S. D., Santana, M. G., De Aquino Lemos, V., Tufik, S., & De Mello, M. T. (2023). Insomnia severity is associated with morning cortisol and psychological health. Sleep Science, 16(1), e56-e63. https://pubmed.ncbi.nlm.nih.gov/37151768/
5. Sahola, N., et al. (2024). Worse sleep architecture but not self-reported insomnia and sleepiness is associated with higher cortisol levels in menopausal women. Maturitas, 187, 108056. https://pubmed.ncbi.nlm.nih.gov/38909441/
6. Velazquez Sanchez, C., & Dalley, J. W. (2025). The cortisol awakening response: fact or fiction? Brain and Neuroscience Advances, 9. https://pubmed.ncbi.nlm.nih.gov/40297522/
7. Lee, K. F. A., et al. (2025). Effects of exposure to life stressors, perceived stress, and psychopathological symptoms on cortisol awakening response: individual differences in resilience. Stress and Health, 41(3), e70044. https://pubmed.ncbi.nlm.nih.gov/40392433/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
