The 3am waking pattern reported across keto, carnivore, and intermittent fasting communities maps onto a documented metabolic mechanism. The body manages overnight glucose through two fuel sources — stored glycogen and gluconeogenesis (the liver producing new glucose from non-carbohydrate substrates). Carbohydrate restriction changes the ratio between them, and that change has consequences for sleep during the second half of the night.
Overnight glucose stability and sleep continuity both independently predict long-term metabolic health, insulin sensitivity, and cognitive function. When carbohydrate restriction disrupts one, it cascades into the other.
This article covers the metabolic cascade from carbohydrate restriction through glycogen depletion, cortisol-driven gluconeogenesis, and nocturnal blood glucose changes that produce 3am waking. For the full metabolic overview, see Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep. For blood sugar drops specifically, see Can a Blood Sugar Drop Wake You Up at 3am?.
What Happens to Liver Glycogen When You Cut Carbs?
Liver glycogen is the body’s overnight glucose buffer. During sleep, while no food is coming in, the brain — which cannot store meaningful amounts of energy — draws on the liver’s glycogen reserves. The liver converts glycogen to glucose through glycogenolysis (the enzymatic breakdown of glycogen into glucose-1-phosphate) and releases it into the bloodstream at a rate that matches the brain’s requirements.
On a standard mixed diet, this reservoir refills with each meal containing carbohydrates. The 80-120g of hepatic glycogen is sufficient to sustain blood glucose for roughly 18-24 hours without food, depending on activity level and body composition. Under these conditions, the overnight fast — typically 8-12 hours — leaves adequate glycogen remaining by morning.
A ketogenic diet changes this math. With daily carbohydrate intake restricted to under 20-50g, the liver cannot replenish glycogen stores to full capacity. After several days of sustained carbohydrate restriction, hepatic glycogen stabilizes at a lower baseline. The body adapts by increasing reliance on the alternative glucose-production pathway: gluconeogenesis, where the liver synthesizes glucose from amino acids (primarily alanine), glycerol released from fat breakdown, and lactate.
Khani and Tayek (2001) quantified this transition in a controlled human study. At 16 hours of fasting — a duration that approximates what happens on a low-carb diet by the middle of the night — gluconeogenesis accounted for 55-57% of total glucose production. This was the baseline before any cortisol was manipulated. The body had already moved more than halfway from glycogen-dependent to gluconeogenesis-dependent glucose supply.
This is the metabolic setup. The glycogen buffer that normally smooths out overnight blood glucose is reduced. The liver compensates with gluconeogenesis, which is a slower, hormone-responsive, and more variable process than glycogenolysis. At the point in the night when cortisol begins its scheduled rise — between 2 and 4am — that gluconeogenic machinery is what cortisol acts on. With less glycogen to release cleanly, the cortisol response produces a different metabolic outcome than it would on a carbohydrate-adequate diet.
How Does the 3-4am Cortisol Rise Affect Blood Sugar on a Low-Carb Diet?
The pre-dawn cortisol rise is a scheduled circadian event. Cortisol secretion follows a 24-hour pattern, reaching its nadir around midnight and beginning to climb between 2 and 4am, peaking at approximately 6-8am. This rise serves a specific function: preparing glucose and fatty acids so the body has fuel available when you wake.
On a standard diet with a full glycogen buffer, this cortisol rise directs the liver to release stored glucose — a fast, regulated process. Glycogenolysis responds predictably to cortisol, and the glucose release is proportionate.
On a ketogenic diet, the glycogen buffer is reduced. Cortisol instead acts on gluconeogenesis — stimulating the liver to convert amino acids and glycerol into glucose. This pathway is more responsive to cortisol dose. Khani and Tayek (2001) demonstrated this in a controlled pituitary-pancreatic clamp study: when high-dose hydrocortisone was infused while holding insulin, growth hormone, and glucagon constant, the entire increase in glucose production was accounted for by gluconeogenesis. It rose from a fasting baseline of approximately 35-40% to 65-66% of total glucose production. Blood glucose increased to 7.9 mmol/L (approximately 142 mg/dL).
Dinneen et al. (1995) isolated the metabolic effects of the normal nocturnal cortisol rise specifically. Using metyrapone to suppress endogenous cortisol and then replacing it with a controlled infusion in type 1 diabetic individuals, they showed that the nocturnal cortisol rise increased glucose appearance in the bloodstream, reduced tissue glucose uptake, and increased beta-hydroxybutyrate concentrations — a ketone body. The cortisol rise at 3-4am simultaneously drives both gluconeogenesis and ketogenesis, meaning the same hormonal event that raises blood glucose also upregulates ketone production.
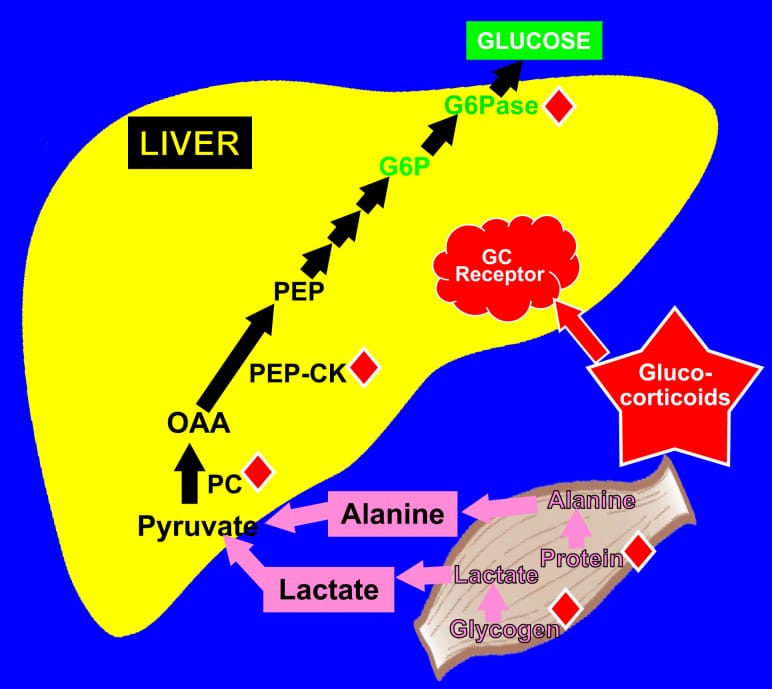
DeFronzo and Auchus (2025) describe what happens when this cortisol-gluconeogenesis interaction compounds. Their review proposes a “reverberating cycle”: cortisol disrupts circadian rhythms, drives insulin resistance, and the resulting hyperinsulinemia further dysregulates the hypothalamic-pituitary-adrenal (HPA) axis — the hormonal control center governing cortisol release. This creates a self-amplifying loop.
On a ketogenic diet, the absence of a glycogen buffer means the nocturnal cortisol rise drives a larger gluconeogenic response. The resulting blood glucose fluctuation — potentially rising, then dipping as insulin responds — can cross the threshold that triggers arousal. The person wakes up because the normal cortisol mechanism is acting on a substrate supply that responds differently than it would with adequate glycogen.
Does a Ketogenic Diet Change Sleep Architecture?
Afaghi, O’Connor, and Chow (2008) conducted the only polysomnographic (overnight brain-wave recording) controlled study using a repeated-measure design comparing a diet with less than 1% carbohydrate and a mixed diet in healthy, non-obese men. Fourteen males (ages 18-35, BMI 23.4 kg/m2) underwent sleep recording under the carbohydrate-restricted diet (38% protein, 61% fat, less than 1% carbohydrate, 2400 kcal) versus an isocaloric control diet (72% carbohydrate).
The findings were specific. REM sleep — the dream-state sleep phase associated with memory consolidation and emotional processing — decreased as a proportion of total sleep time. This reduction was present during both the acute carbohydrate-restriction phase (P=0.006) and the established ketosis phase (P=0.05), compared to the control diet.
Slow-wave sleep (stage 4) — the deepest sleep phase, associated with physical recovery, growth hormone release, and immune function — increased: 17.7% and 17.8% of total sleep time during the acute and ketosis phases, respectively, compared to 13.9% on the control diet (P=0.02 for both comparisons).
This redistribution — less REM, more slow-wave sleep — under carbohydrate restriction is consistent with the brain allocating more time to deep restorative processes when substrate availability changes. Because participants were healthy and non-obese, the dietary change itself was the primary variable.
Xie, Li, and Bai (2026) provided a different angle. In a retrospective analysis of 220 people with polycystic ovary syndrome (PCOS) undergoing a ketogenic diet program, 42% developed sleep disorders during the study period. Impaired blood glucose metabolism was the strongest predictor: an odds ratio of 5.278 (95% CI: 1.533-18.177, P=0.008). That odds ratio was higher than anxiety (OR 1.768), depression (OR 1.494), or elevated LDL cholesterol (OR 1.619). The predictive model achieved an area under the ROC curve of 0.93, demonstrating strong discriminative accuracy.
The Xie et al. finding matters because it quantifies the relative contribution: glycemic disruption carries a 5-fold higher odds of sleep disorder than either psychological factor. This positions blood glucose instability as a primary mechanism of keto-related sleep disruption, not a secondary effect of stress or mood changes.
Pasca et al. (2024) reviewed 20 studies on ketogenic dietary therapies and sleep outcomes. The review found that chronic ketogenic diets were associated with improved overall sleep quality, fewer nighttime awakenings, and decreased daytime sleepiness — but with an important limitation. The majority of evidence came from neurological disease cohorts (drug-resistant epilepsy, autism spectrum disorder, migraine) where the ketogenic diet produces anti-seizure and anti-inflammatory effects that independently benefit sleep. Because the evidence base draws predominantly from these neurological-disease cohorts, direct extrapolation to metabolically healthy adults on a ketogenic diet is uncertain.
The acute-versus-chronic distinction emerges across the available evidence: acute carbohydrate restriction disrupts sleep architecture; long-term adaptation may resolve some of those changes. But the evidence base for healthy populations remains limited.
Does Keto Insomnia Go Away with Adaptation?
The Afaghi et al. (2008) data tracked participants through two distinct phases — an acute carbohydrate-restriction phase and an established ketosis phase. REM suppression persisted into the ketosis phase (P=0.05), not resolving with metabolic adaptation to ketone utilization. The increase in slow-wave sleep also persisted. This suggests the architecture change is not an acute stress response that resolves as the body adapts to burning ketones for fuel.
Pasca et al. (2024) found improved sleep outcomes in chronic ketogenic diet populations — but the populations studied were predominantly neurological disease cohorts. In drug-resistant epilepsy, the ketogenic diet produces anti-seizure effects through multiple mechanisms (GABAergic tone modulation, adenosine receptor activity, circadian clock gene expression) that independently benefit sleep. These benefits are not transferable to metabolically healthy people doing keto for body composition.
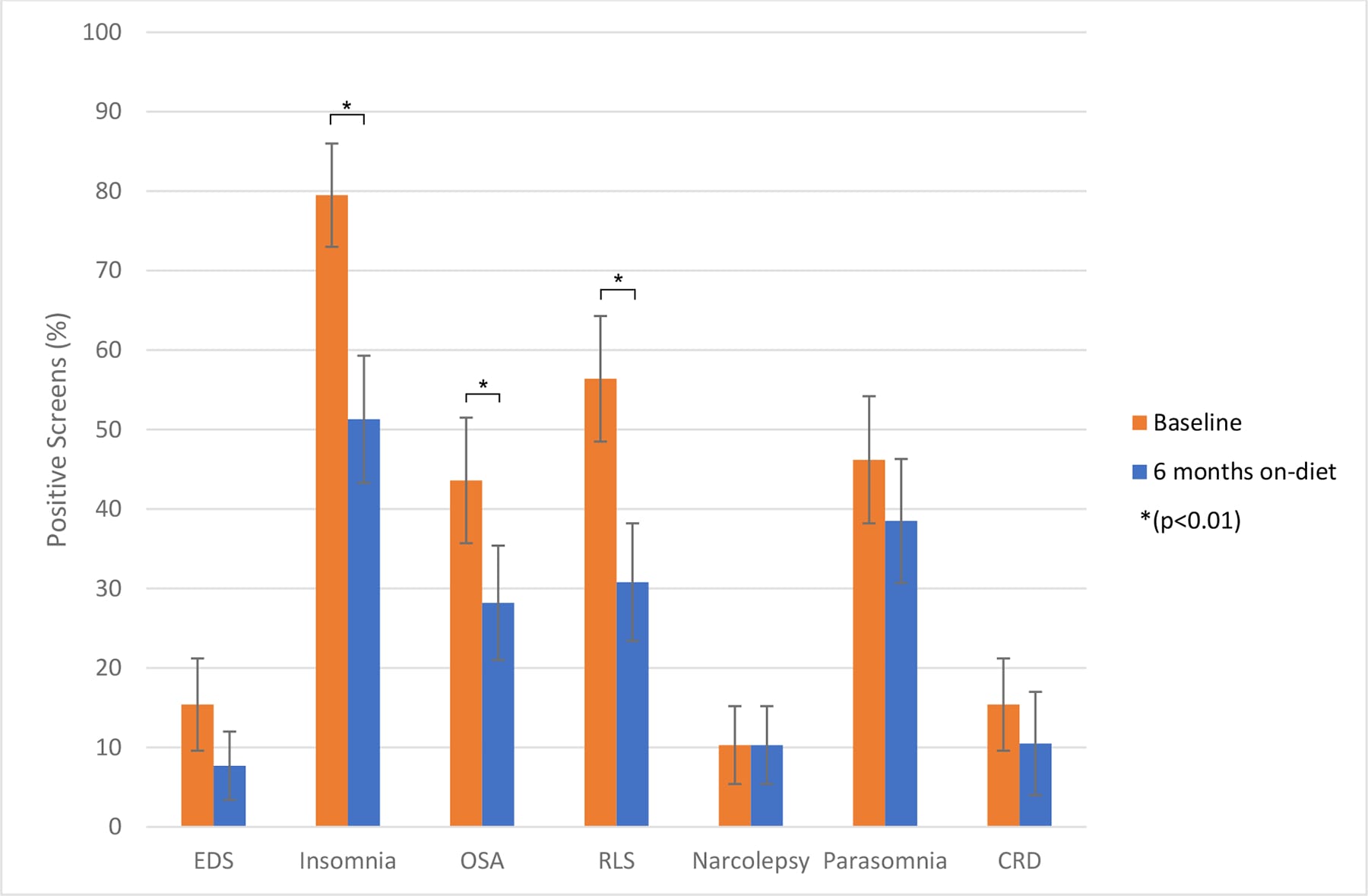
Perlman et al. (2024) provided some of the more detailed quantified evidence for improvement over time. In a 6-month ketogenic diet study in which 39 of 45 enrolled individuals with relapsing multiple sclerosis completed sleep assessments, daytime sleepiness improved (mean reduction of 1.90 points on the Epworth Sleepiness Scale, P<0.001), total sleep disorder burden fell (4.4-point reduction, P=0.002), and domain-level improvements appeared for insomnia, obstructive sleep apnea, and restless leg syndrome. These improvements occurred without changes in total sleep duration — the quality changed, not the quantity.
The summary: adaptation may resolve some acute sleep disruption, particularly the noticeable insomnia and wakefulness in the first weeks. But the evidence supporting this comes from disease populations with confounding therapeutic benefits, not from healthy adults doing keto.
Meanwhile, the cortisol-gluconeogenesis mechanism described in the sections above persists as long as hepatic glycogen remains depleted — which is, by definition, the entire duration of a ketogenic diet. Individual response depends on metabolic flexibility (how well the body transitions between fuel sources), HPA axis regulation (how cleanly cortisol follows its circadian pattern), and baseline glucose tolerance.
Some people on long-term keto report that the early-weeks insomnia resolves. The mechanism-level data suggests this may reflect adaptation in arousal threshold or improved metabolic flexibility rather than elimination of the underlying gluconeogenic response to nocturnal cortisol.
Multiple causes often contribute to the same 3am waking pattern. Blood glucose instability from carbohydrate restriction might be one factor alongside cortisol rhythm disruption, caloric deficit, or stress-driven HPA axis changes. A single dietary pattern can engage several of these pathways at once.
Find out which causes might be driving your 3am wakeups
Frequently Asked Questions
Can a Calorie Deficit Cause Insomnia?
Energy restriction raises diurnal cortisol regardless of whether the diet is low-carb, low-fat, or balanced. When a ketogenic diet is also calorie-restricted — which is common, since many people adopt keto as a weight loss tool — the two pathways stack. Cortisol rises from the energy deficit itself, and the glycogen depletion from carbohydrate restriction amplifies the nocturnal gluconeogenic response to that cortisol.
This compounding effect means keto dieters in a caloric deficit face a double mechanism for 3am waking: more cortisol being produced, and a liver more dependent on gluconeogenesis to respond to it. For full coverage of how energy restriction independently fragments sleep, see Can a Calorie Deficit Cause Insomnia?.
Does Intermittent Fasting Cause Sleep Problems?
An early eating window — stopping food intake at 5pm or 6pm — extends the overnight fast to 9-10 hours by 3am, and 13-15 hours by the time of waking. At the longer duration, the Khani and Tayek (2001) data shows gluconeogenesis is already the dominant glucose production pathway. The 3-4am cortisol rise then acts on a liver with less glycogen available, producing the same mechanism described in this article for ketogenic diets — even if the diet is not low-carb.
Combining intermittent fasting with a ketogenic diet amplifies the glycogen depletion further: fewer carbohydrates coming in, plus a longer window in which the liver draws down whatever glycogen remains. For how meal timing affects circadian clock entrainment — a related but distinct mechanism — see the dedicated article on intermittent fasting and sleep.
Does Sugar Cause Nighttime Waking?
Reactive hypoglycemia from a high-sugar evening meal follows a different timeline than keto-related waking. The insulin overshoot happens within 2-4 hours of the meal, placing the glucose nadir in the first half of the night. Keto-related waking concentrates in the 2-4am window because it depends on the circadian cortisol rise, not on a recent meal’s insulin response.
Both mechanisms produce arousal through the same final pathway: blood glucose instability triggers counterregulatory hormones (epinephrine, cortisol, glucagon), and those hormones activate the brain. The dietary triggers are opposite — too much sugar versus too little carbohydrate — but the endocrine response that wakes you is the same. For the full counterregulatory cascade, see Can a Blood Sugar Drop Wake You Up at 3am?.
Related Reading
- Metabolic Sleep Disruption
- Can a Blood Sugar Drop Wake You Up at 3am?
- Why Do You Wake Up Hungry at 3am?
- Why Does Cortisol Spike at 3am and Wake You Up?
- Can a Calorie Deficit Cause Insomnia?
- Adrenal Fatigue and 3am Waking
- Insulin Resistance and Insomnia
- Prediabetes and Sleep Problems
- CGM, Nocturnal Glucose, and Sleep
- Intermittent Fasting, Blood Sugar, and Sleep
- Does Visceral Fat Block Growth Hormone Pulses During Sleep?
- Dawn Phenomenon in Non-Diabetics
- Fatty Liver and Sleep Problems
- Gut Dysbiosis, Blood Sugar, and Sleep
- Late-Night Eating, Blood Sugar, and Sleep
References
Afaghi, A., O’Connor, H., & Chow, C. M. (2008). Acute effects of the very low carbohydrate diet on sleep indices. Nutritional Neuroscience, 11(4), 146-154. https://pubmed.ncbi.nlm.nih.gov/18681982/
DeFronzo, R. A., & Auchus, R. J. (2025). Cushing syndrome, hypercortisolism, and glucose homeostasis: A review. Diabetes, 74(12), 2168-2178. https://pubmed.ncbi.nlm.nih.gov/40663715/
Dinneen, S., Alzaid, A., Miles, J., & Rizza, R. (1995). Effects of the normal nocturnal rise in cortisol on carbohydrate and fat metabolism in IDDM. The American Journal of Physiology, 268(4 Pt 1), E595-603. https://pubmed.ncbi.nlm.nih.gov/7733257/
Khani, S., & Tayek, J. A. (2001). Cortisol increases gluconeogenesis in humans: Its role in the metabolic syndrome. Clinical Science, 101(6), 739-747. https://pubmed.ncbi.nlm.nih.gov/11724664/
Pasca, L., Quaranta, C. A., Grumi, S., Zanaboni, M. P., Tagliabue, A., Guglielmetti, M., Vitali, H., Capriglia, E., Varesio, C., Toni, F., Nobili, L., Terzaghi, M., & De Giorgis, V. (2024). The effects of ketogenic dietary therapies on sleep: A scoping review. Journal of Sleep Research, 33(4), e14073. https://pubmed.ncbi.nlm.nih.gov/37932966/
Perlman, J., Wetmore, E., Lehner-Gulotta, D., Banwell, B., Bergqvist, A. G. C., Coleman, R., Chen, S., Conaway, M., Goldman, M. D., Morse, A. M., & Brenton, J. N. (2024). Impact of a ketogenic diet on sleep quality in people with relapsing multiple sclerosis. Sleep Medicine, 122, 213-220. https://pubmed.ncbi.nlm.nih.gov/39208520/
Xie, M., Li, J., & Bai, W. (2026). Sleep disorders in patients with PCOS after ketogenic diet intervention: An analysis of influencing factors and development of predictive model. British Journal of Hospital Medicine, 87(2), 50655. https://pubmed.ncbi.nlm.nih.gov/41762102/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 7 references cited
