Lion’s mane is one of the fastest-growing functional mushroom supplements, marketed primarily for cognition and nerve regeneration. But a persistent pattern in user reports shows contradictory sleep effects — some people sleep better, others develop insomnia or vivid dreams. Both outcomes have pharmacological explanations.
This article covers lion’s mane’s dual mechanism (neurotrophic vs cholinergic), the human and animal evidence for sleep effects, the REM-specific pathway, and practical timing guidance. It does not cover the full autonomic model of sleep disruption — for that, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It. Cholinergic modulation is one of several autonomic pathways that regulate sleep architecture; the pillar covers the broader picture.
Can Lion’s Mane Cause Insomnia?
The insomnia side of lion’s mane comes down to one mechanism: acetylcholinesterase (AChE) inhibition. AChE is the enzyme responsible for breaking down acetylcholine in the synaptic cleft. When a compound inhibits AChE, acetylcholine levels rise — the same pharmacological principle behind Alzheimer’s drugs like donepezil and galantamine.
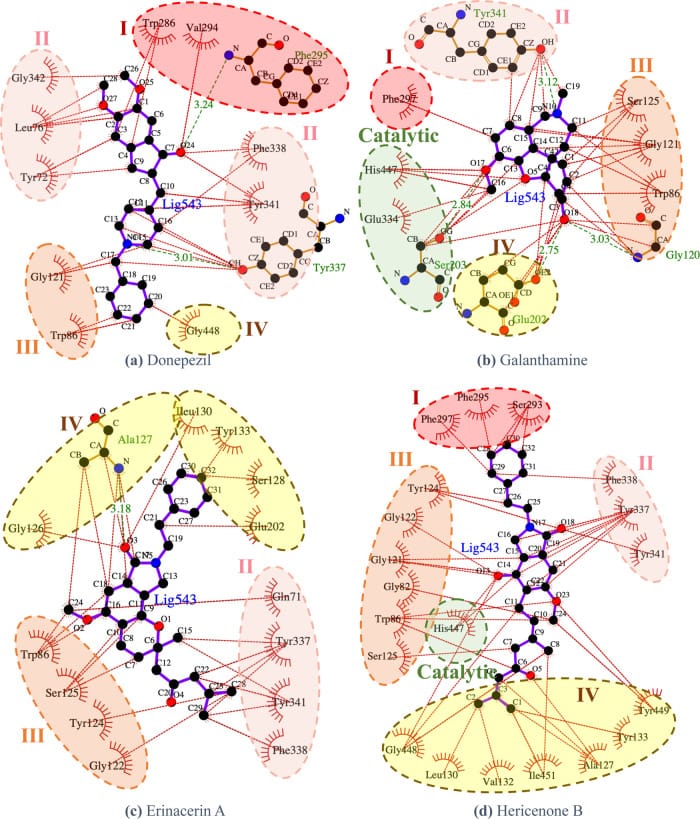
Sutthibutpong et al. (2024) used deep learning and molecular docking to screen lion’s mane compounds for AChE inhibitory potential. Two compounds — erinacerin A and hericenone B — showed binding energy profiles similar to the combined characteristics of donepezil and galantamine at the AChE active site, though with slightly weaker binding free energies (Sutthibutpong et al., 2024). The computational modeling identified these compounds as potential AChE inhibitors with binding characteristics that overlap with FDA-approved cholinesterase inhibitors.
Why does elevated acetylcholine disrupt sleep? Acetylcholine is the neurotransmitter that initiates and sustains REM sleep. Two muscarinic receptor subtypes — M1 (Chrm1) and M3 (Chrm3) — are required for REM generation. Niwa et al. (2018) demonstrated this using CRISPR gene editing: mice lacking both M1 and M3 muscarinic receptors had REM sleep diminished to an almost undetectable level. The study’s triple-target CRISPR method addressed the redundancy challenge inherent in studying whether the cholinergic pathway is essential for REM (Niwa et al., 2018). This is the same receptor pathway involved in anticholinergic drug-induced REM suppression — the Benadryl article covers the opposite direction of that mechanism.
When acetylcholine levels are elevated during the early sleep cycles — when NREM should dominate — the result is fragmented sleep transitions, premature REM intrusions, and difficulty maintaining deep sleep. This is the mechanism behind the insomnia reports.
Dose matters. Li et al. (2021) found that only the high dose of lion’s mane mycelium (150 mg/kg) produced measurable effects on sleep and anxiety in mice. The low dose (75 mg/kg) did not reach statistical significance (Li et al., 2021). This dose-dependence may explain why some users tolerate moderate doses without sleep disruption while others, taking higher amounts, experience insomnia.
Extract type also matters. Mycelium contains erinacines — compounds that cross the blood-brain barrier and are primarily responsible for NGF stimulation. Fruiting body contains hericenones, which have different pharmacokinetic profiles. The AChE inhibitory compounds identified by Sutthibutpong et al. include erinacerin A and hericenone B. Hericenone B is a known fruiting body compound, while erinacerin A’s tissue source was not specified in the computational study — though erinacerins are structurally related to mycelium-derived erinacines. Both extract types may carry cholinergic potential, but the degree and onset may differ.
Why Does Lion’s Mane Help Some People Sleep Better?
The sleep-improvement side of lion’s mane works through a different mechanism than the insomnia side. Where AChE inhibition raises acetylcholine directly, the neurotrophic pathway works through BDNF and NGF — growth factors that support neuronal health, reduce neuroinflammation, and lower anxiety. Sleep improvement appears as a downstream effect of these changes, not as a direct sedative action.
Vigna et al. (2019) conducted an 8-week trial in 77 overweight or obese adults who exhibited mood and sleep disturbances. Participants receiving lion’s mane supplementation showed decreased depression, anxiety, and sleep disorder scores on validated questionnaires, along with increased circulating pro-BDNF levels. Mature BDNF levels did not change at the end of supplementation but decreased significantly during the washout period. The pro-BDNF/BDNF ratio increased at both timepoints (p<0.05), suggesting it may serve as a biomarker for neurotrophin-mediated response. The biomarker analysis included a subset of 10 participants (Vigna et al., 2019). Sleep quality improvements occurred concurrently with mood improvements — consistent with a shared underlying mechanism rather than independent effects.
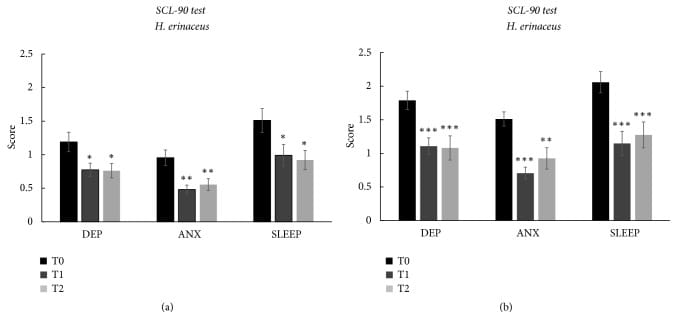
Li et al. (2021) provided supporting evidence in an animal model. Mice subjected to 9 days of stress-induced sleep disruption (via daily tail suspension tests) received lion’s mane mycelium at two doses. The high dose (150 mg/kg) reversed the stress-induced disruption of NREM sleep during the dark (active) period and reduced anxiety behavior in the elevated-plus-maze test. The low dose did not reach statistical significance on either measure (Li et al., 2021). Small group sizes (n = 2-6 per group) limit generalizability, but the convergence of anti-anxiety and pro-sleep effects in a single study suggests a shared neurotrophic mechanism.
A 2025 review by Menon et al. synthesized 26 studies on lion’s mane supplementation and reported evidence for sleep improvement from individual trials, neurogenesis promotion in preclinical models, and BDNF/pro-BDNF increases, though the review noted the evidence base remains limited and heterogeneous. The review also reported a combined weighted mean increase of 1.17 points on the Mini-Mental State Examination (MMSE) in lion’s mane groups within the intervention groups (pre- to post-supplementation) — a modest effect, though the review noted the evidence base is heterogeneous and limited in size (Menon et al., 2025).
The two mechanisms — cholinergic and neurotrophic — are not mutually exclusive. A person can experience BDNF-mediated anxiety reduction (which improves sleep) while simultaneously experiencing AChE-mediated acetylcholine elevation (which can fragment sleep). Which effect dominates depends on individual neurochemistry, dose, and timing.
Does Lion’s Mane Affect Rapid Eye Movement Sleep?
During normal sleep, acetylcholine levels follow a cyclical pattern. They drop during NREM sleep (allowing deep sleep stages to occur) and rise during REM sleep (activating the cortical and thalamic circuits that produce dreaming). This rhythm is regulated by cholinergic neurons in the pedunculopontine tegmental nucleus (PPT) and laterodorsal tegmental nucleus (LDT) — the same brainstem regions that the Niwa 2018 study mapped as essential for REM generation through muscarinic M1 and M3 receptors (Niwa et al., 2018).
When an external compound raises acetylcholine levels — whether through AChE inhibition from lion’s mane or from cholinesterase inhibitors used in Alzheimer’s management — the natural NREM/REM rhythm is disrupted. Acetylcholine that should be low during early NREM-dominant sleep cycles is elevated, promoting premature REM onset and increasing REM intensity.
This is the pharmacological basis for vivid dreams on lion’s mane. More acetylcholine during sleep means more muscarinic receptor activation, which means more REM activity, which means more vivid and memorable dream content. The same mechanism has been documented with AChE inhibitors used for Alzheimer’s management — vivid dreams and sleep disturbance are recognized side effects of donepezil.
The cholinergic brainstem pathway that controls REM is the connection point between lion’s mane and dream changes. Acetylcholine-raising supplements can intensify REM in the same way that acetylcholine-lowering drugs (like diphenhydramine) suppress it. The vivid dream mechanism from cholinergic supplements covers this pathway in full detail.
The timing dimension is important. Taking lion’s mane in the late afternoon or evening means that AChE inhibition is active during the first sleep cycles — when NREM should dominate. The elevated acetylcholine during those early cycles can delay deep sleep onset and fragment the NREM-to-REM transitions that maintain sleep continuity.
Should You Take Lion’s Mane in the Morning or at Night?
The pharmacokinetic reasoning follows directly from the mechanism. AChE inhibition from lion’s mane raises acetylcholine levels within hours of ingestion. If cholinergic activity peaks during the daytime — when the brain benefits from increased acetylcholine for attention, memory, and cognitive performance — and returns toward baseline by evening, the sleep-disrupting effects are minimized. This is the same timing principle applied to AChE inhibitors used in Alzheimer’s management — donepezil, for example, is sometimes switched to morning dosing when sleep disruption occurs.
Docherty et al. (2023) demonstrated that lion’s mane produces measurable acute cognitive effects within 60 minutes of ingestion — improved Stroop task reaction time (p = 0.005) in healthy young adults (Docherty et al., 2023). This indicates that cholinergic activity begins rapidly after dosing, reinforcing the rationale for morning timing if sleep protection is the goal.
Mori et al. (2009), in their landmark trial of lion’s mane for mild cognitive impairment, used a dosing schedule of four 250 mg tablets taken three times daily — distributed throughout the day rather than concentrated in a single evening dose. The study reported cognitive improvement over 16 weeks with no notable adverse sleep effects (Mori et al., 2009). While the study did not compare morning vs evening dosing directly, the distributed daytime schedule avoided the concentrated evening cholinergic spike that evening-only dosing would produce.
Individual variation plays a role. Baseline cholinergic tone differs between people — someone with already elevated acetylcholine activity may be more sensitive to additional AChE inhibition than someone with lower baseline levels. Concurrent supplement use matters: stacking lion’s mane with other cholinergic compounds (alpha-GPC, CDP-choline, huperzine A) amplifies the acetylcholine-raising effect and increases sleep disruption risk. A neuroscience-based approach to nootropic stacking without compromising sleep covers timing and stacking guidance for protecting sleep.
Extract standardization is another variable. Standardized extracts with verified compound concentrations produce more predictable effects than bulk powder, where the amount of AChE-inhibitory compounds can vary between batches and brands. This inconsistency may partially explain why some people report sleep disruption from one product but not another at the same stated dose.
Cholinergic modulation from lion’s mane might not be the only factor affecting your sleep. Autonomic hyperarousal, GABA receptor changes, metabolic disruptions, or inflammatory processes might also be contributing. When multiple causes overlap, identifying which ones might be active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Lion’s Mane Make You Sleepy or Give You Energy?
The confusion comes from categorizing lion’s mane as either calming or energizing — it is neither in the way that melatonin is calming or caffeine is energizing. Lion’s mane acts on two pathways (neurotrophic and cholinergic), and the net effect depends on which pathway is more relevant to your baseline state. In Vigna et al. (2019), participants with anxiety and mood disturbances experienced calming effects, consistent with BDNF-mediated neuronal support. Docherty et al. (2023) found acute cognitive performance improvements in healthy young adults — an alerting effect, consistent with increased cholinergic transmission. Same supplement, different outcomes, different populations.
Is Lion’s Mane Good for Sleep Anxiety?
Li et al. (2021) showed that high-dose lion’s mane mycelium (150 mg/kg) reduced anxiety behavior in sleep-deprived mice using the elevated-plus-maze test and reversed stress-induced NREM disruption (Li et al., 2021). In humans, Vigna et al. (2019) documented parallel improvements in anxiety scores and sleep quality scores over 8 weeks of supplementation, with corresponding increases in circulating pro-BDNF (Vigna et al., 2019). The proposed mechanism — neurotrophin-mediated anxiety reduction leading to improved sleep — is distinct from GABAergic sedation. Lion’s mane does not appear to act as a direct anxiolytic in the pharmacological sense; the anxiety reduction is downstream of neurotrophic support.
Does It Matter Whether You Take Lion’s Mane Fruiting Body or Mycelium?
Spangenberg et al. (2025) conducted a review focused on erinacines — the diterpenoid compounds found exclusively in lion’s mane mycelium — and found that erinacines A and C accumulate the transcription factor Nrf2, a master regulator of antioxidant defense, while promoting hippocampal neurogenesis and reducing neuroinflammation across animal models (Spangenberg et al., 2025). These effects are dose-dependent. While erinacines are unique to mycelium, the review noted that both fruiting body and mycelium preparations showed neuromodulatory activity — erinacines alone do not account for all of the observed neuroprotective effects. If the neurotrophic (BDNF/NGF) pathway is the primary goal, mycelium-based extracts may have an advantage based on the erinacine evidence. If minimizing cholinergic sleep disruption is the priority, the compound profile of each product needs evaluation — both extract types contain AChE-inhibitory compounds, but in different concentrations.
Can You Take Lion’s Mane With Other Nootropics Without Affecting Sleep?
Each cholinergic compound in a stack adds to the total acetylcholine load. Lion’s mane inhibits AChE (reducing acetylcholine breakdown). Alpha-GPC and CDP-choline provide additional choline substrate for acetylcholine synthesis. Huperzine A is another AChE inhibitor. Taken together, these compounds can produce a cumulative cholinergic effect that exceeds what any single compound would produce alone. How different choline forms cross the blood-brain barrier and affect sleep compares the sleep impact of individual choline sources. A timing and stacking approach for protecting sleep while using nootropics provides practical stacking guidance.
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Why Do Cholinergic Supplements Give You Vivid Dreams? — the acetylcholine-REM pathway behind supplement-related vivid dreams
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — how post-viral autonomic disruption can affect sleep continuity
- Why Does Your Heart Race at Night After COVID? What the Vagus Nerve Has to Do With It — why post-COVID autonomic instability can show up as nighttime tachycardia
- Can POTS and Dysautonomia Cause Insomnia? Why Your Autonomic Nervous System Will Not Let You Rest — how orthostatic intolerance and autonomic instability can fragment sleep
- Can a Viral Infection Cause Chronic Insomnia? — how viral infections can disrupt autonomic regulation and sleep
- Why Will Your Nervous System Not Let You Sleep? Polyvagal Theory and Insomnia — how polyvagal states map to insomnia, hyperarousal, and unrefreshing sleep
- Why Does Trauma Cause Insomnia? How Your Nervous System Stays on Guard at Night — how trauma can keep sleep shaped by hyperarousal and vigilance
- Why Are You Sleeping 12 Hours and Still Exhausted? The Dorsal Vagal Shutdown Explained — the dorsal vagal pattern behind long sleep that still feels unrefreshing
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- How Do Probiotics Reach Your Brain? The Vagotomy Evidence — what vagotomy studies show about gut-brain communication
- Does Poor Sleep Damage Your Gut Microbiome? — the relationship between sleep fragmentation, microbiome disruption, and inflammatory load
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
Li, T. J., Lee, T. Y., Lo, Y., Lee, L. Y., Li, I. C., Chen, C. C., & Chang, F. C. (2021). Hericium erinaceus mycelium ameliorate anxiety induced by continuous sleep disturbance in vivo. BMC Complementary Medicine and Therapies, 21(1), 295. https://pubmed.ncbi.nlm.nih.gov/34865649/
Menon, A., Jalal, A., Arshad, Z., Nawaz, F. A., & Kashyap, R. (2025). Benefits, side effects, and uses of Hericium erinaceus as a supplement: a systematic review. Frontiers in Nutrition, 12, 1641246. https://pubmed.ncbi.nlm.nih.gov/40959699/
Mori, K., Inatomi, S., Ouchi, K., Azumi, Y., & Tuchida, T. (2009). Improving effects of the mushroom Yamabushitake (Hericium erinaceus) on mild cognitive impairment: a double-blind placebo-controlled clinical trial. Phytotherapy Research, 23(3), 367-372. https://pubmed.ncbi.nlm.nih.gov/18844328/
Niwa, Y., Kanda, G. N., Yamada, R. G., Shi, S., Sunagawa, G. A., Ukai-Tadenuma, M., Fujishima, H., Matsumoto, N., Masumoto, K. H., Nagano, M., Kasukawa, T., Galloway, J., Perrin, D., Shigeyoshi, Y., Ukai, H., Kiyonari, H., Sumiyama, K., & Ueda, H. R. (2018). Muscarinic acetylcholine receptors Chrm1 and Chrm3 are essential for REM sleep. Cell Reports, 24(9), 2231-2247.e7. https://pubmed.ncbi.nlm.nih.gov/30157420/
Docherty, S., Doughty, F. L., & Smith, E. F. (2023). The Acute and Chronic Effects of Lion’s Mane Mushroom Supplementation on Cognitive Function, Stress and Mood in Young Adults: A Double-Blind, Parallel Groups, Pilot Study. Nutrients, 15(22), 4842. https://pubmed.ncbi.nlm.nih.gov/38004235/
Spangenberg, E. T., Moneypenny, A., Bozzo, G. G., & Perreault, M. L. (2025). Unveiling the role of erinacines in the neuroprotective effects of Hericium erinaceus: a systematic review in preclinical models. Frontiers in Pharmacology, 16, 1582081. https://pubmed.ncbi.nlm.nih.gov/40626304/
Sutthibutpong, T., Posansee, K., Liangruksa, M., Termsaithong, T., Piyayotai, S., Phitsuwan, P., Saparpakorn, P., Hannongbua, S., & Laomettachit, T. (2024). Combining Deep Learning and Structural Modeling to Identify Potential Acetylcholinesterase Inhibitors from Hericium erinaceus. ACS Omega, 9(14), 16311-16321. https://pubmed.ncbi.nlm.nih.gov/38617639/
Vigna, L., Morelli, F., Agnelli, G. M., Napolitano, F., Ratto, D., Occhinegro, A., Di Iorio, C., Savino, E., Girometta, C., Brandalise, F., & Rossi, P. (2019). Hericium erinaceus Improves Mood and Sleep Disorders in Patients Affected by Overweight or Obesity: Could Circulating Pro-BDNF and BDNF Be Potential Biomarkers?. Evidence-Based Complementary and Alternative Medicine, 2019, 7861297. https://pubmed.ncbi.nlm.nih.gov/31118969/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 7 references cited
