The term “adrenal fatigue” persists because the experience it describes is documented: waking between 2am and 4am, difficulty returning to sleep, daytime exhaustion that coffee does not resolve, and a feeling that the body’s stress response is no longer working the way it used to. Millions of people search for this term because it matches what they feel.
But the label points to the wrong organ. The adrenal glands do not fatigue from overuse. What the research documents is a disruption in HPA axis regulation — the feedback loop between the hypothalamus, pituitary gland, and adrenal cortex that governs cortisol output across the 24-hour cycle. That disruption can move in two directions, and which direction matters for how you investigate it.
Understanding what the HPA axis is doing during the hours around 3am matters beyond sleep quality. Chronic cortisol dysregulation in either direction — whether cortisol stays too high at night or does not rise adequately in the morning — is associated with measurable downstream effects on metabolic and cognitive health over time. This article covers what the research shows about the HPA axis and 3am waking: the measurable mechanism behind what people call “adrenal fatigue.”
For the full picture of how metabolic processes fragment sleep, see Metabolic Sleep Disruption: How Blood Sugar, Cortisol, and Metabolic Rate Affect Sleep.
What Is the HPA Axis and How Does It Relate to 3am Waking?
Cortisol follows a predictable daily pattern: it reaches its lowest point between roughly 12am and 3am, then begins rising in the second half of the night, peaking around 30 minutes after waking. This rhythm is tightly coupled to sleep architecture.
Buckley and Schatzberg (2005) found that the early-night sleep period dominated by slow-wave sleep (SWS) is the only segment of the 24-hour cycle during which HPA axis activity undergoes sustained suppression, producing minimum circulating concentrations of both ACTH and cortisol. This suppression is not passive — SWS actively inhibits cortisol release. When that deep sleep is reduced or fragmented, the inhibition lifts, and cortisol remains higher than it should be during the hours when the brain and body are meant to be in recovery.
Rodenbeck et al. (2002) directly measured hourly plasma cortisol throughout the evening and night in people with chronic insomnia and matched healthy controls. The insomnia group showed elevated evening and nocturnal cortisol concentrations across the measurement window. A positive correlation emerged between pre-bedtime cortisol levels and the number of nocturnal awakenings — not only in the insomnia group, but also in healthy controls. The relationship was amplified in insomnia, but present in both groups.
Cortisol release during the first four hours of sleep correlated with wakefulness within the sleep period in the insomnia group. This timing implicates early-night HPA activity as a driver of the fragmentation pattern — including the 2-4am awakenings that people describe when they search for “adrenal fatigue.”
The relationship between cortisol and sleep fragmentation is self-perpetuating. Elevated cortisol during early sleep reduces the depth of slow-wave sleep. Reduced slow-wave sleep removes the only period of active HPA suppression. Without that suppression, cortisol stays elevated. The same night that begins with higher-than-normal cortisol produces the conditions for the next night to follow the same pattern.
Does Sleep Fragmentation Elevate Cortisol, or Does Elevated Cortisol Fragment Sleep?
Directionality — does the cortisol come first, or the sleep disruption? — is one of the reasons the “adrenal fatigue” label persists. If the adrenals are “fatigued,” the implication is that stress came first and the glands stopped producing adequate cortisol. The research shows something more complex: the relationship is bidirectional, and either end — stress or sleep disruption — can initiate the cortisol-sleep feedback loop.
Cohn et al. (2023) isolated the causal direction in a controlled crossover experiment. Twenty-two healthy premenopausal women underwent sleep fragmentation (via auditory tones) and unfragmented sleep in counterbalanced order. Sleep fragmentation alone — without any psychological stress, without sleep deprivation, without hormonal changes — raised bedtime cortisol by 27% (p = .03). The same fragmented nights reduced the cortisol awakening response (CAR) by 57% (p = .01).
What this produced was a recognizable cortisol profile: elevated at night when it should be low, and blunted in the morning when it should spike. This inverted pattern — high-at-night, flat-in-the-morning — is the cortisol signature associated with cardiovascular risk, impaired glucose tolerance, and immune impairment in the endocrine literature.
Polysomnographic data supported this. Wake after sleep onset (WASO) — the total minutes spent awake after initially falling asleep — correlated positively with elevated bedtime cortisol (p = .047). More fragmentation, more cortisol. More cortisol, more fragmentation.

Chen et al. (2024) added a methodological point through a meta-analysis of 24 studies examining acute sleep deprivation and cortisol. The overall effect across studies was null (SMD = 0.18, p = 0.208) — but this headline number is misleading. When only serum-based cortisol measurements were analyzed, the effect became statistically detectable (SMD = 0.46, p = 0.011). And studies that measured cortisol at multiple time points across the day found the effect (p = 0.027), while single-timepoint studies did not (p = 0.777).
The implication: the cortisol elevation from poor sleep is distributed across the day rather than concentrated at one time point. A single morning cortisol draw can miss it. The disruption is measurable — but detecting it requires looking at the full daily cortisol curve, not one snapshot.
This bidirectional relationship between cortisol and sleep fragmentation explains why “adrenal fatigue” feels so persistent. A period of high stress can initiate the cortisol-sleep cycle by elevating nocturnal cortisol. That elevated cortisol fragments sleep. The fragmented sleep sustains the cortisol elevation independent of the original stressor. The stressor can resolve while the sleep-cortisol cycle continues running.
What Does Elevated Cortisol Feel Like Versus Blunted Cortisol?
“Adrenal fatigue” as a term collapses two distinct HPA axis states into one label. In practice, the people searching for this term are experiencing one of two patterns — and the distinction matters because the underlying physiology points in different directions.
Hyperactivation pattern: Cortisol is elevated when it should be low — particularly in the late evening and early-morning hours. This is the pattern documented by Rodenbeck et al. (2002) and Buckley and Schatzberg (2005). The experience: waking between 2am and 4am with a sense of alertness or mental activation, racing or looping thoughts, difficulty returning to sleep, and a feeling of being “wired” despite being tired. Morning energy may be adequate initially but crashes by midday.
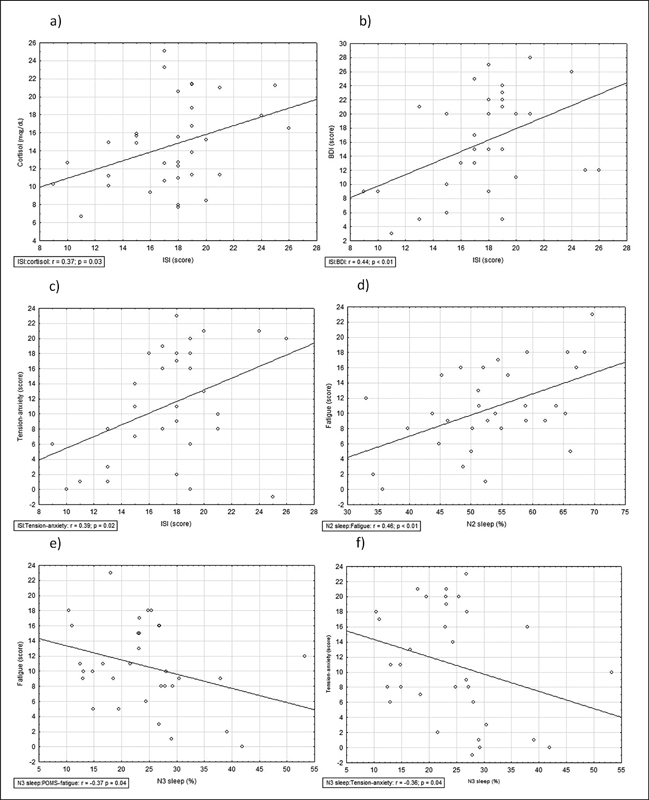
Passos et al. (2023) measured this in 34 adults with chronic insomnia. Insomnia severity (measured by the Insomnia Severity Index) correlated positively with morning cortisol (r = 0.37, p = 0.03). People with more severe insomnia had higher cortisol at waking. The same study found that reduced slow-wave sleep (N3) correlated with increased tension-anxiety (r = -0.36, p = 0.04) and fatigue (r = -0.37, p = 0.04). More time spent in lighter N2 sleep — at the expense of deep sleep — was associated with greater fatigue (r = 0.46, p < 0.01).
HPA axis output and daytime psychological state were connected: insomnia severity correlated with depression scores (r = 0.44, p < 0.01) and tension-anxiety (r = 0.39, p = 0.02). Morning cortisol may be part of what mediates the anxiety and low mood that accompany chronic insomnia — the HPA axis does not reset overnight when it cannot access sufficient deep sleep.
Hypoactivation pattern: Cortisol output is blunted — the morning cortisol rise is suppressed, and the daily rhythm is flat. This is the pattern documented by Cohn et al. (2023), where sleep fragmentation reduced the cortisol awakening response by 57%. The experience: heavy fatigue upon waking, difficulty getting going in the morning, unrefreshing sleep even after 7-8 hours in bed, and tiredness throughout the day. The 3am waking may still occur, but without the sharp mental activation — the person wakes and feels drained rather than alert.
The hyperactivation and hypoactivation states can coexist over time. The Cohn data showed that the same fragmented sleep nights that raised bedtime cortisol by 27% also blunted the morning cortisol awakening response by 57%. One person can have elevated cortisol at night (producing 3am waking) and blunted cortisol in the morning (producing difficulty waking and daytime fatigue). This high-at-night, low-in-the-morning combination is the flattened diurnal cortisol profile that carries the broadest health consequences.
Why does this distinction matter? Because the experience of “adrenal fatigue” is not uniform. Someone waking at 3am with racing thoughts is in a different physiological state than someone who sleeps through the night but wakes exhausted. Both may describe their experience as “adrenal fatigue,” but what the HPA axis is doing in each case is different.
Is Adrenal Fatigue a Recognized Condition?
As a concept, “adrenal fatigue” proposes that prolonged stress exhausts the adrenal glands, reducing their capacity to produce cortisol. No published research supports this mechanism. The adrenal glands maintain cortisol production capacity throughout life, and cortisol output in stressed individuals is typically elevated, not reduced (Buckley & Schatzberg, 2005).
What is measurable is HPA axis dysregulation — a disruption in the timing, amplitude, or feedback sensitivity of the cortisol rhythm. This can produce either elevated nocturnal cortisol (hyperactivation) or a blunted cortisol awakening response (hypoactivation), or both at the same time.
The experiences people attribute to “adrenal fatigue” — fatigue, sleep disruption, difficulty recovering from stress — are documented in the research under HPA axis dysregulation. The label is inaccurate, but the underlying disruption is measurable.
Can You Recover from HPA Axis Dysregulation?
Cohn et al. (2023) showed causation in one direction: sleep fragmentation produced the cortisol disruption. When fragmentation was removed (the unfragmented sleep condition in the same participants), bedtime cortisol and the cortisol awakening response were within normal ranges.
The HPA axis disruption reversed when the sleep disruption resolved. The cortisol rhythm responded to changes in sleep quality — it was not permanently altered by a period of fragmentation or stress.
Buckley and Schatzberg (2005) identified slow-wave sleep as the sleep stage that suppresses HPA axis activity. This means that approaches directed at increasing the depth and duration of slow-wave sleep have a plausible mechanism for normalizing cortisol. Sleep architecture responds to changes in stress load, exercise timing, light exposure, temperature, and hormonal status.
Because fragmented sleep sustains cortisol elevation and cortisol elevation sustains fragmented sleep, breaking the cortisol-fragmentation cycle at either point can resolve both sides. Reducing cortisol through stress management can improve sleep depth. Improving sleep depth can reduce cortisol. Both entry points have evidence behind them.
What Organ Is Active at 3am According to Traditional Chinese Medicine?
In TCM, the body clock assigns each two-hour window to an organ, and the 1-3am period corresponds to the liver. People who wake consistently during this window often encounter this framework and wonder whether the two perspectives connect.
The timing observation is accurate: cortisol does reach its lowest circulating concentration between approximately 1am and 3am, and the subsequent rise coincides with the period when waking becomes more likely if HPA axis regulation is disrupted. The research perspective attributes this to the cortisol circadian rhythm and its relationship to sleep architecture — not to liver-based energy cycling.
Whether the TCM observation and the cortisol rhythm describe different aspects of the same phenomenon or are coincidental in timing is beyond what current research can resolve. What is documented is that the 1-3am cortisol nadir and the subsequent pre-dawn cortisol rise are the period when HPA axis dysregulation produces its noticeable sleep disruption.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the full metabolic cause overview
- Can a Blood Sugar Drop Wake You Up at 3am? — how nocturnal blood sugar drops trigger the hormonal cascade that wakes you up
- Why Do You Wake Up Hungry at 3am? — how leptin, ghrelin, and blood sugar interact during sleep
- Why Does Cortisol Spike at 3am and Wake You Up? — the cortisol-sleep feedback loop and HPA axis timing
- Can a Calorie Deficit Cause Insomnia? — how energy restriction elevates cortisol and fragments sleep
- Does Insulin Resistance Affect Sleep Quality? — the bidirectional relationship between insulin sensitivity and deep sleep
- Can Prediabetes Cause Sleep Problems? — how prediabetes changes sleep architecture and accelerates metabolic decline
Kat Fu, M.S., M.S. | The Longevity Vault
References
Buckley, T. M., & Schatzberg, A. F. (2005). On the interactions of the hypothalamic-pituitary-adrenal (HPA) axis and sleep: Normal HPA axis activity and circadian rhythm, exemplary sleep disorders. Journal of Clinical Endocrinology & Metabolism, 90(5), 3106-3114. https://pubmed.ncbi.nlm.nih.gov/15728214/
Chen, Y., et al. (2024). The effect of acute sleep deprivation on cortisol level: A systematic review and meta-analysis. Endocrine Journal, 71(8), 767-777. https://pubmed.ncbi.nlm.nih.gov/38777757/
Cohn, A. Y., et al. (2023). Effects of sleep fragmentation and estradiol decline on cortisol in a human experimental model of menopause. Journal of Clinical Endocrinology & Metabolism, 108(6), e1347-e1355. https://pubmed.ncbi.nlm.nih.gov/37207451/
Passos, G. S., et al. (2023). Insomnia severity is associated with morning cortisol and psychological health. Sleep Science, 16(1), e50-e56. https://pubmed.ncbi.nlm.nih.gov/37151768/
Rodenbeck, A., et al. (2002). Interactions between evening and nocturnal cortisol secretion and sleep parameters in patients with severe chronic primary insomnia. Neuroscience Letters, 324(2), 159-163. https://pubmed.ncbi.nlm.nih.gov/11988351/
