Nearly every search result on the dawn phenomenon addresses it as a diabetes management problem. That leaves healthy adults — people who notice their morning blood sugar is higher than their bedtime reading, who wake between 4am and 6am without an alarm, or who see an unexplained pre-dawn rise on a consumer CGM — with no explanation of what is happening.
The dawn phenomenon is normal physiology that becomes a problem on a spectrum. It does not begin at a diabetes label. When counter-regulatory hormones are dysregulated — whether from chronic stress, poor sleep quality, or early-stage metabolic decline — the same circadian mechanism that should prepare you for the day instead fragments sleep and destabilizes fasting glucose. In a prospective cohort of 5,542 participants, a severe dawn phenomenon independently predicted 30% higher all-cause mortality regardless of average blood sugar control (Cai et al., 2024).
This article covers the hormonal mechanism behind the dawn phenomenon in non-diabetic adults, what separates it from the Somogyi effect, and why the phenomenon weakens with age. For the broader metabolic sleep disruption picture, see Metabolic Sleep Disruption: The Full Picture.
Does the Dawn Phenomenon Happen in People Without Diabetes?
Bolli et al. (1984) studied eight healthy non-diabetic volunteers at 30-minute intervals from 1am to 9am. After 5:30am, plasma glucose, insulin, and C-peptide all rose (p < 0.05), alongside increased rates of both glucose production and glucose utilization. Cortisol, epinephrine, and norepinephrine rose between 4am and 6:30am. The authors concluded that a dawn-like phenomenon — initiated by increased endogenous glucose production — occurs in non-diabetic individuals as normal circadian physiology, not pathology.
The diabetic dawn phenomenon is an exaggerated version of the same mechanism. What changes across the glycemic spectrum is the beta cell’s ability to compensate with sufficient insulin. In a healthy person, insulin output ramps up to match the cortisol-driven glucose release. When beta cell function declines or insulin resistance worsens, that compensation falls short and the morning glucose reading climbs higher.
Rosenthal and Argoud (1989) documented the non-diabetic dawn phenomenon in a comparison of young (mean age 26) and older (mean age 69) non-diabetic men. Young men showed a glucose rise from 89 to 92 mg/dL (p < 0.05) with increased glucose production rates. Older men showed no rise as a group — and the reason was measurable: their nocturnal growth hormone levels were lower (6.7 vs 10.1 ng/mL in the younger group, p < 0.05).
This age-GH relationship is the clearest evidence that the dawn phenomenon in healthy adults depends on growth hormone. What changes with age is not the mechanism but the hormonal amplitude driving it.
What Hormones Cause Blood Sugar to Rise Before Dawn?
Growth hormone is the dominant driver of the dawn phenomenon in younger adults. Campbell et al. (1985) demonstrated in people with insulin-dependent diabetes that nocturnal GH surges increase hepatic glucose production by 65% and reduce glucose clearance by 50%. When GH was suppressed with somatostatin (a hormone that blocks GH release), the dawn phenomenon disappeared. When exogenous GH was infused to restore the surge, the dawn phenomenon returned. Plasma glucose correlated with peak GH concentration (r = 0.58, p < 0.01), supporting a dose-response relationship. GH acts through two routes at once: it stimulates the liver to produce glucose and it reduces glucose uptake in muscle.
Cortisol adds a second dimension. Van Cauter et al. (1991) measured glucose in eight healthy men across nocturnal sleep, wakefulness, and daytime sleep conditions. During nocturnal sleep, plasma glucose rose by 31 +/- 5% and insulin secretion increased by 60 +/- 11%. Insulin secretion varied inversely with the cortisol rhythm — as cortisol rises in the early morning hours, insulin’s effectiveness is suppressed, and the liver releases more glucose. Even during total sleep deprivation, a 17% glucose rise still occurred, demonstrating that circadian cortisol drives part of the morning elevation regardless of sleep state.
Glucagon acts on a faster timescale. Hansen et al. (2025) established that glucagon acutely stimulates hepatic gluconeogenesis — producing a 3.6-fold rise in hepatic glucose output within minutes at 10 nM concentrations. The acute effect occurred independently of hepatic glycogen — glucagon stimulated gluconeogenesis even in glycogen-depleted livers. However, the effect was absent in overnight-fasted mice, suggesting that the availability of gluconeogenic substrates (rather than glycogen per se) determines the magnitude of glucagon’s acute action. Glucagon also amplified substrate-driven gluconeogenesis by 36-43% when lactate or pyruvate was present, showing that glucagon acts additively with cortisol and GH rather than as a substitute for either.
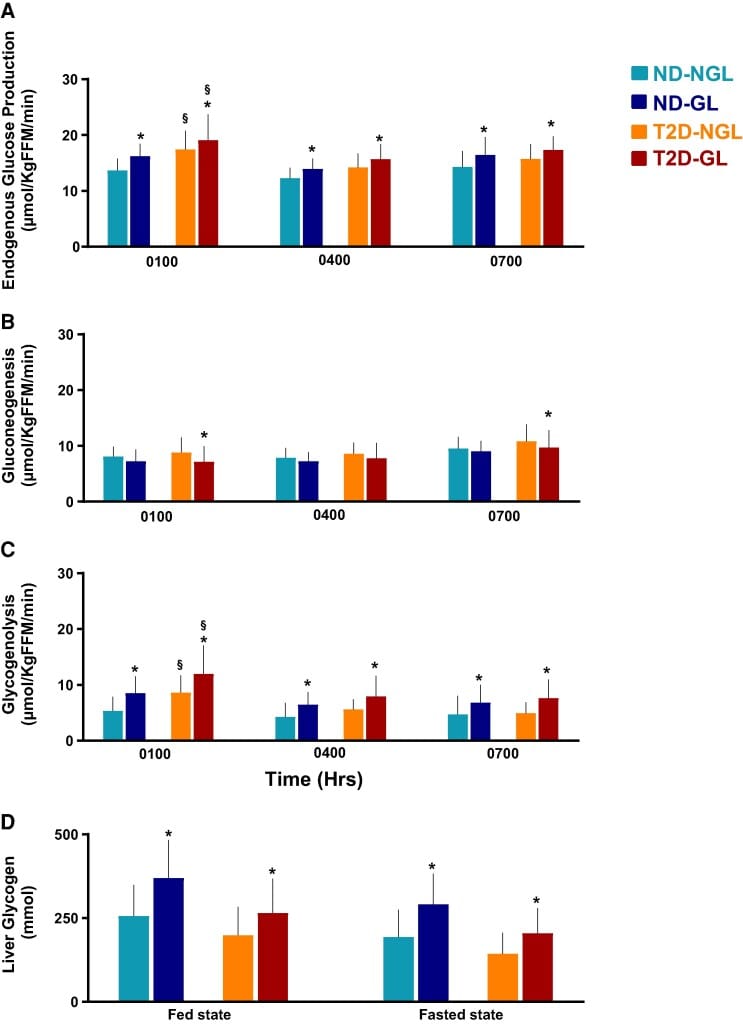
The substrate side matters too. Unni et al. (2025) used stable isotope tracer studies in both non-diabetic controls and people with type 2 diabetes to measure overnight hepatic glucose production directly. Non-diabetic participants maintained approximately 50% gluconeogenesis contribution to overnight glucose production throughout the overnight period. Both groups showed elevated overnight glucose production following high-carbohydrate meals, indicating that dietary glycogen loading the prior evening raises the substrate available for the pre-dawn counter-regulatory surge.
Does Cortisol Drive the Dawn Phenomenon on Its Own?
Atiea et al. (1990) tested this directly using a crossover design. On control nights, glucose rose from 8.4 mmol/L at 4am to 9.4 mmol/L at 8am (p < 0.01). After cortisol suppression with metyrapone (a drug that blocks cortisol production), glucose instead fell from 9.0 to 7.7 mmol/L over the same window (p < 0.05) — a complete reversal. Mean overnight cortisol dropped from 167.2 to 55.9 nmol/L with metyrapone (p < 0.001). No changes in insulin, C-peptide, glucagon, growth hormone, or catecholamines were observed during cortisol suppression — isolating cortisol as the specific causal agent rather than a downstream effect of other hormone changes.
For non-diabetic adults, the implication is direct: chronic elevation of cortisol from poor sleep quality, psychological stress, or circadian misalignment would amplify the pre-dawn glucose rise through the same mechanism Atiea et al. demonstrated pharmacologically.
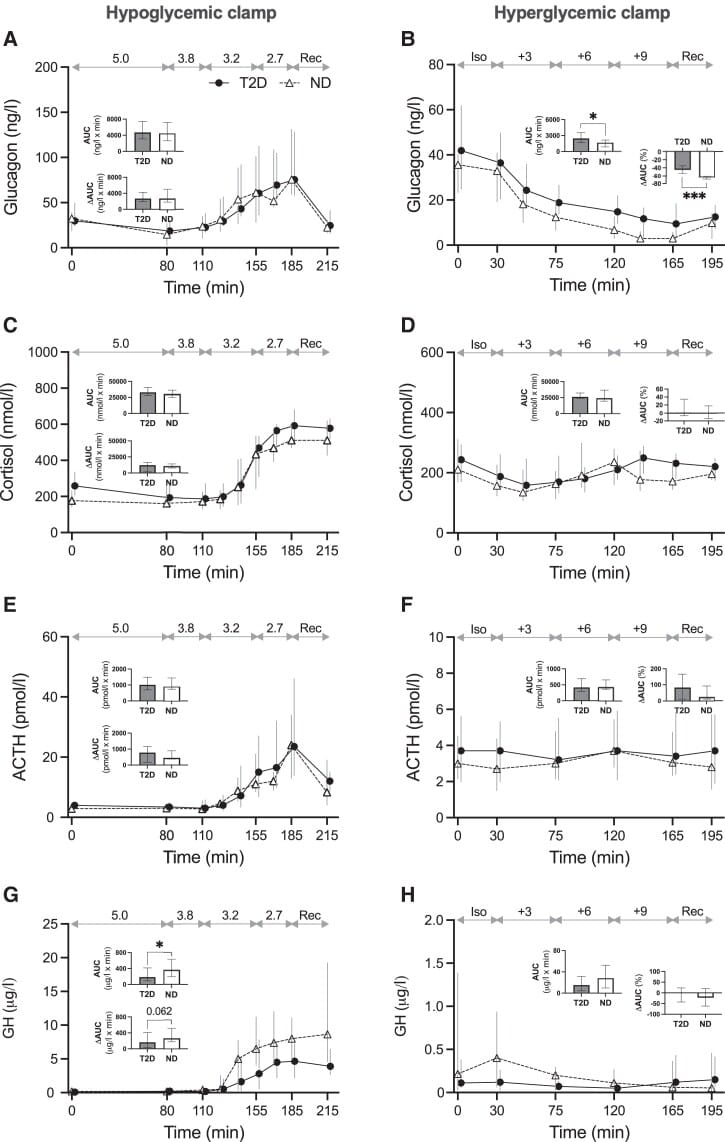
Lundqvist et al. (2024) extended this across the glycemic spectrum using hyperinsulinemic clamp studies in 54 participants (normoglycemic controls, people with prediabetes, and people with type 2 diabetes). Insulin resistance was the strongest predictor of elevated cortisol and ACTH counter-regulatory responses during glucose perturbation. Prediabetic participants showed enhanced ACTH responses (p < 0.05), meaning counter-regulatory cortisol drive increases before a diabetes label is applied. The dawn phenomenon worsens along the glycemic spectrum not because the mechanism changes, but because insulin resistance amplifies cortisol's hepatic glucose effect while blunting the compensatory insulin response.
Many people have more than one cause contributing to their sleep disruption. Cortisol-driven glucose elevation may compound with blood sugar instability, circadian misalignment, or hormonal changes. Identifying which causes may be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Does the Dawn Phenomenon Cause Early Morning Waking?
Van Cauter et al. (1991) established that the glucose rise during sleep is partly sleep-dependent and partly circadian. A 16% glucose rise occurred during daytime sleep, and a 17% rise occurred during total sleep deprivation, demonstrating that the hormonal pattern fires regardless of behavioral sleep state. The circadian component means the hormones rise on schedule even when the body is already awake, producing the familiar experience of early morning alertness that is difficult to override.
The sleep-glucose relationship runs in both directions. Ng et al. (2025) studied 119 healthy non-diabetic university students using CGM and Oura Ring tracking over 14 days. Within-normal-range sleep variation — including a prespecified 1.73-hour sleep restriction — did not affect next-morning glucose in healthy young adults. This matters for interpretation: in metabolically healthy individuals, the dawn glucose rise is driven by endogenous hormonal rhythm rather than night-to-night sleep quality variation. When the dawn phenomenon is exaggerated — producing early waking or unusually high morning readings — the exaggeration points to counter-regulatory dysregulation rather than poor sleep on a given evening.
The long-term relevance is established. Cai et al. (2024) documented in 5,542 participants that a severe dawn phenomenon (glucose increment exceeding 5.55 mmol/L from overnight nadir to pre-breakfast reading) predicted 30% higher all-cause mortality independent of HbA1c. The dawn phenomenon magnitude carries prognostic information beyond average glycemic control. For non-diabetic adults, this means an amplified pre-dawn glucose rise is relevant to track — even when standard blood work returns normal.
Frequently Asked Questions
What Is the Difference Between the Dawn Phenomenon and the Somogyi Effect?
Only a continuous glucose monitor — or fingerstick testing at 2-3am — can reliably distinguish the two, because the Somogyi effect requires evidence of a preceding overnight low.
In non-diabetic adults, the Somogyi effect is less common because endogenous insulin regulation prevents glucose from dropping enough to trigger a large rebound. Consumer CGM data has made the distinction accessible: a dawn phenomenon trace shows a smooth upward curve beginning around 4-5am from a stable overnight baseline; a Somogyi pattern shows a V-shape — a dip below 70 mg/dL followed by a sharp rebound that may exceed the bedtime reading. A true dawn phenomenon responds to earlier breakfast timing and evening carbohydrate composition, while a Somogyi rebound requires addressing the overnight low itself.
Does the Dawn Phenomenon Get Better or Worse With Age?
Rosenthal and Argoud (1989) measured this directly: young men (mean age 26) showed a glucose rise from 89 to 92 mg/dL (p < 0.05) with increased glucose production rates; older men (mean age 69) showed neither, with GH levels at 6.7 vs 10.1 ng/mL (p < 0.05). The practical implication is that a robust dawn phenomenon in a younger adult reflects normal physiology. The same magnitude in an older adult may indicate elevated counter-regulatory hormone activity beyond what GH alone explains — cortisol and stress pathways becoming proportionally more dominant as GH declines. Maintaining metabolic flexibility and cortisol regulation matters more, not less, as the GH buffer decreases with age.
What Should Your Blood Sugar Be When You Wake Up Without Diabetes?
The waking glucose reading is not a true fasting baseline — it is the tail end of the dawn phenomenon rise. A reading of 90 mg/dL at 7am may have come from a 75 mg/dL trough at 3am with a 15 mg/dL cortisol-driven rise, which is a standard pattern. A reading of 105 mg/dL may reflect a larger rise from the same trough due to amplified cortisol or reduced insulin sensitivity.
Lundqvist et al. (2024) showed with clamp data that counter-regulatory cortisol responses are already enhanced in prediabetes, before fasting glucose exceeds the standard threshold. Some individuals will see dawn phenomenon amplification before any laboratory value flags an abnormality — a gap that consumer CGM is beginning to expose.
Related Reading
- Metabolic Sleep Disruption: The Full Picture
- Can a Blood Sugar Drop Wake You Up at 3am?
- Why Do You Wake Up at 3am Hungry?
- Why Does Cortisol Spike at 3am and Wake You Up?
- Can a Calorie Deficit Cause Insomnia?
- Adrenal Fatigue and 3am Waking
- Insulin Resistance and Insomnia
- Prediabetes and Sleep Problems
- CGM, Nocturnal Glucose, and Sleep
- Intermittent Fasting, Blood Sugar, and Sleep
- Keto and 3am Waking
- Visceral Fat, Growth Hormone, and Sleep
- Fatty Liver and Sleep Problems
- Gut Dysbiosis, Blood Sugar, and Sleep
- Late-Night Eating, Blood Sugar, and Sleep
References
1. Bolli, G. B., De Feo, P., De Cosmo, S., Perriello, G., Ventura, M. M., Calcinaro, F., Lolli, C., Campbell, P., Brunetti, P., & Gerich, J. E. (1984). Demonstration of a dawn phenomenon in normal human volunteers. Diabetes, 33(12), 1150-1153. https://pubmed.ncbi.nlm.nih.gov/6389230/
2. Campbell, P. J., Bolli, G. B., Cryer, P. E., & Gerich, J. E. (1985). Pathogenesis of the dawn phenomenon in patients with insulin-dependent diabetes mellitus. The New England Journal of Medicine, 312(23), 1473-1479. https://pubmed.ncbi.nlm.nih.gov/2859524/
3. Rosenthal, M. J., & Argoud, G. M. (1989). Absence of the dawn glucose rise in nondiabetic men compared by age. Journal of Gerontology, 44(2), M57-M61. https://pubmed.ncbi.nlm.nih.gov/2646356/
4. Atiea, J. A., Aslan, S. M., Owens, D. R., & Luzio, S. (1990). Early morning hyperglycaemia “dawn phenomenon” in non-insulin dependent diabetes mellitus (NIDDM): effects of cortisol suppression by metyrapone. Diabetes Research, 14(4), 181-185. https://pubmed.ncbi.nlm.nih.gov/2132191/
5. Van Cauter, E., Blackman, J. D., Roland, D., Spire, J. P., Refetoff, S., & Polonsky, K. S. (1991). Modulation of glucose regulation and insulin secretion by circadian rhythmicity and sleep. The Journal of Clinical Investigation, 88(3), 934-942. https://pubmed.ncbi.nlm.nih.gov/1885778/
6. Lundqvist, M. H., Pereira, M. J., Almby, K., Hetty, S., & Eriksson, J. W. (2024). Regulation of the Cortisol Axis, Glucagon, and Growth Hormone by Glucose Is Altered in Prediabetes and Type 2 Diabetes. The Journal of Clinical Endocrinology and Metabolism, 109(2), e675-e688. https://pubmed.ncbi.nlm.nih.gov/37708362/
7. Cai, J., Peng, P., Lu, J., Shen, Y., Wang, C., Mo, Y., Lu, W., Zhu, W., Xia, T., & Zhou, J. (2024). Severe dawn phenomenon predicts long-term risk of all-cause mortality in patients with type 2 diabetes. Diabetes/Metabolism Research and Reviews, 40(4), e3813. https://pubmed.ncbi.nlm.nih.gov/38767128/
8. Ng, A. S. C., Tai, E. S., & Chee, M. W. L. (2025). Effects of night-to-night variations in objectively measured sleep on blood glucose in healthy university students. Sleep, 48(2), zsae224. https://pubmed.ncbi.nlm.nih.gov/39325824/
9. Hansen, C., Fisker-Andersen, J., Rasmussen, C., Ceutz, F. R., Holst, J. J., Winther-Sorensen, M., & Wewer Albrechtsen, N. J. (2025). Glucagon acutely stimulates hepatic gluconeogenesis. Peptides, 194, 171448. https://pubmed.ncbi.nlm.nih.gov/41093268/
10. Unni, U. S., Bril, F., Mugler, J. P., Carter, R. E., Basu, A., & Basu, R. (2025). Role of Hepatic Glycogen on Nocturnal Gluconeogenesis in Type 2 Diabetes Mellitus. The Journal of Clinical Endocrinology and Metabolism, 110(10), 2790-2799. https://pubmed.ncbi.nlm.nih.gov/39903644/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 10 references cited
