Between 2% and 14% of people with long COVID develop POTS. The majority of people with dysautonomia report sleep disruption. Yet sleep content is rarely written for this population — and the advice they receive does not work because it does not address the underlying autonomic imbalance.
This article covers how POTS and dysautonomia disrupt sleep architecture, why conventional sleep advice does not work when the autonomic nervous system is involved, and which condition-focused approaches have evidence. If you are also looking at the broader picture of how autonomic imbalance affects sleep, the full overview is here: How Does Autonomic Nervous System Impairment Disrupt Sleep?
Autonomic imbalance is one of several causes of sleep disruption. POTS is under-recognized as a cause — and responsive to management once identified.
How Does Postural Orthostatic Tachycardia Syndrome Disrupt Sleep?
The Bagai et al. (2011) study quantified how POTS affects sleep. In that cross-sectional comparison, POTS participants scored 58 (SD 18) on the MOS Sleep Problems Index versus 20 (SD 13) for healthy controls. Daytime sleepiness was elevated: 10.2 (SD 5.7) versus 6.2 (SD 3.7) on the Epworth Sleepiness Scale. Fatigue was 7.5 (SD 2.0) versus 2.8 (SD 2.5) on a visual analogue scale. The MOS Sleep Problems Index and fatigue measures reached p < 0.0001; daytime sleepiness reached p = 0.001.
The finding that matters here: sleep problem severity alone accounted for approximately 50% of the variance in health-related quality of life in the POTS group (R² = 0.53). This positions sleep disruption as a primary driver of functional impairment in POTS — not a secondary complaint.
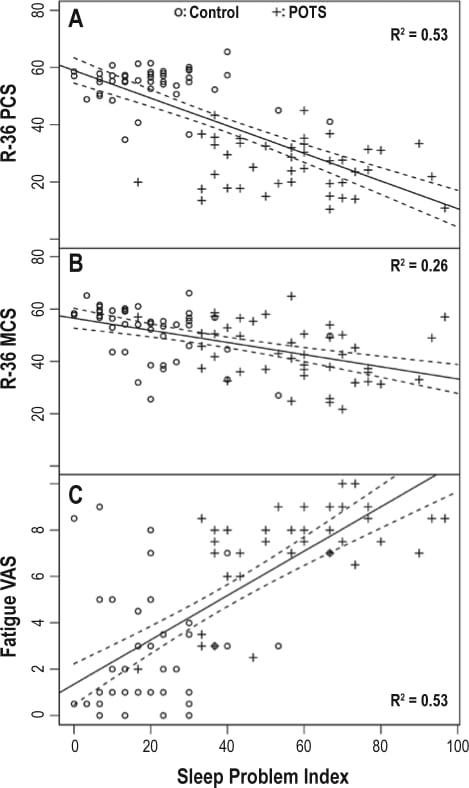
Mallien et al. (2014) added objective data. Using polysomnography with simultaneous heart rate variability recording in 38 people with POTS and 31 controls, they found that POTS participants spent a higher proportion of sleep time in stage 2 non-REM sleep and showed reduced variability in both low-frequency and high-frequency spectral bands across sleep stages. The LF/HF ratio — a measure of sympathovagal balance — was blunted, indicating that the autonomic nervous system was not cycling between sympathetic and parasympathetic dominance the way it does in healthy sleep.
In healthy sleep, parasympathetic tone increases during deep sleep and sympathetic activity rises during REM. In POTS, that cycling is diminished. The sympathovagal imbalance that characterizes POTS during the day does not resolve at night.
Miglis and Barwick (2018) synthesized these findings in a comprehensive review, noting that fatigue and unrefreshing sleep are among the features people with POTS rate as disabling — and that they are not explained by polysomnographic pathology alone. The nocturnal persistence of sympathetic overactivation, including elevated heart rate and reduced parasympathetic tone, prevents restorative sleep and perpetuates a cycle in which poor sleep worsens next-day orthostatic intolerance, which worsens the following night’s sleep.
Why Does Norepinephrine Keep People With Postural Orthostatic Tachycardia Syndrome Awake at Night?
Shannon et al. (2000) identified the first genetic cause of hyperadrenergic orthostatic intolerance: a loss-of-function mutation in the norepinephrine transporter gene. The index case carried a heterozygous mutation that produced greater than 98% loss of transporter function. Standing plasma norepinephrine was 923 pg/mL — more than double the 439 pg/mL average in healthy individuals.
The norepinephrine transporter (NET) is the primary mechanism for clearing norepinephrine from the synapse after release. When NET is impaired, norepinephrine lingers. The result is sustained sympathetic activation: elevated heart rate, heightened alertness, and persistent arousal — including at night when the body should be transitioning to parasympathetic dominance for sleep.
This genetic case established a mechanistic principle. The majority of people with POTS do not carry this mutation, but the downstream state is similar. In post-COVID POTS, vagal nerve damage removes the parasympathetic counter-regulation that normally balances sympathetic activity. The effect is functionally equivalent: norepinephrine-driven arousal persists into the hours when sleep requires the opposite.
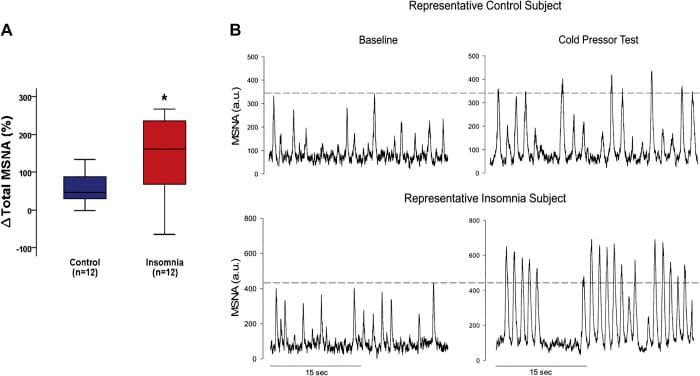
Greenlund and Carter (2022) reviewed the sympathoexcitation-insomnia connection across multiple conditions. Synthesizing studies that used direct microneurography (needle recordings of sympathetic nerve firing) and plasma norepinephrine measurements, the review showed that insomnia involves augmented muscle sympathetic nerve activity (MSNA) responses to stress — not just cortical arousal. People with insomnia showed augmented sympathetic responses to stress compared to people without insomnia. The relationship is bidirectional: sympathetic overactivity disrupts sleep, and disrupted sleep amplifies sympathetic tone.
Okamoto et al. (2024) identified a practical biomarker for the hyperadrenergic POTS subtype. A diastolic blood pressure rise exceeding 17 mmHg during late phase 2 of the Valsalva maneuver identified central sympathetic overactivity with 71% sensitivity and 85% specificity. In the same study’s pharmacological trial, guanfacine — a central alpha-2 adrenergic agonist that reduces norepinephrine release — produced improvement in 85% of hyperadrenergic POTS participants versus 44% of non-hyperadrenergic participants (p = 0.016). This showed that in the hyperadrenergic subtype, the norepinephrine excess is measurable and responsive to pharmacological approaches that reduce central sympathetic drive.
Why Does Standard Sleep Advice Not Work for People With Postural Orthostatic Tachycardia Syndrome?
Sleep hygiene assumes a healthy autonomic nervous system. It assumes that if the environment is right — dark room, cool temperature, consistent bedtime — the body will transition from sympathetic to parasympathetic dominance on its own. For people with POTS, that transition is impaired at the neurological level.
Three POTS-related mechanisms make standard advice insufficient:
Norepinephrine persistence. In hyperadrenergic POTS, elevated norepinephrine does not decrease at bedtime because the underlying cause — impaired norepinephrine clearance or excessive central sympathetic drive — is continuous. Reducing blue light exposure does not lower plasma norepinephrine levels.
Blood volume redistribution. When a person with POTS lies down, blood that was pooling in the lower extremities redistributes centrally. In healthy individuals, this is a minor adjustment. In POTS, reduced blood volume and impaired vasoconstriction can trigger compensatory tachycardia even in the supine position — the heart rate increases to maintain cardiac output despite inadequate venous return. This is a cardiovascular compensation, not a behavioral arousal that sleep hygiene can address.
Vagal insufficiency. Sleep onset requires a transition toward parasympathetic dominance. In post-COVID dysautonomia, vagal nerve damage may reduce the capacity for that transition. Goldstein (2024), writing in Nature Reviews Neurology, noted that existing studies of post-COVID autonomic impairment are “largely observational and descriptive” and that the precise mechanisms — including direct autonomic neurochemical measurements — remain among the largest knowledge gaps in the field. What is established: standard approaches are insufficient for this population.
Meenakshisundaram et al. (2024) demonstrated that COVID infection worsens pre-existing POTS. In a retrospective study of 41 people with established POTS who contracted COVID, 92.7% reported worsening of baseline autonomic features during acute infection. More concerning: 68% experienced persistent worsening for one to six months after infection, and 73.2% required escalation of their medications. For people already managing POTS, COVID does not just add a temporary burden — it compounds the existing autonomic impairment and may worsen sleep disruption that was already present.
What Condition-Focused Approaches Help Dysautonomia-Related Sleep Disruption?
These approaches are organized by what they address. Each counteracts a component of the autonomic imbalance rather than behavioral or environmental factors.
Elevated head-of-bed sleeping. Raising the head of the bed 6 to 10 inches (not just adding pillows — the entire bed angled so feet are lower) reduces venous blood pooling in the upper body and decreases the compensatory tachycardia that occurs when a person with POTS lies flat. This is one of the first changes to consider for POTS-related sleep disruption — it requires no medication and can be implemented the same day.
Compression garments. Abdominal compression is more effective than leg-only compression for reducing blood pooling. Waist-high compression garments worn during the evening and at bedtime help maintain venous return and reduce the heart rate elevations that occur with positional changes during sleep.
Volume expansion before bed. Increasing sodium and fluid intake 2 to 3 hours before bed — not at bedtime, which causes nocturia — expands blood volume and reduces compensatory tachycardia. The timing matters: electrolyte-containing fluids taken too close to sleep increase nighttime urination, replacing one sleep disruption with another.
Slow-exhale breathing. Breathing with a longer exhale than inhale (for example, 4 seconds in, 6 seconds out) directly activates vagal afferents and promotes parasympathetic tone. This is not a relaxation technique in the general sense — it is a direct autonomic input. In people with POTS, where parasympathetic activation is insufficient, this provides a stimulus that sleep hygiene recommendations do not include.
Pharmacological approaches. Okamoto et al. (2024) demonstrated that guanfacine produced improvement in 85% of people with hyperadrenergic POTS, with fatigue scores dropping by 12.9 points versus 2.2 in non-responders (p = 0.005). Other pharmacological options include low-dose propranolol (reduces heart rate; low doses may minimize blood pressure effects), ivabradine (selectively reduces heart rate through the sinoatrial node), and fludrocortisone (expands blood volume by promoting sodium and water retention).
Eastin et al. (2025) followed 526 adults with long-COVID-associated autonomic impairment and found median autonomic feature duration of 36 months, with 37.5% unable to work or attend school. This timeline underscores that these approaches may need to be sustained over months to years, not days to weeks. POTS-related sleep disruption is a chronic management challenge, and the autonomic approaches that help need to be maintained as long as the underlying condition persists.
Does COVID Cause Postural Orthostatic Tachycardia Syndrome?
The relationship between COVID and POTS is now well-documented across multiple study designs. The Eastin et al. (2025) cohort of 526 adults with long COVID found that 71.9% scored in the moderate-to-severe range on the COMPASS-31 autonomic impairment scale, and of those, 40.5% received a new POTS identification. Female sex and joint hypermobility were risk factors for greater severity.
For people who already had POTS before COVID, the picture is more concerning. Meenakshisundaram et al. (2024) found that COVID did not just temporarily exacerbate features — 73.2% of their cohort required medication escalation, indicating that the infection produced a durable worsening of the underlying autonomic state.
If you experienced a COVID infection and developed new nighttime heart racing, insomnia that began after an infection, or sleep disruption alongside other long COVID features, a POTS evaluation is a reasonable next step — especially if these features worsen with standing or positional changes.
How Common Is Sleep Disruption in Postural Orthostatic Tachycardia Syndrome?
The Bagai et al. (2011) data showed that sleep problems in POTS are not mild inconveniences — the scores were in a range associated with functional impairment in other conditions. The Ahmed et al. (2026) two-year follow-up of 93 neurology outpatients with post-COVID features (44 of whom completed the two-year follow-up) found that 66% of follow-up participants met criteria for autonomic impairment at the two-year mark, and sleep problems were among the features associated with higher COMPASS-31 severity scores. Each additional post-COVID feature was associated with an average 1.5-point increase in autonomic severity.
This means sleep disruption in POTS is not something that resolves on its own timeline. It tracks with the autonomic condition itself — and managing one requires managing the other.
Can Postural Orthostatic Tachycardia Syndrome Be Managed Long-Term, or Does It Resolve?
The honest answer: the long-term trajectory of post-COVID POTS is not yet well-characterized. Goldstein (2024) noted this as a primary gap — the field lacks prospective studies using direct autonomic testing to track recovery. What the cohort data shows is that over a third of people continue to experience functional impairment at 2 to 3 years.
For sleep, this means that the condition-focused approaches described in this article — head-of-bed elevation, compression, volume expansion, breathing practices, and in some cases pharmacological management — are often sustained strategies rather than short-term fixes. The autonomic impairment and the sleep disruption it produces tend to move together: as the autonomic state improves, sleep improves with it.
Postural orthostatic tachycardia syndrome and dysautonomia may not be the only factor affecting your sleep. Autonomic hyperarousal, GABA receptor changes, metabolic disruptions, or inflammatory processes may also be contributing. When multiple causes overlap, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does COVID Cause Postural Orthostatic Tachycardia Syndrome?
COVID-19 can trigger postural orthostatic tachycardia syndrome through several pathways: direct vagal nerve damage, autoimmune-mediated small fiber neuropathy, and persistent neuroinflammation that disrupts autonomic regulation. The Seeley et al. (2025) study used standardized standing heart rate testing and COMPASS-31 questionnaires to quantify the autonomic burden, finding that the majority of long COVID participants met the 30-beat-per-minute heart rate increase threshold. For people who already had autonomic instability before COVID, the infection added a second layer of impairment — Meenakshisundaram et al. (2024) documented worsened and prolonged autonomic experiences in this pre-existing population.
How Common Is Sleep Disruption in People With Postural Orthostatic Tachycardia Syndrome?
Sleep disruption in postural orthostatic tachycardia syndrome is not a secondary complaint — it is a primary contributor to functional impairment. The Bagai et al. (2011) data showed that sleep disturbance severity alone explained approximately 50% of the variability in health-related quality of life (R²=0.53). Fatigue scores were also elevated nearly three-fold (7.5±2.0 vs 2.8±2.5). The Ahmed et al. (2026) longitudinal data confirmed that this pattern persists — sleep impairment remained a primary contributor to autonomic burden 2 years after COVID infection.
Can Postural Orthostatic Tachycardia Syndrome Be Resolved or Does It Require Long-Term Management?
The recovery timeline varies. Some people improve over months as vagal function recovers. Others experience persistent autonomic instability years after the initial infection. The Eastin et al. (2025) cohort data — with a 36-month median duration — suggests that this is not a short-term post-illness effect for many people. The condition-focused approaches described in this article (elevated head sleeping, volume expansion, norepinephrine management) may need to be sustained for the duration of the autonomic impairment rather than applied as a short-term course.
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Does Lion’s Mane Affect Your Sleep? Why It Helps Some People and Keeps Others Awake — lion’s mane’s dual mechanism: neurotrophic activity, cholinergic activity, and sleep architecture
- Why Do Cholinergic Supplements Give You Vivid Dreams? — the acetylcholine-REM pathway behind supplement-related vivid dreams
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — how post-viral autonomic disruption can affect sleep continuity
- Why Does Your Heart Race at Night After COVID? What the Vagus Nerve Has to Do With It — why post-COVID autonomic instability can show up as nighttime tachycardia
- Can a Viral Infection Cause Chronic Insomnia? — how viral infections can disrupt autonomic regulation and sleep
- Why Will Your Nervous System Not Let You Sleep? Polyvagal Theory and Insomnia — how polyvagal states map to insomnia, hyperarousal, and unrefreshing sleep
- Why Does Trauma Cause Insomnia? How Your Nervous System Stays on Guard at Night — how trauma can keep sleep shaped by hyperarousal and vigilance
- Why Are You Sleeping 12 Hours and Still Exhausted? The Dorsal Vagal Shutdown Explained — the dorsal vagal pattern behind long sleep that still feels unrefreshing
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- How Do Probiotics Reach Your Brain? The Vagotomy Evidence — what vagotomy studies show about gut-brain communication
- Does Poor Sleep Damage Your Gut Microbiome? — the relationship between sleep fragmentation, microbiome disruption, and inflammatory load
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
- Bagai, K., Song, Y., Ling, J. F., Malow, B., Black, B. K., Biaggioni, I., Robertson, D., & Raj, S. R. (2011). Sleep disturbances and diminished quality of life in postural tachycardia syndrome. Journal of Clinical Sleep Medicine, 7(2), 204-210. https://pubmed.ncbi.nlm.nih.gov/21509337/
- Mallien, J., Isenmann, S., Mrazek, A., & Haensch, C. A. (2014). Sleep disturbances and autonomic dysfunction in patients with postural orthostatic tachycardia syndrome. Frontiers in Neurology, 5, 118. https://pubmed.ncbi.nlm.nih.gov/25071706/
- Shannon, J. R., Flattem, N. L., Jordan, J., Jacob, G., Black, B. K., Biaggioni, I., Blakely, R. D., & Robertson, D. (2000). Orthostatic intolerance and tachycardia associated with norepinephrine-transporter deficiency. The New England Journal of Medicine, 342(8), 541-549. https://pubmed.ncbi.nlm.nih.gov/10684912/
- Miglis, M. G., & Barwick, F. (2018). Sleep disorders in patients with postural tachycardia syndrome: A review of the literature and guide for clinicians. Autonomic Neuroscience: Basic & Clinical, 215, 62-69. https://pubmed.ncbi.nlm.nih.gov/29773483/
- Greenlund, I. M., & Carter, J. R. (2022). Sympathetic neural responses to sleep disorders and insufficiencies. American Journal of Physiology: Heart and Circulatory Physiology, 322(3), H337-H349. https://pubmed.ncbi.nlm.nih.gov/34995163/
- Okamoto, L. E., Urechie, V., Rigo, S., Abner, J. J., Giesecke, M., Muldowney, J. A. S., Furlan, R., Shibao, C. A., Shirey-Rice, J. K., Pulley, J. M., Diedrich, A., & Biaggioni, I. (2024). Hyperadrenergic postural tachycardia syndrome: Clinical biomarkers and response to guanfacine. Hypertension, 81(11), 2237-2247. https://pubmed.ncbi.nlm.nih.gov/39109428/
- Goldstein, D. S. (2024). Post-COVID dysautonomias: What we know and (mainly) what we don’t know. Nature Reviews Neurology, 20(2), 99-113. https://pubmed.ncbi.nlm.nih.gov/38212633/
- Meenakshisundaram, C., Moustafa, A., Ranabothu, M., Maraey, A., & Grubb, B. (2024). Impact of COVID-19 infection on baseline autonomic symptoms in patients with preexisting postural tachycardia syndrome and orthostatic intolerance: A retrospective study. The American Journal of the Medical Sciences, 367(5), 323-327. https://pubmed.ncbi.nlm.nih.gov/38340983/
- Eastin, E. F., Machnik, J. V., Stiles, L. E., Larsen, N. W., Seliger, J., Geng, L. N., Bonilla, H., Yang, P. C., & Miglis, M. G. (2025). Chronic autonomic symptom burden in long-COVID: A follow-up cohort study. Clinical Autonomic Research, 35(3), 453-464. https://pubmed.ncbi.nlm.nih.gov/39907931/
- Ahmed, S., Greenberg, J., Kenney, R., Marini, C., Hyman, S., Fung, S., Edeoga, N., Baltazar, M., Grossman, S. N., Seixas, A., Jean-Louis, G., Osorio, R. S., Condos, R., Frontera, J., Gonzalez-Duarte Briseno, M. A., Galetta, S. L., Balcer, L. J., & Thawani, S. P. (2026). Autonomic dysfunction and quality of life in a cohort of neurology outpatients with post-acute sequelae of COVID-19, a two-year follow-up study. Journal of Clinical Neuroscience, 143, 111719. https://pubmed.ncbi.nlm.nih.gov/41202571/
- Seeley, M.-C., Gallagher, C., Ong, E., Langdon, A., Chieng, J., Bailey, D., Page, A., Lim, H. S., & Lau, D. H. (2025). High incidence of autonomic dysfunction and postural orthostatic tachycardia syndrome in patients with long COVID. The American Journal of Medicine, 138(2), 354-361.e1. https://pubmed.ncbi.nlm.nih.gov/37391116/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 11 references cited
