Sleep disturbance affects an estimated 38-46% of people with long COVID (Chinvararak & Chalder, 2023) — and standard explanations like stress, disrupted routine, or health anxiety do not account for the severity or persistence. The emerging evidence points to a structural explanation: damage to the vagus nerve itself.
This article covers how SARS-CoV-2 physically damages the vagus nerve, how vagal impairment produces insomnia, why the “exhausted but unable to sleep” paradox occurs, and what approaches target this mechanism. It does not cover the full autonomic framework — for that, see the autonomic sleep disruption pillar, which maps multiple autonomic causes of disrupted sleep. Vagal impairment is one of several autonomic causes, and understanding it in isolation helps explain a pattern that many people with long COVID recognize but cannot make sense of.
How Does COVID Damage the Vagus Nerve?
The strongest evidence for direct vagal infection comes from a 2023 postmortem study by Woo et al. The research team examined vagus nerve tissue from COVID-19 decedents and detected SARS-CoV-2 RNA accompanied by inflammatory cell infiltration composed predominantly of monocytes. RNA sequencing of that same vagal tissue revealed a pronounced inflammatory response, and the magnitude of inflammation correlated with the tissue SARS-CoV-2 RNA load (Woo et al., 2023).
The relevance for sleep is in the cell types affected. Schwann cells — the glial cells responsible for maintaining myelin sheaths and nerve conduction velocity in peripheral nerves — showed inflammatory damage. Schwann cell impairment would be expected to reduce the speed and reliability of autonomic transmission carried by the vagus nerve, including the parasympathetic transmission that regulates heart rate, respiratory rate, and the autonomic transitions required for sleep.
In living people, a 2024 pilot cross-sectional study by Lladós et al. measured the vagus nerve directly using ultrasound in three groups: 30 individuals with post-COVID experiences suggestive of vagal involvement, 14 COVID-recovered asymptomatic controls, and 16 never-infected participants. The post-COVID group showed thickening of the vagus nerve (2.4 mm² vs. 2.0 mm² in recovered controls and 1.9 mm² in never-infected individuals; p = 0.08). Functional markers were affected too — 34% of the post-COVID group had reduced intestinal peristalsis, and 47% showed diaphragmatic flattening, both consistent with impaired vagal and phrenic nerve function (Lladós et al., 2024).
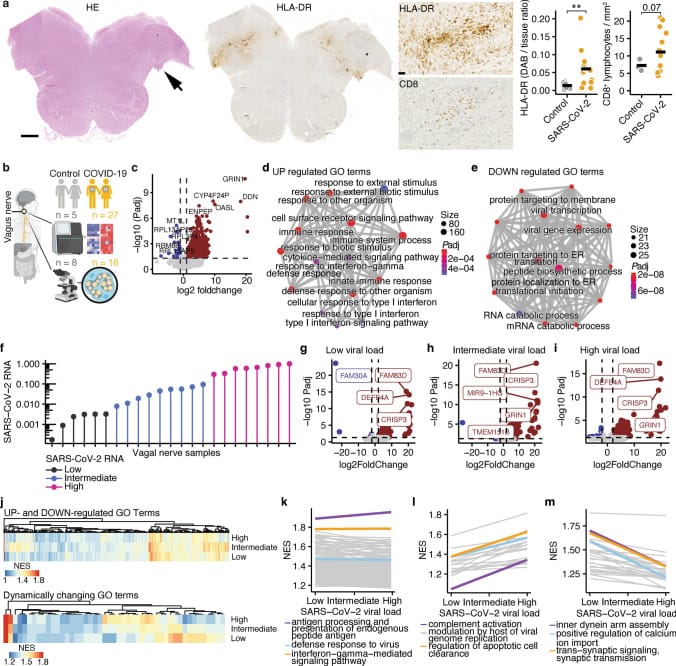
The damage extends beyond the vagus nerve itself. Small fiber neuropathy — injury to unmyelinated C-fibers and thinly myelinated A-delta fibers — has been confirmed in post-COVID adults via skin punch biopsy, with autonomic co-involvement documented in a subset of those cases (Abrams et al., 2022). A 2026 review by Morcos and Theoharides detailed the cellular mechanism: the SARS-CoV-2 spike protein activates mast cells through both ACE2 receptor binding and toll-like receptor 4 (TLR4) engagement, triggering release of interleukin-1-beta, interleukin-6, tumor necrosis factor-alpha, histamine, and tryptase. These mediators sensitize peripheral nerve fibers and disrupt the blood-brain barrier, driving small-fiber injury and autonomic instability (Morcos & Theoharides, 2026).
If you have experienced nighttime heart racing after COVID, the vagal damage pathway described here is one plausible mechanism — impaired parasympathetic regulation of heart rate during sleep.
Why Does Vagus Nerve Damage Cause Insomnia?
Sleep onset requires an active increase in parasympathetic activity: heart rate needs to decrease, blood pressure needs to drop, cortisol needs to fall, and respiratory rate needs to slow. The vagus nerve is the primary conduit for this parasympathetic output. When it is impaired, the body cannot execute the autonomic transition that sleep requires.
Acanfora et al. (2022) measured this directly using 24-hour ECG-derived heart rate variability in 30 long COVID adults and 20 healthy controls. Total HRV power was reduced in the long COVID group (7.46 vs. 8.08 log units; p < 0.0001). The high-frequency component of HRV — the spectral band that reflects vagal tone — was also lower (4.65 vs. 5.33; p = 0.015). The LF/HF ratio, a measure of sympathovagal balance, was elevated in the long COVID group (1.46 vs. 1.23; p = 0.001), indicating a tilt toward sympathetic dominance.
In practical terms: autonomic activity in these individuals was measurably insufficient for sleep. Heart rate stayed elevated, sympathetic activity remained dominant, and the autonomic conditions for sleep onset were not met — even when fatigue was present.
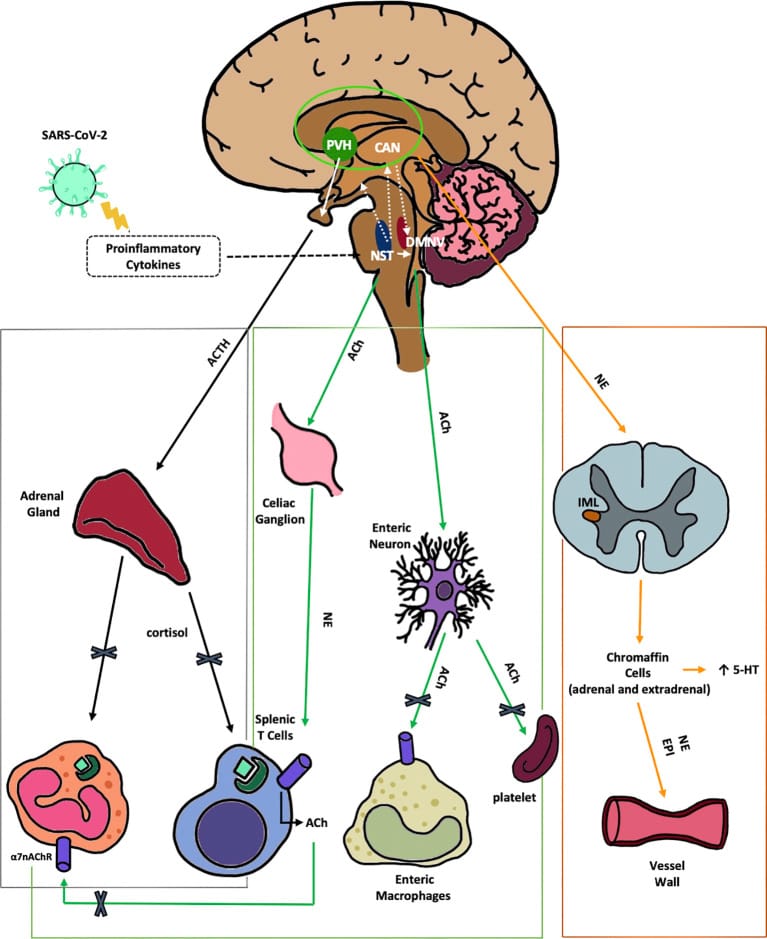
Dani et al. (2021) proposed autonomic disruption as a key mechanism underlying multiple long COVID symptoms, arguing that virus- or immune-mediated autonomic dysfunction — rather than isolated organ damage — explains persistent tachycardia, orthostatic intolerance, and blood pressure variability. Their framework described excessive catecholamine drive resulting from impaired parasympathetic counterregulation, a mechanism that would also be expected to impair sleep onset.
The damage does not stop at the heart-brain axis. Camici et al. (2024) extended the framework to include the hypothalamic-pituitary-adrenal (HPA) axis and mitochondrial function. In their model, vagal damage disrupts the cholinergic anti-inflammatory reflex, which normally suppresses cytokine production. Without this reflex, sustained inflammation dysregulates HPA feedback and cortisol rhythm. Since both intact vagal tone and a normal diurnal cortisol rhythm are required for sleep architecture, disruption of both simultaneously would be expected to impair both sleep onset and sleep continuity.
If you track overnight HRV, these numbers have a direct personal application — reduced high-frequency HRV and elevated LF/HF ratio during sleep are measurable markers of this vagal impairment. The HRV and vagal tone article covers how to interpret overnight HRV data in this context.
Why Are You Exhausted But Unable to Sleep After COVID?
This is the experience that brings people to this topic: bone-deep exhaustion that does not translate into the ability to sleep. It feels contradictory, but the mechanism explains why fatigue and insomnia co-exist.
The fatigue side: vagal damage disrupts the cholinergic anti-inflammatory reflex and HPA axis regulation, which downstream impairs mitochondrial oxidative phosphorylation — the primary energy-producing pathway in cells (Camici et al., 2024). Sustained inflammation also reduces serotonin availability — which may limit melatonin precursor supply (Camici et al., 2024). The result is cellular-level exhaustion.
The insomnia side: the same vagal impairment that disrupts energy metabolism also leaves sympathetic activation without adequate parasympathetic counterregulation. Heart rate stays elevated, cortisol does not follow its normal nocturnal decline, and the autonomic conditions for sleep onset are not present. The body is exhausted at the mitochondrial level while sympathetic activation remains elevated — increased heart rate, raised cortisol, heightened arousal.
Larsen et al. (2022) surveyed 2,314 adults with long COVID and found that difficulty sleeping was among the commonly reported symptoms, though daytime sleepiness scores remained below clinical thresholds in both hospitalized and non-hospitalized groups. Autonomic severity did not differ between those who had been hospitalized and those who had not (median COMPASS-31 scores: 28.95 vs. 26.4; p = 0.06) — meaning even mild initial illness can produce this pattern.
A 2-year follow-up study by Ahmed et al. (2026) found that sleep impairment was associated with increased autonomic severity at the 24-month mark — positioning sleep disruption as both a consequence and an indicator of ongoing autonomic injury. At two years, 66% of their cohort still met criteria for autonomic impairment.
This dual mechanism — mitochondrial fatigue plus autonomic hyperarousal — is what makes post-COVID insomnia resistant to standard sleep advice. Melatonin supplementation, sleep hygiene adjustments, and relaxation techniques may not be sufficient when the underlying vagal infrastructure is impaired. The insomnia has an autonomic basis, and approaches need to address that basis rather than behavioral factors alone.
For people with post-COVID insomnia who also experience orthostatic intolerance or POTS, the vagal impairment described here and the autonomic disruption in that condition share a common mechanism. If your experience extends to insomnia after other viral infections, the vagus nerve pathway may not be unique to SARS-CoV-2 — other viruses can produce similar autonomic damage.
What Can You Do About Post-COVID Sleep Disruption?
The hierarchy below moves from free, low-risk approaches toward more specialized ones. Each targets the vagal impairment described in the preceding sections rather than treating insomnia as an isolated experience.
Breathing-based vagal tone exercises. Slow exhalation breathing (inhale 4 seconds, exhale 8 seconds) and humming or chanting both activate vagal afferents. These are mechanical stimulation of the vagus nerve through the respiratory and laryngeal branches. Regular practice over weeks can measurably increase high-frequency HRV, the marker of parasympathetic tone that is reduced in long COVID.
Sleep environment modifications for autonomic impairment. Standard sleep hygiene takes on a different significance when autonomic regulation is compromised. A cooler room (65-67 degrees F) reduces sympathetic load — relevant because sympathetic activity is already elevated. For people with orthostatic co-involvement, elevating the head of the bed 10-15 degrees can reduce nocturnal blood pressure variability.
Supplements that support parasympathetic recovery. Magnesium glycinate (200-400 mg before bed) supports GABA receptor activity and muscle relaxation. L-theanine (200 mg) promotes alpha-wave activity associated with calm wakefulness and the transition to sleep. Neither is a vagus nerve repair agent, but both support the parasympathetic conditions that vagal impairment undermines.
Vagus nerve stimulation devices. Transcutaneous vagus nerve stimulation (tVNS) delivers electrical stimulation through the auricular branch of the vagus nerve. The rationale: if the vagus nerve is underperforming, external stimulation may partially restore parasympathetic tone. The VNS devices article covers device options, evidence, and practical considerations in detail.
Protecting sleep as a prevention measure. Quan et al. (2025) analyzed 24,803 U.S. adults and found that pre-existing insomnia was associated with a 30-52% increased risk of developing long COVID (adjusted odds ratios 1.30-1.52 across three PASC definition frameworks). Poor sleep quality showed an even stronger association — adjusted odds ratios of 1.77-2.00. This suggests that sleep health before and after infection influences the trajectory, making early sleep support a practical step during and after any SARS-CoV-2 infection.
Vagus nerve damage from COVID may not be the only factor affecting your sleep. Autonomic hyperarousal, GABA receptor changes, metabolic disruptions, or inflammatory processes may also be contributing. When multiple causes overlap, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
How Long Does Post-COVID Insomnia Last?
Ahmed et al. (2026) followed 93 long COVID adults over two years, with 44 completing the full assessment. At 24 months, sleep impairment showed a strong and statistically meaningful association with increased autonomic severity — each additional PASC symptom corresponded to a 1.5-point increase in the COMPASS-31 composite score. The duration of insomnia appears tied to the timeline of vagal and autonomic recovery rather than following a predictable resolution window.
Can COVID Cause Permanent Sleep Problems?
The 2-year data from Ahmed et al. (2026) shows persistent impairment, but the study cannot distinguish between permanent nerve damage and slow regeneration. Peripheral nerves can regenerate — Schwann cells, the glial cells damaged in COVID vagal neuropathy, are involved in peripheral nerve repair — but the timeline for vagal regeneration in this context is not yet characterized. The Quan et al. (2025) population data adds a nuance: people who entered their SARS-CoV-2 infection with existing sleep problems or poor sleep quality were more likely to develop long COVID, suggesting that pre-infection vagal tone and sleep health may affect long-term outcomes.
Is Post-COVID Insomnia Different From Regular Insomnia?
In standard insomnia, cognitive behavioral therapy for insomnia (CBT-I) is the first-line approach because the insomnia is maintained by conditioned arousal and maladaptive sleep behaviors. In post-COVID insomnia, the arousal arises from structural vagal damage that impairs parasympathetic output (Acanfora et al., 2022). CBT-I may still help with behavioral components, but it does not address the vagal impairment. Approaches that target vagal tone — breathing exercises, VNS devices, and reducing inflammatory load — address the mechanism that makes post-COVID insomnia different.
Does Vagus Nerve Stimulation Help Post-COVID Insomnia?
The rationale is direct: vagal impairment reduces parasympathetic output, and tVNS delivers electrical stimulation to the auricular branch of the vagus nerve to compensate. The evidence base for tVNS in insomnia is growing, and the mechanism aligns with the vagal damage model described in this article. For device options, stimulation parameters, and the current evidence, see the VNS devices article.
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Does Lion’s Mane Affect Your Sleep? Why It Helps Some People and Keeps Others Awake — lion’s mane’s dual mechanism: neurotrophic activity, cholinergic activity, and sleep architecture
- Why Do Cholinergic Supplements Give You Vivid Dreams? — the acetylcholine-REM pathway behind supplement-related vivid dreams
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Does Your Heart Race at Night After COVID? What the Vagus Nerve Has to Do With It — why post-COVID autonomic instability can show up as nighttime tachycardia
- Can POTS and Dysautonomia Cause Insomnia? Why Your Autonomic Nervous System Will Not Let You Rest — how orthostatic intolerance and autonomic instability can fragment sleep
- Can a Viral Infection Cause Chronic Insomnia? — how viral infections can disrupt autonomic regulation and sleep
- Why Will Your Nervous System Not Let You Sleep? Polyvagal Theory and Insomnia — how polyvagal states map to insomnia, hyperarousal, and unrefreshing sleep
- Why Does Trauma Cause Insomnia? How Your Nervous System Stays on Guard at Night — how trauma can keep sleep shaped by hyperarousal and vigilance
- Why Are You Sleeping 12 Hours and Still Exhausted? The Dorsal Vagal Shutdown Explained — the dorsal vagal pattern behind long sleep that still feels unrefreshing
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- How Do Probiotics Reach Your Brain? The Vagotomy Evidence — what vagotomy studies show about gut-brain communication
- Does Poor Sleep Damage Your Gut Microbiome? — the relationship between sleep fragmentation, microbiome disruption, and inflammatory load
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
Abrams, R. M. C., Simpson, D. M., Navis, A., Jette, N., Zhou, L., & Shin, S. C. (2022). Small fiber neuropathy associated with SARS-CoV-2 infection. Muscle & Nerve, 65(4), 440-443. https://pubmed.ncbi.nlm.nih.gov/34766365/
Acanfora, D., Nolano, M., Acanfora, C., Colella, C., Provitera, V., Caporaso, G., Rodolico, G. R., Bortone, A. S., Galasso, G., & Casucci, G. (2022). Impaired vagal activity in long-COVID-19 patients. Viruses, 14(5), 1035. https://pubmed.ncbi.nlm.nih.gov/35632776/
Ahmed, S., Greenberg, J., Kenney, R., Marini, C., Hyman, S., Fung, S., Edeoga, N., Baltazar, M., Grossman, S. N., Seixas, A., Jean-Louis, G., Osorio, R. S., Condos, R., Frontera, J., Gonzalez-Duarte Briseno, M. A., Galetta, S. L., Balcer, L. J., & Thawani, S. P. (2026). Autonomic dysfunction and quality of life in a cohort of neurology outpatients with post-acute sequelae of COVID-19, a two-year follow-up study. Journal of Clinical Neuroscience, 143, 111719. https://pubmed.ncbi.nlm.nih.gov/41202571/
Chinvararak, C., & Chalder, T. (2023). Prevalence of sleep disturbances in patients with long COVID assessed by standardised questionnaires and diagnostic criteria: A systematic review and meta-analysis. Journal of Psychosomatic Research, 175, 111535. https://pubmed.ncbi.nlm.nih.gov/37898059/
Camici, M., Del Duca, G., Brita, A. C., & Antinori, A. (2024). Connecting dots of long COVID-19 pathogenesis: A vagus nerve-hypothalamic-pituitary-adrenal-mitochondrial axis dysfunction. Frontiers in Cellular and Infection Microbiology, 14, 1501949. https://pubmed.ncbi.nlm.nih.gov/39735263/
Dani, M., Dirksen, A., Taraborrelli, P., Torocastro, M., Panagopoulos, D., Sutton, R., & Lim, P. B. (2021). Autonomic dysfunction in ‘long COVID’: Rationale, physiology and management strategies. Clinical Medicine, 21(1), e63-e67. https://pubmed.ncbi.nlm.nih.gov/33243837/
Larsen, N. W., Stiles, L. E., Shaik, R., Schneider, L., Muppidi, S., Tsui, C. T., Geng, L. N., Bonilla, H., & Miglis, M. G. (2022). Characterization of autonomic symptom burden in long COVID: A global survey of 2,314 adults. Frontiers in Neurology, 13, 1012668. https://pubmed.ncbi.nlm.nih.gov/36353127/
Lladós, G., Massanella, M., Coll-Fernández, R., Rodríguez, R., Hernández, E., Lucente, G., López, C., Loste, C., Santos, J. R., España-Cueto, S., Nevot, M., Muñoz-López, F., Silva-Arrieta, S., Brander, C., Durà, M. J., Cuadras, P., Bechini, J., Tenesa, M., Martinez-Piñeiro, A., Herrero, C., Chamorro, A., Garcia, A., Grau, E., Clotet, B., Paredes, R., & Mateu, L. (2024). Vagus nerve dysfunction in the post-COVID-19 condition: A pilot cross-sectional study. Clinical Microbiology and Infection, 30(4), 515-521. https://pubmed.ncbi.nlm.nih.gov/37984511/
Morcos, Z. L., & Theoharides, T. C. (2026). Long COVID neuropathy: The role of mast cells. Journal of Neuropathology & Experimental Neurology, 85(5), 413-424. https://pubmed.ncbi.nlm.nih.gov/41790576/
Quan, S. F., Weaver, M. D., Czeisler, M. É., Barger, L. K., Booker, L. A., Howard, M. E., Jackson, M. L., Lane, R. I., McDonald, C. F., Ridgers, A., Robbins, R., Varma, P., Wiley, J. F., Rajaratnam, S. M. W., & Czeisler, C. A. (2025). Sleep and long COVID: Preexisting sleep issues and the risk of post-acute sequelae of SARS-CoV-2 infection in a large general population using 3 different model definitions. Journal of Clinical Sleep Medicine, 21(2), 249-259. https://pubmed.ncbi.nlm.nih.gov/39324686/
Woo, M. S., Shafiq, M., Fitzek, A., Dottermusch, M., Altmeppen, H., Mohammadi, B., Mayer, C., Bal, L. C., Raich, L., Matschke, J., Krasemann, S., Pfefferle, S., Brehm, T. T., Lütgehetmann, M., Schädler, J., Addo, M. M., Schulze Zur Wiesch, J., Ondruschka, B., Friese, M. A., & Glatzel, M. (2023). Vagus nerve inflammation contributes to dysautonomia in COVID-19. Acta Neuropathologica, 146(3), 387-394. https://pubmed.ncbi.nlm.nih.gov/37452829/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 11 references cited
