Rapid eye movement sleep is the stage that appears on wearable dashboards and generates concern when the numbers look low. Understanding what controls it — a brainstem circuit modulated by acetylcholine — moves the question from “what am I doing wrong” to “what is happening in the circuit.” Continuity in this sleep stage matters for brain health, emotional regulation, and long-term cognitive function. Declining amounts may be an early marker of cholinergic neurodegeneration.
This article covers the brainstem flip-flop circuit, the dual role of acetylcholine in onset versus maintenance, what happens when episodes fragment, and which medications suppress this sleep stage through the cholinergic pathway. For the broader picture, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It.
Cholinergic disruption is one of several autonomic causes of fragmented sleep. The autonomic pillar covers the full landscape: GABA-related impairment, hyperarousal, and stress-driven fragmentation.
What Is the Brainstem Switch That Controls Rapid Eye Movement Sleep?
The architecture was mapped in a 2006 study by Lu and colleagues, published in Nature. They identified two populations of GABAergic neurons in the mesopontine tegmentum that form a bistable circuit: a REM-off population in the vlPAG and lateral pontine tegmentum (LPT), and a REM-on population in the sublaterodorsal nucleus (SLD). Chemical lesions of the SLD eliminated REM. Lesions of the vlPAG produced continuous REM-like states. The mutual GABAergic inhibition between these populations is what makes REM transitions rapid and complete — the circuit flips between states rather than transitioning gradually (Lu et al., 2006).
Within the REM-on region, two anatomically distinct subpopulations serve different functions. One projects to the basal forebrain to generate the cortical activation that characterizes REM sleep — the vivid, dream-associated brain activity. The other projects to the brainstem and spinal cord to produce the muscle atonia (paralysis) that prevents acting out dreams (Lu et al., 2006).
A 2023 review by Vetrivelan and Bandaru confirmed that the glutamatergic neurons in the SLD are the exclusive generators of REM sleep. Selective genetic ablation of SLD glutamatergic neurons in mice eliminated all REM sleep — the definitive evidence to date that this single cell group is required for REM to occur. Cholinergic neurons in the pedunculopontine tegmentum (PPT) and laterodorsal tegmental nucleus (LDT) modulate REM but are not required for spontaneous REM generation. This repositions acetylcholine as a modulator of the circuit rather than its primary driver (Vetrivelan & Bandaru, 2023).
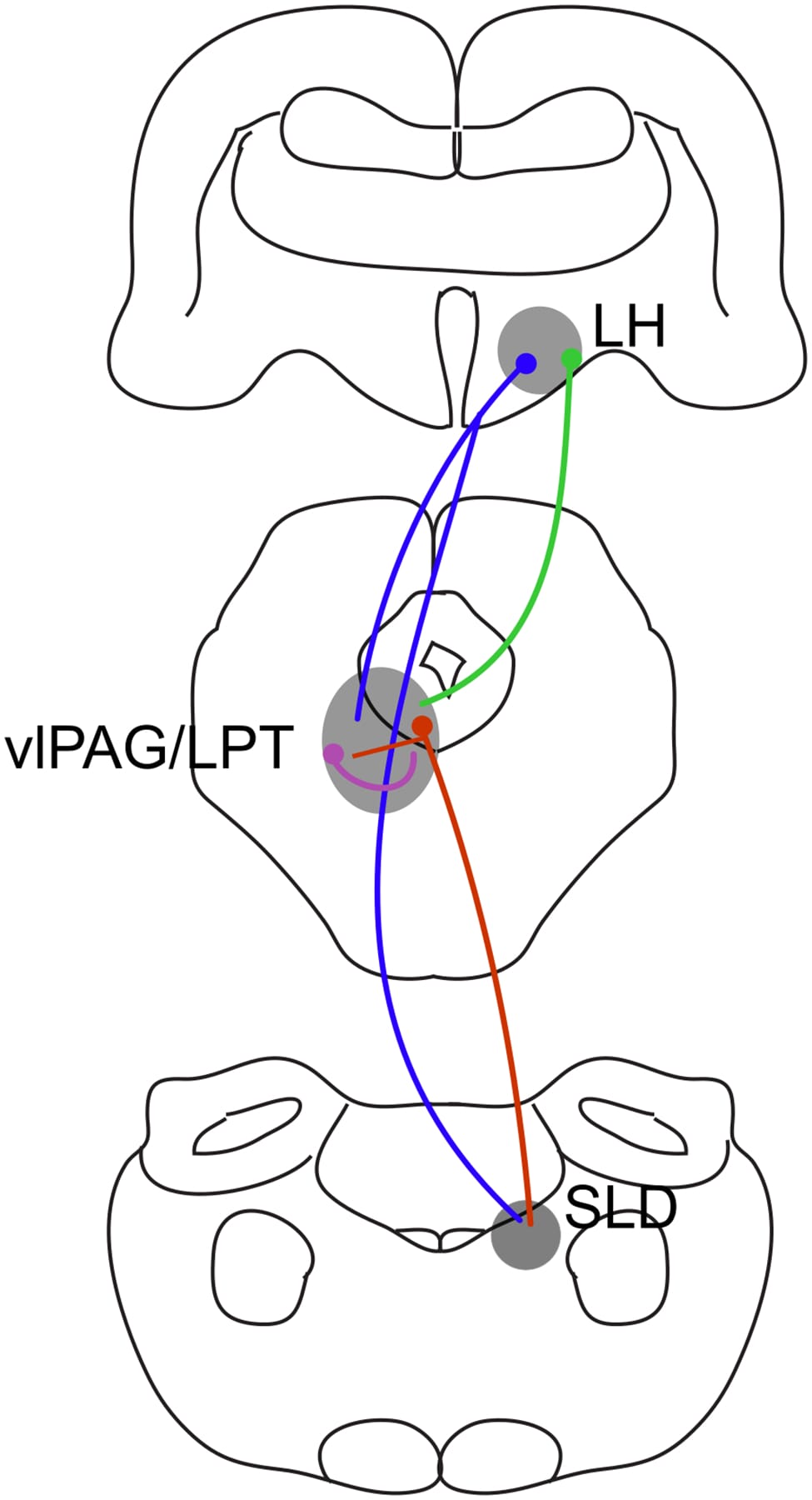
A 2025 review by Luppi integrated these findings into an updated circuit model. Melanin-concentrating hormone (MCH) neurons and GABAergic neurons in the lateral hypothalamus gate REM entry by inhibiting vlPAG REM-off neurons — providing a forebrain pathway that integrates sleep pressure and homeostatic demands into the brainstem circuit. The updated model describes “a brainstem generator controlled by forebrain structures involved in autonomic control,” positioning REM sleep as a distributed brain state rather than a purely brainstem event (Luppi, 2025).
The bistable architecture has a direct consequence for fragmentation: if either side of the circuit weakens — through neurodegeneration, medication, or altered neurotransmitter tone — the mutual inhibition becomes unstable. The circuit no longer latches cleanly into REM. Instead of sustained REM episodes, the result is brief, incomplete transitions that break REM into fragments.
How Does Acetylcholine Control When Dream Sleep Starts and How Long It Lasts?
The assumption that acetylcholine has a single role in REM — that it “turns on” dream sleep — is incomplete. Nissen and colleagues tested this directly in a double-blind, randomized, placebo-controlled crossover trial with 20 healthy volunteers. They compared the muscarinic M1 agonist RS-86 against donepezil (an acetylcholinesterase inhibitor that increases acetylcholine levels broadly) and placebo (Nissen et al., 2006).
The results revealed a clean pharmacological dissociation. RS-86 shortened REM latency — meaning it accelerated how quickly the first REM episode began after sleep onset — without changing total REM duration or the frequency of rapid eye movements during REM. Donepezil did the opposite: it extended total REM duration and increased rapid eye movement frequency without changing when REM started. Two different drugs acting on the same neurotransmitter produced two non-overlapping effects on two different components of REM architecture (Nissen et al., 2006).
This dissociation maps onto the brainstem circuit. M1 muscarinic receptors at the REM-on nuclei in the pons appear to control the timing of REM initiation — when the circuit flips from NREM into REM. A different muscarinic subtype — likely M2 — stabilizes each REM episode once it has begun, sustaining the suppression of vlPAG REM-off neurons that keeps the circuit latched in the REM-on state (Nissen et al., 2006).
The relevance to human variation was demonstrated earlier by Riemann and colleagues in a 1994 trial that administered RS-86 to 40 people with major depressive disorder, 43 people with schizophrenia, and 36 healthy controls. Both affected groups showed pronounced REM latency shortening after the muscarinic agonist — more than healthy controls — consistent with muscarinic receptor hypersensitivity in these conditions. This established that the degree of muscarinic sensitivity at brainstem REM-on nuclei varies across individuals and is altered in psychiatric conditions (Riemann et al., 1994).
The muscarinic receptor requirement was confirmed at the genetic level by Niwa and colleagues in 2018. Using CRISPR gene editing in mice, they knocked out M1 (Chrm1) and M3 (Chrm3) muscarinic receptors simultaneously. The double knockout virtually eliminated REM sleep. Single knockouts of either receptor reduced REM but did not abolish it, confirming that multiple muscarinic receptor subtypes contribute to REM generation and that their roles are partially redundant but collectively essential (Niwa et al., 2018).
The practical implication: drugs that block M1 muscarinic receptors would be expected to delay REM onset — producing temporally disorganized REM architecture where the first REM episode arrives late and subsequent episodes are compressed. Drugs that block acetylcholine more broadly (reducing overall cholinergic tone) would be expected to shorten individual REM episodes. Both effects fragment REM, but through different mechanisms.
What Happens to the Brain When Rapid Eye Movement Sleep Fragments?
Viselli and colleagues published a 2025 study in Sleep that isolated the consequences of REM fragmentation from total sleep loss. They enrolled 17 healthy young adults in a randomized crossover design and used vibrotactile wrist stimulation during polysomnographically verified REM sleep to selectively increase the REM fragmentation index. On fragmentation nights, the index reached 75.52 events per hour versus 23.12 events per hour on control nights — without altering total sleep time, sleep efficiency, or time spent awake after sleep onset (Viselli et al., 2025).
The measured outcome was heart rate deceleration (HRD) habituation — the autonomic nervous response in which heart rate progressively slows with repeated exposure to the same emotional stimulus. This is a marker of the brain’s ability to downregulate emotional reactivity at the autonomic level.
Selective REM fragmentation impaired HRD habituation to emotional images at both the immediate post-sleep assessment and at a 48-hour follow-up. Electrodermal activity, self-reported emotional responses, and recognition memory were unaffected. The impairment was isolated to the autonomic channel — the body’s cardiac response to emotion did not habituate, while the conscious experience of emotion and the ability to remember the images remained intact (Viselli et al., 2025).
The degree of impairment tracked with a measurable EEG marker: parieto-occipital alpha power intrusions during fragmented REM predicted impaired cardiac habituation (r = 0.710, p = .003 at immediate post-sleep; r = 0.618, p = .014 at 48-hour follow-up). Alpha intrusions into REM represent brief moments where the cortex transitions toward a waking-like state — interrupting the neural processing that REM provides for emotional regulation (Viselli et al., 2025).
This finding reframes what REM fragmentation costs. The primary measurable consequence in this study was not impaired memory, not subjective mood changes, and not daytime sleepiness. It was a loss of the autonomic nervous regulation that allows the body to calibrate its cardiac response to emotional stimuli — a function that runs below conscious awareness.
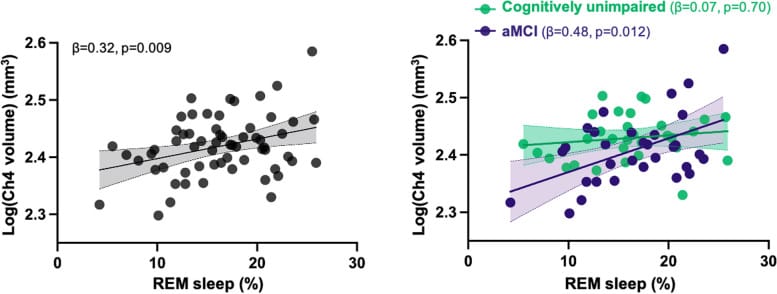
The long-term implications connect to cholinergic neurodegeneration. Andre and colleagues showed in a 2023 study that reduced REM sleep percentage in individuals with amnestic mild cognitive impairment (aMCI) correlated with lower volume of the nucleus basalis of Meynert — the brain region that provides the primary cholinergic innervation of the neocortex and hippocampus (beta = 0.48, p = 0.012). In cognitively healthy controls, this association was not present, suggesting that cholinergic degeneration must exceed a threshold before it produces detectable REM disruption. The authors concluded that REM sleep disturbances may constitute an early biomarker of basal forebrain cholinergic degeneration — appearing before the onset of Alzheimer’s dementia (Andre et al., 2023).
Which Medications Suppress Dream Sleep by Blocking This Cholinergic Pathway?
The mechanism described in the previous sections — muscarinic receptors governing both REM onset and REM maintenance — predicts that any drug blocking those receptors would suppress REM sleep. The pharmacological evidence supports this across multiple drug classes.
Diphenhydramine (Benadryl and common over-the-counter sleep aids): Diphenhydramine is classified as a first-generation antihistamine, but its anticholinergic activity is what affects REM. A 2025 expert consensus review reported that diphenhydramine reduces REM sleep by approximately 16.2% and redistributes sleep architecture toward lighter N1 and N2 stages. Many people take diphenhydramine to help with sleep, unaware that it suppresses the sleep stage associated with emotional regulation and cognitive maintenance (Expert Consensus, 2025).
Bladder antimuscarinics (oxybutynin, tolterodine): These drugs are prescribed for overactive bladder and act by blocking muscarinic receptors in the bladder wall. However, oxybutynin and tolterodine also cross the blood-brain barrier and block central muscarinic receptors. A 2005 study by Diefenbach and colleagues in adults aged 50 and older found that both drugs reduced REM sleep by approximately 15% (Diefenbach et al., 2005).
The same study included trospium — a bladder antimuscarinic that does not cross the blood-brain barrier because of its quaternary amine structure. Trospium had no effect on REM sleep. This dissociation between drugs that do and do not cross the blood-brain barrier demonstrates that the REM suppression is a central nervous effect — it occurs because these drugs reach the brainstem muscarinic receptors that the flip-flop circuit depends on, not because of peripheral anticholinergic activity (Diefenbach et al., 2005).
The genetic anchor: The Niwa 2018 CRISPR study provides the mechanistic basis. When M1 and M3 muscarinic receptors were genetically eliminated, REM was virtually abolished. The drugs described above are doing a partial, pharmacological version of the same thing — blocking the receptor subtypes that the brainstem circuit requires to initiate and maintain REM episodes (Niwa et al., 2018).
SSRIs suppress REM through a different pathway. Selective serotonin reuptake inhibitors reduce REM by increasing serotonergic tone — serotonin activates the REM-off side of the brainstem circuit through monoaminergic inputs. This is a different mechanism from anticholinergic suppression. A person taking both an SSRI and an anticholinergic medication may be suppressing REM through two independent pathways simultaneously.
Long-term cumulative exposure carries additional risk. Gray and colleagues analyzed data from 3,434 participants aged 65 and older over a median follow-up of 7.3 years. Cumulative anticholinergic use of three or more years was associated with 54% higher risk of dementia overall and 63% higher risk of Alzheimer’s disease, compared to no anticholinergic use. The dose-response relationship was present: higher cumulative exposure corresponded to higher risk. The study could not determine causality, but the association was consistent after adjusting for multiple confounders (Gray et al., 2015).
The connection between these findings runs in a single direction: muscarinic receptors are required for REM, anticholinergic drugs block muscarinic receptors, REM is suppressed, REM loss is associated with cholinergic neurodegeneration, and cumulative anticholinergic exposure is associated with higher dementia risk. Whether chronic REM suppression contributes to cognitive decline or whether both are downstream effects of a shared process remains an open question. The epidemiological and mechanistic data are consistent with both possibilities.
Fragmented REM might not have a single cause. Cholinergic pathway disruption can overlap with GABA receptor impairment, autonomic hyperarousal, or medication-induced suppression — and these causes might compound each other. When REM fragments, identifying which mechanisms might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Alcohol Reduce Rapid Eye Movement Sleep?
The mechanism has two phases. In the first half of the night, alcohol’s GABA-A agonist activity tips the brainstem circuit toward the REM-off state, delaying and shortening early REM episodes. As blood alcohol falls in the second half of the night, the GABA-A enhancement subsides and a compensatory excitability rebound occurs. This rebound destabilizes the mutual inhibition between the vlPAG REM-off and SLD REM-on populations, producing incomplete state transitions — the circuit attempts to enter REM but cannot sustain it (Lu et al., 2006).
The timing matters because REM episodes normally grow longer across the night. The longest and best-consolidated REM episodes occur in the final 2-3 hours of sleep. Alcohol’s rebound effect is strongest during this same window, disrupting the REM periods that would otherwise be the longest and allow the greatest amount of sustained REM processing.
Can Stress Cause Rapid Eye Movement Sleep Fragmentation?
Cortisol activates the locus coeruleus (norepinephrine) and dorsal raphe (serotonin), both of which project to the vlPAG REM-off population. Elevated monoaminergic input strengthens the REM-off side of the circuit, making it harder for the SLD REM-on neurons to overcome the inhibition and initiate a sustained REM episode.
The effect is pronounced in the early morning hours when cortisol naturally begins its pre-waking rise. In people with elevated baseline cortisol from chronic stress, this rise starts earlier and reaches higher levels — compressing and fragmenting the late-night REM episodes that are normally the longest. The Viselli 2025 study showed that even partial REM fragmentation is sufficient to impair autonomic emotional habituation, creating a feedback pattern where stress fragments REM, fragmented REM impairs emotional regulation, and impaired emotional regulation increases stress reactivity (Viselli et al., 2025).
How Much Rapid Eye Movement Sleep Should You Get Per Night?
In a typical 7-8 hour sleep period, a healthy adult spends approximately 90-120 minutes total in REM. The first REM episode may last only 5-10 minutes, while late-night episodes can extend to 30-45 minutes. This progressive lengthening means that cutting sleep short by even one hour disproportionately reduces REM, because it eliminates the cycle where REM is heavily concentrated (Vetrivelan & Bandaru, 2023).
Total REM percentage varies with age and individual physiology. The quality of REM — whether episodes are sustained or fragmented — may be more informative than the total amount. A person with 22% REM in sustained episodes has a different sleep architecture than a person with 22% REM broken into dozens of brief fragments.
Can You See Dream Sleep Fragmentation in Wearable Data?
Patterns to look for in wearable data include: REM episodes that rarely exceed 10-15 minutes, multiple brief awakenings during the 4:00-7:00 AM window (when REM should dominate), and consistently low REM percentage across multiple nights. A single night of low REM can reflect normal variation — the pattern across weeks is more informative than any individual number.
The limitation is that wearables cannot distinguish between true REM fragmentation (the brainstem circuit not sustaining REM) and REM that was not detected due to sensor limitations. Polysomnography — overnight EEG-based sleep recording — remains the only method that can directly measure REM fragmentation indices (Andre et al., 2023).
Related Reading:
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — how autonomic imbalance disrupts sleep architecture across all stages, not just REM
- Can Your Nervous System Get Stuck in Fight or Flight and Ruin Your Sleep? — sympathetic dominance and its effect on sleep onset and continuity
- What Is Hyperarousal Insomnia? Why You’re Wired but Tired Every Night — cortical and autonomic hyperarousal as a cause of sleep-onset and sleep-maintenance insomnia
- Why Won’t Your Brain Shut Off at Night? The Autonomic Nervous System Connection — autonomic drive and racing thoughts at bedtime
- Does Benadryl Destroy Your Sleep? How Anticholinergic Drugs Suppress REM Sleep — anticholinergic sleep damage through the muscarinic receptor pathway
- What Your Overnight HRV Is Telling You About Your Sleep: The Vagal Tone Connection — how vagal tone measured through HRV reflects autonomic sleep quality
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — vagal gut-brain communication and its role in sleep regulation
- I’ve Been Taking Benadryl Every Night for Years. Is It Bad? — anticholinergic sleep damage and long-term cognitive risk
- Alcohol Sedates You, Then Wakes You at 3AM — the GABA-A rebound mechanism that fragments REM in the second half of the night
- Why Won’t Your Brain Shut Off at Night? The Autonomic Connection — how GABA receptor impairment drives the sympathetic overactivation that prevents sleep
- Does Benadryl Degrade Your Sleep? How Anticholinergic Drugs Suppress Rapid Eye Movement Sleep — how anticholinergic medications block the muscarinic receptors REM requires
- What Your Overnight Heart Rate Variability Is Telling You About Your Sleep: The Vagal Tone Connection — using HRV as a window into parasympathetic recovery during sleep
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut bacteria, GABA production, and the vagal pathway to sleep regulation
- Do Antidepressants Suppress Rapid Eye Movement Sleep? — SSRIs, SNRIs, and tricyclics affect sleep architecture
- Can Vagus Nerve Stimulation Devices Improve Insomnia? — clinical trial evidence for taVNS devices
- Does Your Sleep Tracker Accuracy Matter? — what Oura, WHOOP, and Apple Watch get right and wrong
- Is Your Insomnia a Nervous System Problem? — how to tell if autonomic dysregulation is driving sleep disruption
References
Andre, C., Rehel, S., Kuhn, E., Landeau, B., Moulinet, I., Touron, E., Ourry, V., Le Du, G., Mezenge, F., Tomadesso, C., de Flores, R., Bejanin, A., Sherif, S., Schimmer, M., Bouet, R., Heckemann, R. A., Peres, K., Amieva, H., Eustache, F., … Rauchs, G. (2023). REM sleep is associated with the volume of the cholinergic basal forebrain in aMCI individuals. Alzheimer’s Research & Therapy, 15(1), 151. https://pubmed.ncbi.nlm.nih.gov/37684650/
Diefenbach, K., Donath, F., Maurer, A., Schreiner, V., Roots, I., & Bauer, S. (2005). Effects on sleep of anticholinergics used for overactive bladder treatment in healthy volunteers aged >= 50 years. BJU International, 95(3), 346-349. https://pubmed.ncbi.nlm.nih.gov/15679791/
Expert Consensus. (2025). Expert consensus on the use of diphenhydramine for short-term insomnia: Efficacy, safety, and clinical applications. Journal of Clinical Medicine, 14(10), 3297. https://pmc.ncbi.nlm.nih.gov/articles/PMC12112657/
Gray, S. L., Anderson, M. L., Dublin, S., Hanlon, J. T., Hubbard, R., Walker, R., Yu, O., Crane, P. K., & Larson, E. B. (2015). Cumulative use of strong anticholinergics and incident dementia: a prospective cohort study. JAMA Internal Medicine, 175(3), 401-407. https://pubmed.ncbi.nlm.nih.gov/25621434/
Lu, J., Sherman, D., Devor, M., & Saper, C. B. (2006). A putative flip-flop switch for control of REM sleep. Nature, 441(7093), 589-594. https://pubmed.ncbi.nlm.nih.gov/16688184/
Luppi, P. H. (2025). Neuronal network controlling REM sleep. Journal of Sleep Research, 34(2), e14266. https://pubmed.ncbi.nlm.nih.gov/38972672/
Nissen, C., Nofzinger, E. A., Feige, B., Waldheim, B., Radosa, M. P., Riemann, D., & Berger, M. (2006). Differential effects of the muscarinic M1 receptor agonist RS-86 and the acetylcholine-esterase inhibitor donepezil on REM sleep regulation in healthy volunteers. Neuropsychopharmacology, 31(6), 1294-1300. https://pubmed.ncbi.nlm.nih.gov/16192980/
Niwa, Y., Kanda, G. N., Yamada, R. G., Shi, S., Sunagawa, G. A., Ukai-Tadenuma, M., Fujishima, H., Matsumoto, N., Masumoto, K. H., Nagano, M., Kasukawa, T., Galloway, J., Perrin, D., Bhatt, D. K., Mori, T., Bhatt, A., Takashima, N., Ebihara, S., & Ueda, H. R. (2018). Muscarinic acetylcholine receptors Chrm1 and Chrm3 are essential for REM sleep. Cell Reports, 24(8), 2231-2247. https://pubmed.ncbi.nlm.nih.gov/30157420/
Riemann, D., Hohagen, F., Krieger, S., Gann, H., Muller, W. E., Olbrich, R., Wark, H. J., Bohus, M., Low, H., & Berger, M. (1994). Cholinergic REM induction test: muscarinic supersensitivity underlies polysomnographic findings in both depression and schizophrenia. Journal of Psychiatric Research, 28(3), 195-210. https://pubmed.ncbi.nlm.nih.gov/7932282/
Vetrivelan, R., & Bandaru, S. S. (2023). Neural control of REM sleep and motor atonia: current perspectives. Current Neurology and Neuroscience Reports, 23(12), 907-923. https://pubmed.ncbi.nlm.nih.gov/38060134/
Viselli, L., Annarumma, L., Ferrara, M., Galbiati, A., Ferini-Strambi, L., & De Gennaro, L. (2025). Experimentally induced REM sleep fragmentation affects psychophysiological habituation to emotional stimuli. Sleep, 48(2), zsae275. https://pubmed.ncbi.nlm.nih.gov/41432255/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 11 references cited
