Conventional explanations for excessive sleep include thyroid problems, sleep apnea, or depression. For many people, those workups come back normal — yet the pattern persists. The autonomic explanation is different: the body is in a conservation state that produces quantity of sleep without quality.
This article covers the dorsal vagal state and its effect on sleep architecture, how it differs from depression-related hypersomnia, the connection to freeze responses, and what gentle reactivation approaches look like. For the full autonomic framework, see the autonomic sleep disruption pillar. Dorsal vagal shutdown is one of several autonomic causes — the opposite end from the hyperarousal pattern.
What Is the Dorsal Vagal State and How Does It Affect Sleep?
The vagus nerve has two branches with different functions. The ventral vagal branch supports social engagement and the parasympathetic dominance that produces restorative sleep — heart rate variability cycling, parasympathetic surges during deep sleep, and the autonomic dynamics that make time asleep translate into recovery. The dorsal vagal branch supports immobilization and metabolic conservation.
When the autonomic state enters dorsal vagal dominance, it is not relaxing. It is conserving. Heart rate slows, metabolic rate drops, and the person becomes immobilized — but the autonomic dynamics required for restorative sleep are absent. The person sleeps for 10 or 12 hours and wakes feeling as though they did not sleep at all.
Roelofs (2017) mapped the neural circuitry underlying this response. The pathway runs from the basolateral amygdala through the central nucleus to the ventrolateral periaqueductal gray (vlPAG), which activates the vagal pathway to produce heart rate deceleration and muscular rigidity. Human neuroimaging shows comparable circuits.
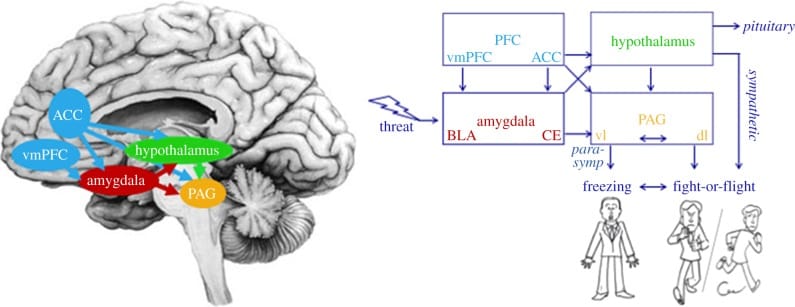
The review distinguishes tonic immobility — an extreme parasympathetically dominated state with loss of voluntary control — from the more common danger-induced freezing. Both involve vagal dominance, but they differ in reversibility. When the autonomic state cannot transition back from conservation, the result is sustained fatigue, dissociation, and prolonged unrefreshing sleep.
The ventromedial prefrontal cortex and anterior cingulate cortex normally modulate the transition between freezing and active engagement. When that regulation is disrupted — as Roelofs notes in anxiety disorders and PTSD — the person can become stuck in a freeze-conservation pattern. Sleep becomes long. Sleep does not become restorative.
What Does Dorsal Vagal Sleep Look Like on a Sleep Study?
Fatt et al. (2020) enrolled 24 people with chronic fatigue and 24 matched healthy controls, measuring heart rate variability across all sleep stages. Normalized high-frequency HRV power — the spectral band that reflects parasympathetic tone — was reduced in the chronic fatigue group compared to controls during stage N2 and slow-wave sleep (both p < .001). No between-group differences emerged during stage N1, REM sleep, or resting wakefulness.
The parasympathetic withdrawal was stage-specific — it occurred during the deeper sleep phases that matter for physical restoration. The chronic fatigue group spent a greater proportion of time in slow-wave sleep compared to controls yet received less restorative benefit from it. Parasympathetic withdrawal during deep sleep correlated with poorer subjective sleep quality.
Mombelli et al. (2025) examined 134 people with idiopathic hypersomnia using overnight polysomnography followed by a Multiple Sleep Latency Test. When classified by self-reported nap quality, no differences emerged in standard sleep architecture metrics between refreshing and unrefreshing naps — pointing toward a mechanism that standard scoring does not capture.
Miglis et al. (2020) measured autonomic impairment burden in idiopathic hypersomnia using the COMPASS-31 scale. Median autonomic scores: 43.6 (online cohort) and 32.9 (confirmed group) vs. 17.6 in healthy controls (p < .001). Autonomic severity correlated with sleepiness and fatigue severity — a dose-response relationship between autonomic impairment and symptom burden.
This is what makes the dorsal vagal framework useful: it explains why a person can spend 10-12 hours asleep and appear normal on a sleep study while experiencing none of the restoration that sleep is supposed to provide. The architecture may look normal. The autonomic support within that architecture is not.
How Is Dorsal Vagal Shutdown Different From Depression?
Many people with the dorsal vagal pattern are told they are depressed. The overlap exists: both present with fatigue, withdrawal, excessive sleep, and reduced engagement. A person can have both simultaneously.
The distinction matters because the approaches diverge. Depression-related hypersomnia often responds to serotonergic medication — SSRIs can improve both mood and sleep when the underlying mechanism is monoaminergic. Dorsal vagal shutdown may not respond to antidepressants because the mechanism is autonomic, not neurotransmitter-based.
How to recognize the difference: a person whose sleep study shows normal architecture, whose thyroid function is normal, whose depression screening is borderline, but whose heart rate variability is low and whose pattern looks like freeze-conservation — that points toward the dorsal vagal pathway.
Beutler et al. (2022) reviewed 28 studies encompassing approximately 1,300 participants examining dissociation and autonomic profiles. The review found no consistent trend across physiological markers in trauma-related dissociation. The observation that dissociation and dorsal vagal states overlap is well-established in polyvagal theory, but the psychophysiological evidence measured through heart rate, blood pressure, and skin conductance is more mixed than the theory predicts.
In practice: if the pattern is excessive sleep, low HRV, fatigue despite long sleep, and a freeze-conservation presentation — the dorsal vagal framework offers a coherent explanation regardless of whether dissociation is co-present. And if antidepressants are not improving the sleep pattern, the autonomic approach outlined in the next section targets a different mechanism.
What Does Gentle Reactivation From Dorsal Vagal Shutdown Look Like?
A person in dorsal vagal shutdown is already in parasympathetic conservation. Adding more parasympathetic input — deep breathing, body scans, progressive muscle relaxation — can deepen the shutdown rather than resolve it. The direction of recovery is toward engagement, not further into rest.
In polyvagal theory, the dorsal vagal conservation state and the ventral vagal social engagement state are mediated by different branches of the vagus nerve. Reactivation means stimulating the ventral branch — associated with social connection, facial expression, and vocal prosody — rather than the dorsal branch that is already dominant.
Orienting exercises. Slowly scanning the room, naming objects, noticing textures and colors. This engages the visual and attentional circuitry that connects to ventral vagal social engagement.
Bilateral engagement. Alternating tapping (sometimes called butterfly tap), alternating hand pressure, or slow side-to-side head movement. Bilateral engagement activates both hemispheres and can facilitate the state transition from freeze to engagement.
Vocalization. Humming, soft singing, or sustained vowel sounds. The pharyngeal and laryngeal muscles innervated by the ventral vagal branch are activated during vocalization — targeting social engagement circuitry rather than conservation circuitry.
Gradual sensory input. Cool water on the face or hands, textured objects, specific scents. Sensory engagement counters the dissociation and reduced sensory awareness that characterize the dorsal vagal state.
Slow, voluntary movement. Rocking, stretching, walking at a deliberately slow pace. Voluntary movement counters the immobilization that defines the dorsal vagal response. Vigorous exercise can trigger a sympathetic spike that the autonomic state responds to with another dorsal vagal crash — a cycle of exertion followed by collapse.
Recovery from chronic dorsal vagal shutdown is gradual — weeks to months of consistent practice. The autonomic state that encoded this conservation pattern did so over time, and the transition back to ventral vagal engagement takes time to stabilize. For the broader state-specific framework — including how these exercises map to the polyvagal three-state model and sleep — that article covers the full picture of autonomic state transitions and sleep.
Dorsal vagal shutdown may not be the only factor affecting your sleep. Autonomic hyperarousal, metabolic disruptions, inflammatory processes, or hormonal changes may also be contributing. When multiple causes overlap, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups
Frequently Asked Questions
Can You Have Both Hyperarousal Insomnia and Dorsal Vagal Shutdown?
van der Burg et al. (2025) studied 257 people with PTSD undergoing intensive trauma-focused therapy and measured tonic immobility (the freeze/shutdown response) during trauma re-experiencing. Baseline tonic immobility predicted higher post-PTSD scores, though this association became non-significant when dissociative tendencies were controlled — indicating that immobility and dissociation share overlapping variance, consistent with shared underlying processes. Tonic immobility scores decreased from pre- to post-therapy, and the magnitude of decrease correlated with PTSD reduction.
The study identified four groups by tonic immobility trajectory: high-stable, high-decrease, moderate, and low. People in the high-stable and moderate groups showed the poorest outcomes — a persistent tonic immobility pattern was associated with more resistant presentations.
If sleep problems alternate between periods of insomnia (unable to sleep) and hypersomnia (sleeping 12+ hours without restoration), the pattern may reflect this autonomic oscillation rather than two separate conditions. For more on the hyperarousal side, see What Is Hyperarousal Insomnia? Why You Are Wired but Tired Every Night.
Is Sleeping Too Much a Freeze Response?
When the autonomic state encodes a freeze pattern during a traumatic event, that pattern can persist as chronic dorsal vagal conservation — which includes prolonged, unrefreshing sleep.
de Souza Junior et al. (2024) followed healthcare workers exposed to COVID-19-related traumatic events over 6 to 12 months. Each one-unit increase in tonic immobility score was associated with a 1.5% increase in average PTSD score at follow-up. At threshold levels: significant tonic immobility conferred 3.5-fold increased likelihood of meeting probable PTSD criteria, and extreme tonic immobility conferred 7.3-fold increased likelihood.
When freeze becomes the default autonomic response, the dorsal vagal state becomes chronic — and the sleep pattern reflects that: long sleep, poor restoration, difficulty engaging, and fatigue that does not improve regardless of hours spent in bed.
If trauma and insomnia through the hyperarousal lens describes one side of the trauma-sleep relationship, dorsal vagal shutdown describes the other. They can co-exist in the same person, alternating or present simultaneously.
What Does Autonomic Collapse Look Like in Post-Viral Fatigue?
Post-viral fatigue conditions — long COVID and ME/CFS — produce a pattern that closely parallels dorsal vagal shutdown: excessive unrestorative sleep, persistent fatigue, cognitive impairment, and difficulty engaging with normal activity.
Novak et al. (2026) assessed 143 people with long COVID, 170 with ME/CFS, and 73 healthy controls. Autonomic impairment was detected in 95% of the long COVID group and 89% of the ME/CFS group. Small fiber neuropathy was identified in 67% and 53%, respectively.
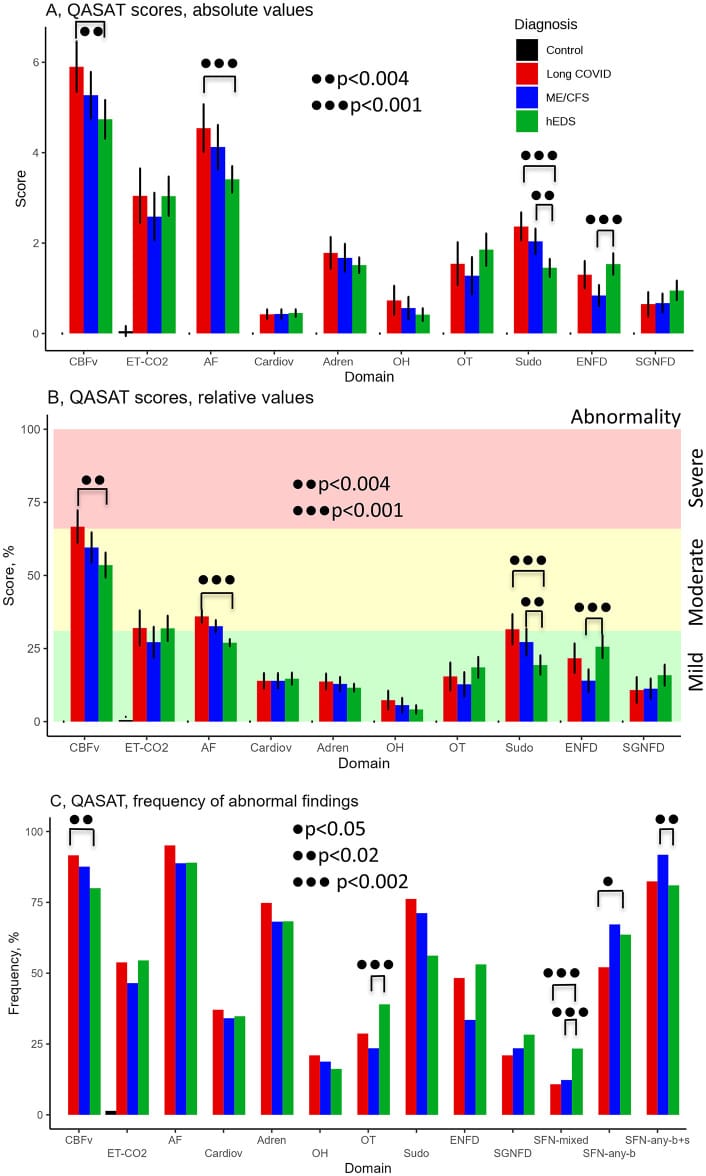
Issa et al. (2025) found comparable results in a multi-site US study: mean COMPASS-31 autonomic scores were 34.1 in 301 people with ME/CFS compared to 6.8 in 141 healthy controls — a five-fold difference.
The similarity across post-infectious conditions supports a shared autonomic mechanism. For people whose excessive-sleep-with-fatigue pattern began after a viral infection, the dorsal vagal framework may explain why standard fatigue workups do not capture what is happening.
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Does Lion’s Mane Affect Your Sleep? Why It Helps Some People and Keeps Others Awake — lion’s mane’s dual mechanism: neurotrophic activity, cholinergic activity, and sleep architecture
- Why Do Cholinergic Supplements Give You Vivid Dreams? — the acetylcholine-REM pathway behind supplement-related vivid dreams
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — how post-viral autonomic disruption can affect sleep continuity
- Why Does Your Heart Race at Night After COVID? What the Vagus Nerve Has to Do With It — why post-COVID autonomic instability can show up as nighttime tachycardia
- Can POTS and Dysautonomia Cause Insomnia? Why Your Autonomic Nervous System Will Not Let You Rest — how orthostatic intolerance and autonomic instability can fragment sleep
- Can a Viral Infection Cause Chronic Insomnia? — how viral infections can disrupt autonomic regulation and sleep
- Why Will Your Nervous System Not Let You Sleep? Polyvagal Theory and Insomnia — how polyvagal states map to insomnia, hyperarousal, and unrefreshing sleep
- Why Does Trauma Cause Insomnia? How Your Nervous System Stays on Guard at Night — how trauma can keep sleep shaped by hyperarousal and vigilance
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- How Do Probiotics Reach Your Brain? The Vagotomy Evidence — what vagotomy studies show about gut-brain communication
- Does Poor Sleep Damage Your Gut Microbiome? — the relationship between sleep fragmentation, microbiome disruption, and inflammatory load
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
Beutler, S., Mertens, Y. L., Ladner, L., Schellong, J., Croy, I., & Daniels, J. K. (2022). Trauma-related dissociation and the autonomic nervous system: a systematic literature review of psychophysiological correlates of dissociative experiencing in PTSD patients. European Journal of Psychotraumatology, 13(2), 2132599. https://pubmed.ncbi.nlm.nih.gov/36340007/
de Souza Junior, S., Monteiro Fabricio Gama, C., Menezes Goncalves, R., Lorrany Campos Guerra, T., Volchan, E., Erthal, F. S., Mocaiber, I., de Paula Antunes David, I., Catarina Lima Portugal, L., Mendlowicz, M. V., Berger, W., de Oliveira, L., & Garcia Pereira, M. (2024). Tonic immobility triggered by COVID-19-related trauma is associated with long-term PTSD symptoms. Journal of Anxiety Disorders, 105, 102894. https://pubmed.ncbi.nlm.nih.gov/38959538/
Fatt, S. J., Beilharz, J. E., Joubert, M., Wilson, C., Lloyd, A. R., Vollmer-Conna, U., & Cvejic, E. (2020). Parasympathetic activity is reduced during slow-wave sleep, but not resting wakefulness, in patients with chronic fatigue syndrome. Journal of Clinical Sleep Medicine, 16(1), 19-28. https://pubmed.ncbi.nlm.nih.gov/31957647/
Issa, A., Lin, J.-M. S., Chen, Y., Attell, J., Brimmer, D., Bertolli, J., Natelson, B. H., Lapp, C. W., Podell, R. N., Kogelnik, A. M., Klimas, N. G., Peterson, D. L., Bateman, L., & Unger, E. R. (2025). Autonomic dysfunction in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): Findings from the multi-site clinical assessment of ME/CFS (MCAM) study in the USA. Journal of Clinical Medicine, 14(17), 6269. https://pubmed.ncbi.nlm.nih.gov/40944028/
Miglis, M. G., Schneider, L., Kim, P., Cheung, J., & Trotti, L. M. (2020). Frequency and severity of autonomic symptoms in idiopathic hypersomnia. Journal of Clinical Sleep Medicine, 16(5), 749-756. https://pubmed.ncbi.nlm.nih.gov/32039754/
Mombelli, S., Deshaies-Rugama, A.-S., Blais, H., Sekerovic, Z., Thompson, C., Desautels, A., Montplaisir, J., Nigam, M., Moderie, C., Carrier, J., & Gosselin, N. (2025). Unrefreshing naps and sleep architecture during the multiple sleep latency test in idiopathic hypersomnia. Journal of Sleep Research, 34(1), e14261. https://pubmed.ncbi.nlm.nih.gov/38859728/
Novak, P., Systrom, D. M., Witte, A., Marciano, S. P., Felsenstein, D., Milunsky, J. M., Milunsky, A., Krier, J., & Fishman, M. C. (2026). Shared autonomic phenotype of long COVID and myalgic encephalomyelitis/chronic fatigue syndrome. PLoS ONE, 21(1), e0341278. https://pubmed.ncbi.nlm.nih.gov/41576003/
Roelofs, K. (2017). Freeze for action: neurobiological mechanisms in animal and human freezing. Philosophical Transactions of the Royal Society B: Biological Sciences, 372(1718), 20160206. https://pubmed.ncbi.nlm.nih.gov/28242739/
van der Burg, N. C., Hagenaars, M. A., de Jongh, A., & van Minnen, A. (2025). The role of tonic immobility during re-experiencing trauma in PTSD treatment. Journal of Anxiety Disorders, 115, 103059. https://pubmed.ncbi.nlm.nih.gov/40882493/
Written by Kat Fu, M.S., M.S. — Last reviewed: May 2026 — 9 references cited
