Waking at 2 or 3 AM with a pounding heart, drenched in sweat, unable to fall back asleep — this is one of the more commonly reported post-COVID experiences. Many people assume the problem is cardiac. The evidence points to an autonomic one: impaired vagal tone that does not suppress heart rate during sleep the way it should.
This article covers why nighttime tachycardia occurs after COVID, how the normal nocturnal catecholamine suppression breaks down, and when a cardiologist visit matters versus when the picture points to autonomic evaluation. For the broader autonomic framework, see the autonomic sleep disruption pillar.
Why Does Your Heart Race When You Lie Down to Sleep After COVID?
Dodt et al. (1997) established the normal nocturnal catecholamine profile by continuously sampling plasma epinephrine and norepinephrine every 7 minutes in healthy men throughout the night. Both reached their lowest concentrations during REM sleep — lower than in any other sleep stage. Heart rate and blood pressure tracked this suppression in parallel.
This is the baseline that post-COVID autonomic impairment disrupts.
Mooren et al. (2023) enrolled 103 people with post-COVID condition at 252 days post-infection and compared them with 103 people with coronary artery disease and 90 healthy controls using 24-hour HRV monitoring. Frequency-domain HRV measures — LF/HF power, LF/HFnu, and LF/HF ratio — differed between groups at p less than or equal to 0.001. The post-COVID group showed impaired parasympathetic activity at night. Those who required hospitalization during acute infection showed more pronounced sympathetic overactivation across the full 24-hour period.
The magnitude of these HRV changes in post-COVID was comparable to what the researchers observed in the coronary artery disease comparison group — persistent sympathovagal imbalance at eight months post-infection.
A 2026 meta-analysis by Schoene et al. synthesized 11 studies encompassing 1,162 participants (593 with post-COVID condition, 565 healthy controls). The analysis found consistent trends toward reduced HRV: SDNN effect size 0.26 (95% CI: -0.03 to 0.56), rMSSD effect size 0.11 (95% CI: -0.15 to 0.36), LF/HF ratio effect size -0.271 (95% CI: -0.61 to 0.07). Heterogeneity was moderate-to-high across studies, but the direction was consistent: a trend toward reduced HRV, particularly in parasympathetic markers.
Is Post-COVID Nighttime Tachycardia a Heart Problem or an Autonomic Problem?
The first question many people ask after weeks of nighttime heart racing is whether something is wrong with the heart itself. In many post-COVID cases, the answer is no — the heart’s structure and electrical conduction are intact. The problem is what is driving it.
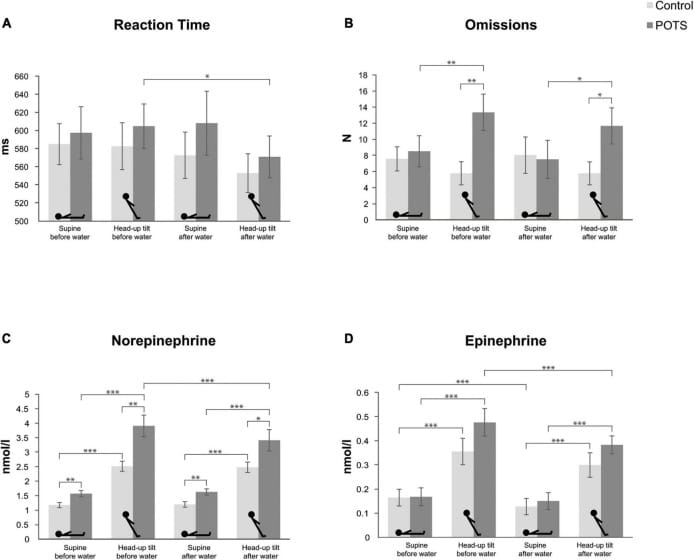
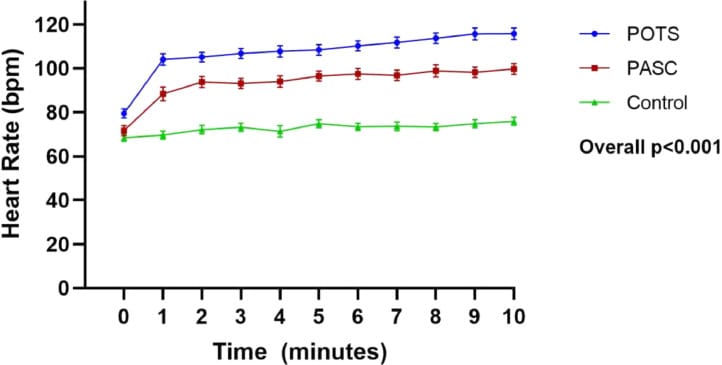
Seeley et al. (2025) prospectively enrolled 33 people with post-acute COVID-19 sequelae (PASC), 33 with established POTS, and 33 healthy controls. Median age of 32 years, 85.9% female. Seventy-nine percent of the PASC group met internationally established criteria for POTS during formal autonomic testing. Both PASC and POTS groups showed reduced respiratory sinus arrhythmia compared to healthy controls (P < .001) — reflecting impaired cardiac vagal tone, the same mechanism that does not suppress heart rate during sleep.
Heart rate increase during 10-minute active standing was greater in both groups compared to controls (P < .001), consistent with excessive sympathetic recruitment. COMPASS-31 autonomic scores were elevated across all subdomains (P < .001), confirming broad multi-domain autonomic involvement beyond palpitations alone.
Fedorowski et al. (2024) in Nature Reviews Cardiology established that cardiovascular autonomic impairment might affect one-third of highly symptomatic post-COVID cases. POTS and inappropriate sinus tachycardia are the two prevalent autonomic phenotypes. Proposed mechanisms discussed in the review include autoantibodies against adrenergic and muscarinic receptors, viral injury to autonomic ganglia, persistent neuroinflammation, and renin-angiotensin dysregulation.
This matters because the standard cardiac workup — echocardiogram, Holter monitor, troponin — is designed to detect structural and electrical problems. When those come back normal, many people are told nothing is wrong. What is often missing is formal autonomic testing: tilt-table evaluation, COMPASS-31 scoring, and heart rate variability analysis.
Bonnet and Arand (2010) established the hyperarousal model of insomnia: persistent heightened activation — abnormal hormone secretion, elevated catecholamines, increased metabolic rate, and increased high-frequency EEG activation during sleep. Post-COVID adds an organic injury (vagal damage) to this hyperarousal model, producing a nocturnal environment where sympathetic activity remains elevated and cannot be suppressed.
When a cardiac evaluation matters: Myocarditis, pericarditis, and new arrhythmias can occur after COVID and require echocardiography and cardiac MRI. Chest pain with exertion, syncope, or new irregular rhythms — as opposed to regular fast heart rate — are reasons to pursue cardiac imaging alongside autonomic evaluation.
What Happens to Your Autonomic Nervous System During Sleep After COVID?
The transition from wakefulness to sleep requires active parasympathetic engagement: the vagus nerve increases its output, heart rate drops, blood pressure falls, and catecholamine secretion declines. When this transition is incomplete — when sympathetic activity remains elevated at sleep onset — heart rate stays up, alertness persists, and consolidated sleep does not happen.
Qin et al. (2025) used short-duration ECG-based HRV recordings in 173 participants and found that young adults under 25 with long COVID showed lower HRV in the very-low-frequency domain (p = 0.012). The long COVID group had a higher proportion of participants with aberrant autonomic neuroactivity compared to those who recovered from COVID-19 without persistent symptoms (p = 0.048). This was in people who had mild-to-moderate COVID-19 — not those who were hospitalized or critically ill. The changes appeared within approximately one month of infection.
Eastin et al. (2025) quantified the chronic burden in a cohort of 526 adults with long COVID. Seventy-one point nine percent scored 20 or above on the COMPASS-31 autonomic questionnaire, meeting the threshold for moderate-to-severe autonomic impairment. Median duration was 36 months (IQR: 30-40 months). Thirty-seven point five percent could no longer work or had to drop out of school. Among those with confirmed autonomic impairment, 40.5% received new POTS identification.
Rodriguez et al. (2022) tested the norepinephrine mechanism in 13 people with neuropathic POTS versus 15 healthy controls. POTS participants demonstrated elevated plasma norepinephrine concentrations independent of body position — meaning sympathetic overactivation was not purely postural but constitutively elevated. Upon standing, norepinephrine rose excessively beyond the normal orthostatic response. A 500 ml water bolus produced measurable reductions in plasma norepinephrine and heart rate, demonstrating that the hyperarousal state is dynamically modifiable.
This norepinephrine-mediated arousal state parallels the post-COVID nocturnal pattern: excessive catecholamine secretion prevents the parasympathetic engagement that sleep onset requires. The result is fragmented sleep, repeated awakenings with pounding heart rate, and inadequate overnight recovery.
For a deeper look at the vagal nerve damage itself, see How COVID damages the vagus nerve and why it affects sleep. For the POTS-related picture, see POTS, dysautonomia, and sleep after COVID. And for the broader post-viral pattern beyond COVID, see post-viral insomnia and nervous system disruption.
What Helps Post-COVID Nighttime Heart Racing and Sleep Disruption?
Treadwell et al. (2025) conducted a scoping review synthesizing 11 effectiveness studies, 11 guidelines, and 15 ongoing trials on treatments for long COVID autonomic impairment. The authors note that existing effectiveness studies “are often small and uncontrolled, making it unclear whether the observed pre-post changes were due solely to the administered treatments.” With that calibration in mind, here is what the evidence covers.
Non-pharmacological approaches:
- Slow-exhale breathing — extended exhalation (4-second inhale, 6-8-second exhale) activates vagal afferents and increases parasympathetic output. This can be done at bedtime and during nocturnal wakeups.
- Cool sleeping environment — ambient temperature of 65-68 degrees Fahrenheit (18-20 degrees Celsius) reduces sympathetic load. Post-COVID autonomic impairment makes thermoregulation less efficient, increasing sensitivity to warmth-driven sympathetic activation at night.
- Elevated head positioning — raising the head of the bed 10-15 degrees reduces venous pooling, which is relevant for people with POTS overlap.
- Compression garments — abdominal and lower-extremity compression can reduce blood pooling and blunt the orthostatic heart rate surge.
- Increased fluid and salt intake — expands plasma volume, reducing the compensatory tachycardia that accompanies low circulating volume. Consensus guidelines support this as a first-line approach.
Supplements with parasympathetic support:
- Magnesium glycinate — supports parasympathetic tone without sedation. May help reduce nocturnal heart rate in people with low magnesium status.
- L-theanine — promotes alpha-wave activity and has calming effects without sedation. May support the sympathetic-to-parasympathetic transition at sleep onset.
Pharmacological options (discussed with a physician):
- Low-dose beta-blockers — reduce heart rate by blocking beta-adrenergic receptors. Used in POTS management at lower doses than cardiac indications.
- Ivabradine — a selective sinus node inhibitor that reduces heart rate without affecting blood pressure. Among the frequently investigated candidate therapies in ongoing long COVID trials.
- Fludrocortisone — expands plasma volume for POTS overlap.
Monitoring and measurement:
Overnight heart rate data from wearables can help characterize the pattern and track whether approaches produce measurable changes. TLV covers how to interpret overnight HRV data in a separate article on HRV and vagal tone. For vagus nerve stimulation devices, see TLV’s coverage of transcutaneous VNS for insomnia.
Post-COVID autonomic disruption may not be the only factor affecting your sleep. GABA changes, metabolic disruptions, inflammatory processes, or circadian misalignment may also be contributing. When multiple causes overlap, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
How Long Do Post-COVID Heart Palpitations Last?
The 36-month median means half of the 526 participants had durations longer than three years at follow-up. Whether this represents permanent change or slow recovery with eventual resolution is not yet established from the available data.
Can Post-COVID Nighttime Tachycardia Happen After Mild COVID?
Qin et al. (2025) enrolled people with mild-to-moderate infections. Young adults under 25 showed reduced HRV in the very-low-frequency domain (p = 0.012) — broadening the at-risk population beyond the severe-illness cohorts that dominated early research.
Should You Wear a Heart Rate Monitor to Bed After COVID?
A heart rate that stays elevated above daytime resting levels during the first 90 minutes of sleep, or spikes that coincide with awakenings at 2-3 AM, are consistent with the sympathetic dominance pattern described throughout this article. Wearable data does not replace autonomic testing, but it provides a longitudinal view that office-based tests cannot. For a guide on wearable accuracy, see TLV’s coverage of sleep tracker accuracy.
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Does Lion’s Mane Affect Your Sleep? Why It Helps Some People and Keeps Others Awake — lion’s mane’s dual mechanism: neurotrophic activity, cholinergic activity, and sleep architecture
- Why Do Cholinergic Supplements Give You Vivid Dreams? — the acetylcholine-REM pathway behind supplement-related vivid dreams
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — how post-viral autonomic disruption can affect sleep continuity
- Can POTS and Dysautonomia Cause Insomnia? Why Your Autonomic Nervous System Will Not Let You Rest — how orthostatic intolerance and autonomic instability can fragment sleep
- Can a Viral Infection Cause Chronic Insomnia? — how viral infections can disrupt autonomic regulation and sleep
- Why Will Your Nervous System Not Let You Sleep? Polyvagal Theory and Insomnia — how polyvagal states map to insomnia, hyperarousal, and unrefreshing sleep
- Why Does Trauma Cause Insomnia? How Your Nervous System Stays on Guard at Night — how trauma can keep sleep shaped by hyperarousal and vigilance
- Why Are You Sleeping 12 Hours and Still Exhausted? The Dorsal Vagal Shutdown Explained — the dorsal vagal pattern behind long sleep that still feels unrefreshing
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- How Do Probiotics Reach Your Brain? The Vagotomy Evidence — what vagotomy studies show about gut-brain communication
- Does Poor Sleep Damage Your Gut Microbiome? — the relationship between sleep fragmentation, microbiome disruption, and inflammatory load
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
- Bonnet, M. H., & Arand, D. L. (2010). Hyperarousal and insomnia: state of the science. Sleep Medicine Reviews, 14(1), 9-15. https://pubmed.ncbi.nlm.nih.gov/19640748/
- Dodt, C., Breckling, U., Derad, I., Fehm, H. L., & Born, J. (1997). Plasma epinephrine and norepinephrine concentrations of healthy humans associated with nighttime sleep and morning arousal. Hypertension, 30(1 Pt 1), 71-76. https://pubmed.ncbi.nlm.nih.gov/9231823/
- Eastin, E. F., Machnik, J. V., Stiles, L. E., Larsen, N. W., Seliger, J., Geng, L. N., Bonilla, H., Yang, P. C., & Miglis, M. G. (2025). Chronic autonomic symptom burden in long-COVID: a follow-up cohort study. Clinical Autonomic Research, 35(3), 453-464. https://pubmed.ncbi.nlm.nih.gov/39907931/
- Fedorowski, A., Fanciulli, A., Raj, S. R., Sheldon, R., Shibao, C. A., & Sutton, R. (2024). Cardiovascular autonomic dysfunction in post-COVID-19 syndrome: a major health-care burden. Nature Reviews Cardiology, 21(6), 379-395. https://pubmed.ncbi.nlm.nih.gov/38163814/
- Mooren, F. C., Bockelmann, I., Waranski, M., Kotewitsch, M., Teschler, M., Schafer, H., & Schmitz, B. (2023). Autonomic dysregulation in long-term patients suffering from Post-COVID-19 Syndrome assessed by heart rate variability. Scientific Reports, 13(1), 15814. https://pubmed.ncbi.nlm.nih.gov/37739977/
- Qin, M., Lee, K., & Yoo, S. J. (2025). The impact of long COVID on heart rate variability: a cross-sectional study. BMC Infectious Diseases, 25(1), 261. https://pubmed.ncbi.nlm.nih.gov/39994668/
- Rodriguez, B., Hochstrasser, A., Eugster, P. J., Grouzmann, E., Muri, R. M., & Z’Graggen, W. J. (2022). Brain fog in neuropathic postural tachycardia syndrome may be associated with autonomic hyperarousal and improves after water drinking. Frontiers in Neuroscience, 16, 968725. https://pubmed.ncbi.nlm.nih.gov/35992935/
- Schoene, D., Deckert, S., Barlinn, K., Huttner, H. B., Kosters, M., & Siepmann, T. (2026). Neurocardiac Autonomic Dysfunction in Patients With Post-COVID-19 Condition: A Systematic Review and Meta-Analysis. European Journal of Neurology, 33(3), e70561. https://pubmed.ncbi.nlm.nih.gov/41814525/
- Seeley, M. C., Gallagher, C., Ong, E., Langdon, A., Chieng, J., Bailey, D., Page, A., Lim, H. S., & Lau, D. H. (2025). High Incidence of Autonomic Dysfunction and Postural Orthostatic Tachycardia Syndrome in Patients with Long COVID. The American Journal of Medicine, 138(2), 354-361.e1. https://pubmed.ncbi.nlm.nih.gov/37391116/
- Treadwell, J. R., Wagner, J., Reston, J. T., Phillips, T., Hedden-Gross, A., & Tipton, K. N. (2025). Treatments for Long COVID autonomic dysfunction: a scoping review. Clinical Autonomic Research, 35(1), 5-29. https://pubmed.ncbi.nlm.nih.gov/39658729/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 10 references cited
