Trauma survivors are routinely told to practice sleep hygiene — consistent bedtime, dark room, no screens. For many, this is insufficient. The insomnia persists because its origin is autonomic, not behavioral. The nervous system, having learned that the environment is dangerous, maintains vigilance during sleep. Sleep hygiene assumes a nervous system that can downregulate when conditions are right. After trauma, the nervous system does not downregulate.
This article covers the autonomic and neuroendocrine mechanisms through which trauma produces insomnia, the prevalence data establishing insomnia as a core feature of PTSD, why standard sleep advice is insufficient for this population, and which approaches target the nervous system origin. For the full autonomic model, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It. Trauma-induced autonomic sensitization is one of several causes covered in that pillar; this article goes deeper on the trauma-specific pathway.
How Does Trauma Change the Nervous System’s Relationship With Sleep?
Van Liempt et al. (2013) enrolled 45 participants — 13 combat veterans with PTSD, 17 trauma-exposed controls, and 15 healthy controls — and monitored sleep using full polysomnography with blood sampling every 20 minutes throughout the night. The PTSD group had elevated heart rate compared to both control groups, confirming sustained sympathetic nervous system activation during sleep. Nocturnal ACTH and cortisol concentrations during the first half of the night were inversely correlated with slow-wave sleep duration — the higher the nocturnal stress hormones, the less deep sleep (van Liempt et al., 2013).
This is not the same mechanism as conditioned insomnia. In conditioned insomnia, the bedroom becomes an arousal cue — the person associates the sleep environment with wakefulness, and the arousal is context-dependent. In trauma-related insomnia, the entire autonomic nervous system is sensitized. The arousal is constitutive. It persists regardless of environment because it originates from HPA axis sensitization and autonomic memory.
Inslicht et al. (2018) tested this causal link directly. Their study compared 33 people with chronic PTSD and 33 matched controls over three laboratory nights with polysomnography. Metyrapone — a drug that blocks cortisol synthesis and disinhibits ACTH release — produced a greater ACTH increase and greater decrease in delta sleep in the PTSD group than in controls. Within the PTSD group, increased ACTH response correlated with decreased delta power. That correlation was absent in healthy controls (Inslicht et al., 2018). When the HPA axis is pharmacologically provoked, PTSD produces a larger deep sleep deficit — evidence that HPA sensitization causally degrades restorative sleep.
Kobayashi et al. (2014) documented the other side of the equation: parasympathetic insufficiency. They compared 20 people with current PTSD and 18 trauma-resilient individuals (trauma-exposed but never developed PTSD) using overnight electrocardiography. Normalized high-frequency heart rate variability — a measure of cardiac parasympathetic activity — was lower in the PTSD group throughout the time-in-bed period (p = .041). In the resilient group, total sleep time correlated with autonomic measures (r = -.72 and .75), indicating healthy coupling between sleep and nocturnal autonomic regulation. That coupling was absent in the PTSD group (Kobayashi et al., 2014). The nervous system that developed PTSD does not show the normal parasympathetic recovery during sleep that the trauma-resilient nervous system maintains.
How Common Is Insomnia in People With Post-Traumatic Stress?
Ahmadi et al. (2022) synthesized 75 studies with a combined total of more than 573,000 participants. Across 44 studies with 57,618 participants, the correlation between insomnia and PTSD reached a medium effect size (ES = 0.52, CI: 0.47-0.57). In the prevalence analysis, 63% of people with PTSD or post-traumatic stress had insomnia (CI: 45-78%). Military service as the primary trauma source and the COVID-19 pandemic context amplified the PTSD-insomnia relationship (Ahmadi et al., 2022).
63% prevalence makes insomnia more common than avoidance or re-experiencing in many PTSD populations. This is not a secondary complaint. It is a defining feature.
Sinha (2016) proposed trauma-induced insomnia as a distinct entity — separable from both primary insomnia and PTSD. The model positions trauma-mediated sensitization of central nervous system arousal circuits as the driver. Trauma-induced insomnia can develop and persist without meeting full criteria for PTSD (Sinha, 2016). This matters because it means that insomnia may need separate attention even when other PTSD features improve. The autonomic sensitization that drives the sleep disruption can outlast the other trauma responses.
Why Does Standard Sleep Advice Fail After Trauma?
Sleep hygiene works when the barrier is behavioral. When the person reduces caffeine, limits screen time, and maintains a consistent schedule, a nervous system capable of parasympathetic transition will respond. The conditions enable the transition.
After trauma, the barrier is autonomic. The HPA axis is sensitized. Sympathetic activation remains elevated during sleep — not at sleep onset, but throughout the night. Van Liempt et al. (2013) documented this with blood sampling every 20 minutes: heart rate remains elevated and ACTH levels correlate with sleep fragmentation throughout the night, not just at the transition point. The arousal is ongoing, independent of what the bedroom looks like or what the pre-bed routine includes.
In polyvagal terms, the nervous system’s neuroception — its continuous below-conscious-awareness assessment of safety — is calibrated to “danger.” It will not permit the ventral vagal state that supports rest, regardless of how optimized the sleep environment is. How polyvagal states determine whether your nervous system allows sleep covers this three-state framework in detail.
The persistent insomnia is the expected outcome of a sensitized autonomic nervous system encountering conditions — darkness, stillness, vulnerability — that the post-trauma nervous system reads as danger-relevant.
What Approaches Target the Autonomic Origin of Trauma-Related Insomnia?
How Does Transcutaneous Vagus Nerve Stimulation Affect Trauma-Related Sleep?
Bottari et al. (2025) enrolled 21 veterans with PTSD and tested nine tVNS parameter combinations against sham stimulation using overnight polysomnography. The optimal combination — 20 Hz frequency, 100 microsecond pulse width, 80% discomfort threshold — improved slow-wave sleep percentage compared to sham. The same parameters reduced next-day startle response (Bottari et al., 2025). Improved autonomic regulation during sleep carried over into reduced daytime hyperarousal. Can Vagus Nerve Stimulation Devices Improve Insomnia? covers the broader tVNS evidence for sleep.
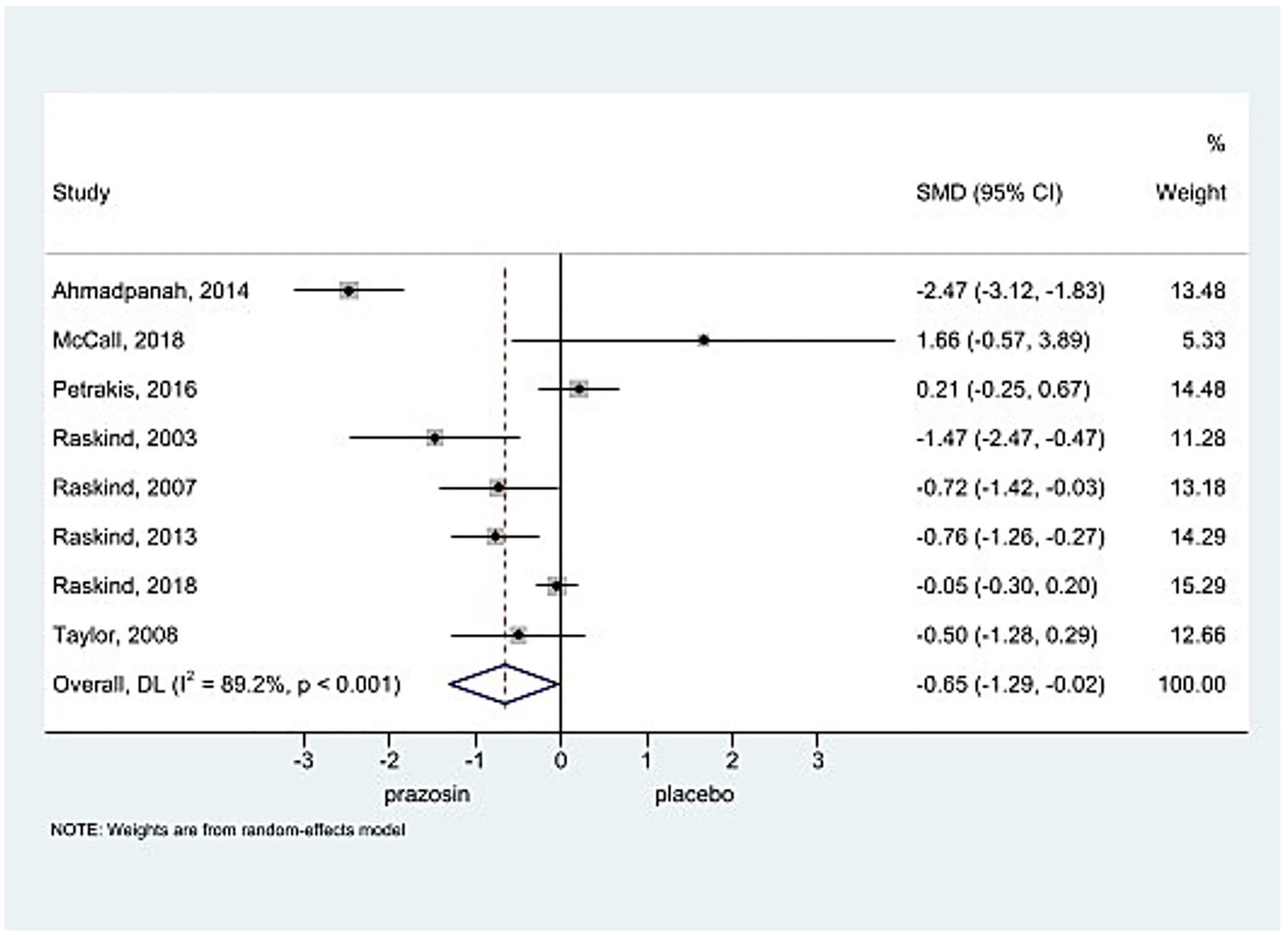
How Does Prazosin Target Nocturnal Noradrenergic Activation?
Prazosin blocks alpha-1 adrenergic receptors, reducing noradrenergic activation during sleep. Lappas et al. (2024) conducted the largest pharmacotherapy network meta-analysis for PTSD sleep — 99 RCTs with 10,481 participants. Prazosin may be the most effective treatment: insomnia (SMD = -0.88, 95% CI: -1.22 to -0.54), nightmares (SMD = -0.44, 95% CI: -0.84 to -0.04), and overall sleep quality (SMD = -0.55, 95% CI: -1.01 to -0.10). Evidence was scarce and indicated lack of efficacy for SSRIs, mirtazapine, z-drugs, and benzodiazepines for PTSD-related sleep (Lappas et al., 2024).
Mendes et al. (2025) analyzed moderators across 10 RCTs with 648 participants. Prazosin improved insomnia (SMD = -0.654, p = 0.043) and nightmare severity (SMD = -0.641, p = 0.025), but did not improve overall PTSD scores (SMD = -0.428, p = 0.077) — confirming that its mechanism is specific to nocturnal noradrenergic activity rather than global PTSD (Mendes et al., 2025).
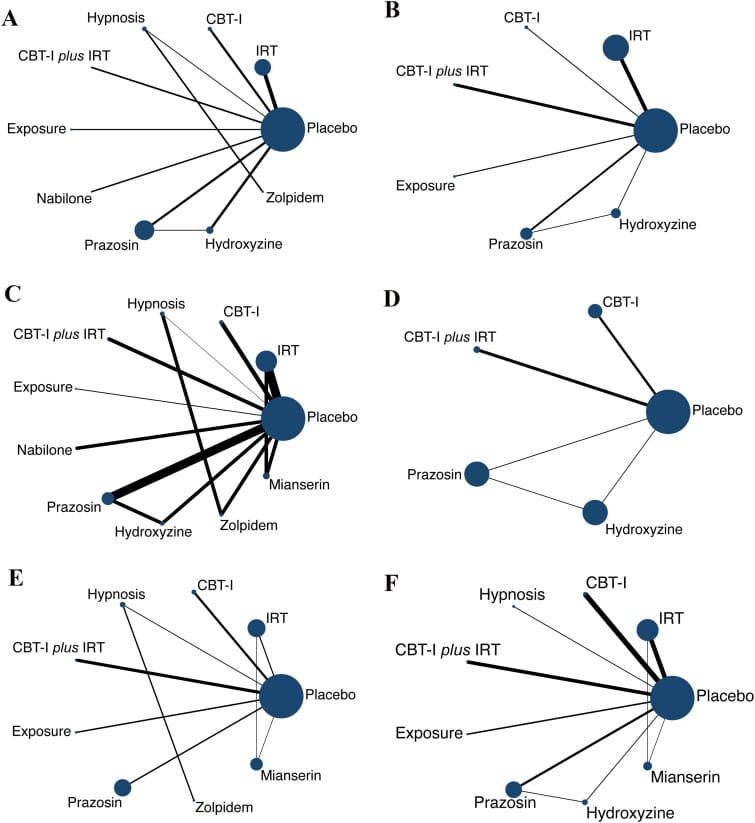
How Does Cognitive Behavioral Therapy for Insomnia Compare to Pharmacological Approaches for Post-Traumatic Stress Sleep?
Huang et al. (2024) compared psychotherapy and pharmacotherapy across 24 RCTs with 1,647 participants. CBT-I produced the largest improvement in sleep quality of any approach (SMD = -5.61, 95% CI: -8.82 to -2.40 versus placebo). For nightmare severity, prazosin outperformed all other agents (SMD = -1.20). CBT-I combined with imagery rehearsal therapy achieved comparable PTSD severity reduction (SMD = -1.71) (Huang et al., 2024).
The evidence supports matching the approach to the primary complaint: CBT-I for insomnia and hyperarousal reconditioning, prazosin for nightmares driven by noradrenergic activation, tVNS for autonomic restoration. Extended exhale breathing and somatic grounding address autonomic activation during wakefulness — the polyvagal framework for nervous system state regulation covers these in context.
Trauma-related autonomic sensitization may not be the only factor affecting your sleep. Metabolic disruptions, inflammatory processes, hormonal changes, or circadian misalignment may also be contributing. When multiple causes overlap, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Is Trauma-Related Insomnia Different From Regular Insomnia?
The distinction matters for choosing an approach. Conditioned insomnia responds to stimulus control and sleep restriction — reconditioning the association between the bedroom and sleep. Trauma-related insomnia involves a nervous system that is not responding to environmental cues in the first place. The arousal originates from HPA axis and sympathetic nervous system sensitization that persists independently of context. Approaches that address only the conditioned component may improve sleep onset but leave the autonomic sensitization — and the nocturnal HPA axis dysregulation and sympathetic activation it produces — intact.
Can Trauma-Related Insomnia Be Resolved Without Addressing the Trauma Itself?
Prazosin targets norepinephrine at the receptor level. tVNS modulates autonomic balance through vagal afferent stimulation. Neither requires processing the traumatic memory. CBT-I reconditions sleep behaviors and addresses hyperarousal cognitions. Each of these produces measurable improvement in sleep metrics — the evidence from Lappas et al. (2024) and Huang et al. (2024) confirms this across thousands of participants.
Where the approaches have limits is in the underlying sensitization. The HPA axis and sympathetic nervous system calibration that trauma produced may persist even as sleep improves. For cases where autonomic sensitization is entrenched — where insomnia returns when the pharmacological or behavioral approach is discontinued — addressing the trauma origin alongside the sleep disruption produces more durable outcomes.
Does Childhood Trauma Affect Sleep Differently Than Adult-Onset Trauma?
When trauma occurs during development, the HPA axis calibrates to a dangerous environment as its baseline. Cortisol patterning, sympathetic tone, and parasympathetic capacity are shaped during a period when the nervous system is establishing its baseline norms. Lawrence and Scofield (2024) reviewed the downstream consequences: PTSD-associated HPA sensitization drives cardiovascular disease, metabolic impairment, autoimmune conditions, and neurocognitive decline across the lifespan. Women with PTSD bear a disproportionate burden of reproductive health consequences driven by HPA-immune interactions (Lawrence & Scofield, 2024).
For sleep, the implication is that childhood-origin autonomic sensitization may not respond as readily to approaches that work for adult-onset trauma. A nervous system that was shaped by danger from the beginning has a deeper calibration to undo than one that was sensitized by a discrete event in adulthood. This does not mean the sensitization is permanent — autonomic tone is responsive to sustained change — but the timeline tends to be longer, and multiple approaches working together tend to produce better results than any single modality. The dorsal vagal state and sleep shutdown in complex trauma covers the autonomic collapse pattern that can accompany prolonged developmental stress.
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Does Lion’s Mane Affect Your Sleep? Why It Helps Some People and Keeps Others Awake — lion’s mane’s dual mechanism: neurotrophic activity, cholinergic activity, and sleep architecture
- Why Do Cholinergic Supplements Give You Vivid Dreams? — the acetylcholine-REM pathway behind supplement-related vivid dreams
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — how post-viral autonomic disruption can affect sleep continuity
- Why Does Your Heart Race at Night After COVID? What the Vagus Nerve Has to Do With It — why post-COVID autonomic instability can show up as nighttime tachycardia
- Can POTS and Dysautonomia Cause Insomnia? Why Your Autonomic Nervous System Will Not Let You Rest — how orthostatic intolerance and autonomic instability can fragment sleep
- Can a Viral Infection Cause Chronic Insomnia? — how viral infections can disrupt autonomic regulation and sleep
- Why Will Your Nervous System Not Let You Sleep? Polyvagal Theory and Insomnia — how polyvagal states map to insomnia, hyperarousal, and unrefreshing sleep
- Why Are You Sleeping 12 Hours and Still Exhausted? The Dorsal Vagal Shutdown Explained — the dorsal vagal pattern behind long sleep that still feels unrefreshing
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- How Do Probiotics Reach Your Brain? The Vagotomy Evidence — what vagotomy studies show about gut-brain communication
- Does Poor Sleep Damage Your Gut Microbiome? — the relationship between sleep fragmentation, microbiome disruption, and inflammatory load
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
Ahmadi, R., Rahimi-Jafari, S., Olfati, M., Javaheripour, N., Emamian, F., Ghadami, M. R., Khazaie, H., Knight, D. C., Tahmasian, M., & Sepehry, A. A. (2022). Insomnia and post-traumatic stress disorder: A meta-analysis on interrelated association (n = 57,618) and prevalence (n = 573,665). Neuroscience and Biobehavioral Reviews, 141, 104850. https://pubmed.ncbi.nlm.nih.gov/36058403/
Bottari, S. A., Trifilio, E. R., Rohl, B., Wu, S. S., Miller-Sellers, D., Waldorff, I., Hadigal, S., Jaffee, M. S., Ferri, R., Lamb, D. G., Porges, E. C., & Williamson, J. B. (2025). Optimizing transcutaneous vagus nerve stimulation parameters for sleep and autonomic function in veterans with posttraumatic stress disorder with or without mild traumatic brain injury. Sleep, 48(8), zsaf152. https://pubmed.ncbi.nlm.nih.gov/40489292/
Huang, C. Y., Zhao, Y. F., Zhang, Z. X., Liu, R. B., Liu, J. L., Li, X. Z., Luo, J., Yue, L., & Zhang, C. (2024). Psychotherapeutic and pharmacological agents for post-traumatic stress disorder with sleep disorder: Network meta-analysis. Annals of Medicine, 56(1), 2381696. https://pubmed.ncbi.nlm.nih.gov/39061119/
Inslicht, S. S., Rao, M. N., Richards, A., O’Donovan, A., Gibson, C. J., Baum, T., Metzler, T. J., & Neylan, T. C. (2018). Sleep and hypothalamic pituitary adrenal axis responses to metyrapone in posttraumatic stress disorder. Psychoneuroendocrinology, 88, 136-143. https://pubmed.ncbi.nlm.nih.gov/29268182/
Kobayashi, I., Lavela, J., & Mellman, T. A. (2014). Nocturnal autonomic balance and sleep in PTSD and resilience. Journal of Traumatic Stress, 27(6), 712-716. https://pubmed.ncbi.nlm.nih.gov/25403523/
Lappas, A. S., Glarou, E., Polyzopoulou, Z. A., Goss, G., Huhn, M., Samara, M. T., & Christodoulou, N. G. (2024). Pharmacotherapy for sleep disturbances in post-traumatic stress disorder (PTSD): A network meta-analysis. Sleep Medicine, 119, 467-479. https://pubmed.ncbi.nlm.nih.gov/38795401/
Lawrence, S., & Scofield, R. H. (2024). Post traumatic stress disorder associated hypothalamic-pituitary-adrenal axis dysregulation and physical illness. Brain, Behavior, & Immunity – Health, 41, 100849. https://pubmed.ncbi.nlm.nih.gov/39280087/
Mendes, T. P., Pereira, B. G., Coutinho, E. S. F., Melani, M. S., Neylan, T. C., & Berger, W. (2025). Factors impacting prazosin efficacy for nightmares and insomnia in PTSD patients – a systematic review and meta-regression analysis. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 136, 111253. https://pubmed.ncbi.nlm.nih.gov/39828080/
Sinha, S. S. (2016). Trauma-induced insomnia: A novel model for trauma and sleep research. Sleep Medicine Reviews, 25, 74-83. https://pubmed.ncbi.nlm.nih.gov/26140870/
van Liempt, S., Arends, J., Cluitmans, P. J. M., Westenberg, H. G. M., Kahn, R. S., & Vermetten, E. (2013). Sympathetic activity and hypothalamo-pituitary-adrenal axis activity during sleep in post-traumatic stress disorder: A study assessing polysomnography with simultaneous blood sampling. Psychoneuroendocrinology, 38(1), 155-165. https://pubmed.ncbi.nlm.nih.gov/22776420/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 10 references cited
