The experience of a brain that will not turn off at bedtime is one of the more common reasons people search for sleep help. The reason it happens is physiological — the autonomic nervous system fails to transition from sympathetic dominance (elevated heart rate, heightened alertness, increased cortisol output) to parasympathetic dominance (lower heart rate, reduced arousal, conditions that allow sleep onset). GABA — gamma-aminobutyric acid, the brain’s primary inhibitory neurotransmitter — is central to that transition. When GABAergic inhibition is insufficient, sympathetic activation persists into the hours when the body needs to be transitioning toward sleep.
This article covers how GABA receptor impairment drives autonomic overactivation, the difference between GABA levels and receptor function, region-specific GABA roles in sleep stages, and evidence for restoring GABAergic tone. It does not cover hormonal GABA interactions or full autonomic sleep theory. For the broader picture of how autonomic regulation affects sleep, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It.
Autonomic dysregulation is one of several causes of sleep disruption. The autonomic pillar covers the broader framework, including heart rate variability, hyperarousal, and gut-vagus pathways.
How Does an Inhibitory Receptor Deficit Cause Autonomic Overactivation?
The hypothalamic paraventricular nucleus (PVN) is a region of the brain that regulates sympathetic nervous system output. Under normal conditions, GABAergic neurons provide tonic inhibition to the PVN — a continuous low-level suppression that keeps sympathetic activity in check. When this inhibition is intact, the PVN constrains heart rate, blood pressure, and sympathetic nerve firing within ranges compatible with sleep onset.
Perry et al. (2014) tested what happens when that inhibition is removed. In a rodent model, 14 days of sleep restriction (repeated cycles of 20 hours of sleep deprivation followed by 4-hour recovery) produced measurable downregulation of GABA-A receptors in the PVN. The researchers then confirmed the causal direction: microinjection of bicuculline — a selective GABA-A receptor antagonist — into the PVN of control animals elevated renal sympathetic nerve activity, mean arterial pressure, and heart rate. In sleep-restricted animals, the same challenge produced even greater sympathetic responses, both in amplitude and frequency.
This establishes a mechanistic pathway: sleep disruption reduces GABAergic inhibition in the PVN, and reduced inhibition in the PVN elevates sympathetic tone. The cardiovascular consequences — higher resting heart rate, elevated blood pressure — are measurable and sustained.
A 2024 randomized controlled trial adds a human dimension. Guimaraes et al. (2024) found that 200 mg of oral GABA daily for 90 days increased parasympathetic heart rate variability and improved sleep efficiency. The autonomic change was progressive over weeks, not an acute sedative effect — consistent with a gradual restoration of inhibitory tone rather than a one-time suppression of arousal.
The PVN is a regulatory hub where GABA receptor function directly determines whether the autonomic nervous system can transition from sympathetic dominance to the parasympathetic state required for sleep.
Is the Problem Low Inhibitory Neurotransmitter Levels or Impaired Receptors?
The assumption that “low GABA” explains the inability to sleep is incomplete. Xiang et al. (2023) compared peripheral blood samples from 30 people meeting DSM-5 criteria for insomnia disorder against 30 age-matched healthy controls. Serum GABA concentrations showed no difference between groups. The GABA was there. What differed was the receptor response.
GABA-A receptor alpha-1 subunit mRNA expression was lower in the insomnia group compared to controls (p < 0.001). Alpha-2 subunit mRNA was also reduced (p < 0.01). Sleep quality scores correlated negatively with alpha-1 subunit expression — the more downregulated the receptor, the worse the sleep. Daytime functioning scores showed an inverse relationship with alpha-2 expression, extending the association beyond nighttime to the functional impairment that characterizes insomnia.
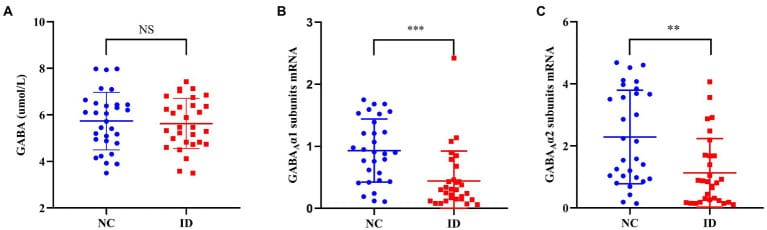
This matters because the receptor subtype determines which aspect of sleep is affected. Winsky-Sommerer (2009) established that GABA-A receptors containing different alpha subunits serve different functions: alpha-1-containing receptors mediate the sedative component of sleep onset, while alpha-2 and alpha-3 subunits contribute to anxiolytic and muscle-relaxant effects. Downregulation of alpha-1 and alpha-2 subunits in insomnia is consistent with impairment at both the sleep-onset and the anxiety-related dimensions of the disorder.
The distinction between GABA availability and receptor responsiveness has practical implications. If the problem were a production deficit — not enough GABA — then increasing GABA through supplementation or precursor support would be the primary approach. But if the receptors are downregulated, restoring receptor sensitivity becomes equally important. These are not mutually exclusive. Some people may have both reduced GABA production and impaired receptor expression, and the combination compounds the inhibitory deficit.
Why Does Sleep Require Different Types of Inhibition at Different Stages?
GABA acts through at least two modes of receptor-mediated inhibition, each with a different role in sleep architecture.
Phasic inhibition occurs at synapses — the junctions between neurons. When GABA is released into the synapse, it binds to GABA-A receptors containing alpha-1 subunits, producing a brief, strong suppression of neural firing. This is the mechanism that initiates sleep onset: fast, transient pulses of inhibition that reduce cortical arousal enough for the transition from wakefulness to NREM sleep (Winsky-Sommerer, 2009).
Tonic inhibition works outside the synapse. Extrasynaptic GABA-A receptors — particularly those containing alpha-4 and delta subunits — respond to low ambient GABA concentrations in the extracellular space. Instead of brief pulses, tonic inhibition produces a continuous, low-level suppression that maintains sleep depth during slow-wave sleep. This sustained inhibitory tone is what keeps neural activity low enough to preserve deep sleep once it has been initiated.
This subunit-level architecture explains a well-documented pharmacological observation. Benzodiazepines enhance GABA-A receptor activity broadly, across multiple subunit types. They increase total sleep time. But they also suppress slow-wave sleep and alter sleep architecture — because broad enhancement does not replicate the specific coordination of phasic and tonic inhibition that natural sleep requires (Winsky-Sommerer, 2009).
Oishi et al. (2023) introduced a further distinction. GABAergic neurons in different brain regions serve opposing functions. In the preoptic area (POA), GABAergic neurons suppress monoaminergic and cholinergic arousal circuits during sleep onset — this is the classical sleep-promoting role. But in the ventral tegmental area (VTA), GABAergic neurons sustain wakefulness by disinhibiting arousal pathways. The same neurotransmitter, through different circuits, promotes both sleep and waking.
This region-specificity is why broad GABA enhancement produces sedation without restoring the architecture of natural sleep. Sedation suppresses cortical activity globally. Natural sleep requires coordinated inhibition in some regions and disinhibition in others, through different receptor subtypes, at different times across the night.
Can Restoring Inhibitory Tone Reverse the Autonomic Imbalance That Prevents Sleep?
The previous three sections describe the problem: GABA receptor downregulation elevates sympathetic tone, receptor impairment can exist independently of GABA levels, and different receptor subtypes govern different sleep stages. The Guimaraes et al. (2024) trial tested whether restoring GABAergic tone could reverse the autonomic imbalance.
In this double-blind, placebo-controlled randomized controlled trial, 30 sedentary overweight women received either 200 mg of oral GABA daily combined with physical exercise, or placebo combined with physical exercise, over 90 days. Outcomes were assessed at baseline, 45 days, and 90 days. The GABA group showed increased heart rate variability attributed to heightened parasympathetic predominance. Sleep efficiency improved as measured by Pittsburgh Sleep Quality Index scores. Depression scores on the DASS-21 scale also decreased in the GABA group compared to placebo.
The progressive nature of the change — measured at three timepoints over 90 days — is informative. An acute sedative effect would produce rapid changes that plateau. The trajectory documented in this trial is consistent with a gradual restoration of autonomic balance, where parasympathetic tone builds incrementally rather than being forced by a one-time suppression of arousal.
Kong et al. (2023) provide context for why this autonomic restoration matters beyond sleep onset. In a study of 78 older adults combining fMRI with overnight polysomnography, higher parasympathetic HRV during slow-wave sleep correlated with stronger functional connectivity between the right anterior insula and posterior midcingulate cortex (F = 3.98, P = 0.022) — regions within the central autonomic network. Elevated parasympathetic HRV during slow-wave sleep also correlated with enhanced right amygdala-thalamic connectivity (F = 6.21, P = 0.005).
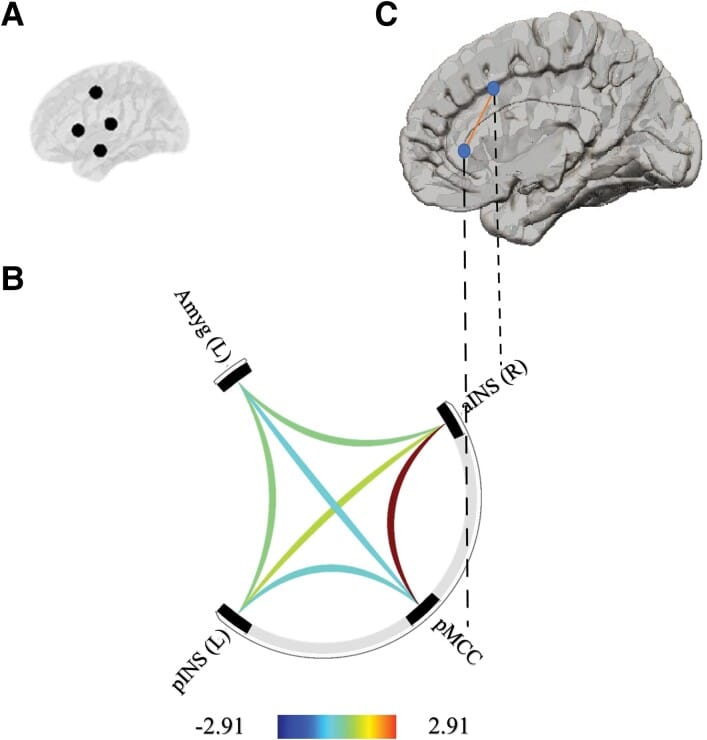
These associations were absent during REM sleep and wakefulness — slow-wave sleep is the window where parasympathetic regulation and central autonomic network integrity co-express. Disruption of slow-wave-specific parasympathetic function, as occurs in insomnia where slow-wave sleep is often fragmented, may impair both peripheral autonomic recovery and central brain network maintenance.
Two lines of evidence converge: a trial showing that restoring GABA tone changes autonomic balance, and a neuroimaging study showing that parasympathetic function during deep sleep correlates with brain network connectivity. Restoring GABAergic inhibition affects both the ability to fall asleep and the quality of the autonomic state during deep sleep — including the brain network maintenance that depends on parasympathetic dominance during slow-wave sleep.
A brain that stays in a state of arousal at night might involve more than one mechanism. GABA receptor impairment can overlap with hyperarousal from chronic stress, HRV dysregulation, or gut-vagus pathway disruption — and these causes might compound each other. When multiple autonomic pathways are involved, addressing only one might not be enough.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Are Racing Thoughts at Night an Inhibitory Neurotransmitter Problem or a Psychological One?
The framing matters because it changes the approach. If racing thoughts are “just anxiety” or “just stress,” the response is often cognitive — journaling, breathing exercises, thought redirection. These can help with the experiential dimension. But when the underlying issue is that GABA-A receptors in the hypothalamic paraventricular nucleus are downregulated, sympathetic outflow stays elevated regardless of what the conscious mind is doing.
In a rodent model, Perry et al. (2014) demonstrated that reduced GABAergic inhibition in the PVN directly elevates sympathetic nerve activity, heart rate, and blood pressure. The physiological state that results — heightened arousal, faster heart rate, increased alertness — is the same state that the mind experiences as racing thoughts, difficulty disengaging from the day, or an inability to wind down. Addressing only the cognitive experience without addressing the autonomic imbalance may leave the physiological driver unresolved.
Does Attention Deficit Hyperactivity Disorder Affect the Ability to Switch Off at Night?
The overlap is in the neurotransmitter pathway. GABAergic neurons in the preoptic area suppress arousal circuits during sleep onset, and GABAergic neurons in other regions — including prefrontal circuits involved in attention regulation — modulate executive function during wakefulness (Oishi et al., 2023). ADHD involves altered function in both of these domains: difficulty regulating attention during the day and difficulty transitioning to sleep at night.
This does not mean that ADHD and insomnia share a single cause. ADHD-related sleep difficulty may involve dopaminergic, noradrenergic, and circadian factors in addition to GABAergic impairment. But the shared GABAergic component may explain why the subjective experience — a brain that will not disengage — feels similar across both conditions, even when the full underlying pathology differs.
Can Vagus Nerve Exercises Improve Parasympathetic Tone?
The vagus nerve is the primary conduit for parasympathetic communication between the brain and the body. Higher vagal tone is associated with greater parasympathetic heart rate variability and a more efficient transition from sympathetic to parasympathetic dominance at sleep onset.
Kong et al. (2023) found that parasympathetic HRV during slow-wave sleep was associated with stronger connectivity in the central autonomic network — the brain regions that regulate autonomic output. This suggests that interventions supporting vagal tone may influence the central circuits governing autonomic balance, not only peripheral heart rate patterns.
Is Autonomic Dysregulation the Same as Anxiety?
Ma et al. (2024) measured HRV at the sleep onset transition in 328 participants. People with insomnia showed higher heart rate (p = 0.004), lower overall HRV measured by SDNN (p = 0.003), and reduced parasympathetic markers — pNN20 was lower (p < 0.001) and pNN50 was lower (p = 0.010) — compared to normal sleepers. These deficits persisted after adjusting for age, sex, BMI, and sleep apnea severity, indicating that the autonomic impairment is specific to insomnia rather than a byproduct of other conditions.
This is a physiological measurement, not a psychological assessment. A person with these autonomic markers may or may not report anxiety. Similarly, a person reporting anxiety may or may not have measurable autonomic dysregulation. The two can co-occur, and often do — but they are separate phenomena that may require separate approaches.
Related Reading:
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of how autonomic regulation affects sleep architecture, HRV, and 3am wakeups
- Can Your Nervous System Get Stuck in Fight or Flight and Ruin Your Sleep? — chronic sympathetic activation and its effects on sleep onset and maintenance
- What Is Hyperarousal Insomnia? Why You’re Wired but Tired Every Night — the hyperarousal model of insomnia, cortisol patterns, and measurable markers
- Does Benadryl Destroy Your Sleep? How Anticholinergic Drugs Suppress REM Sleep — how anticholinergic medications suppress REM sleep through acetylcholine blockade
- What Your Overnight HRV Is Telling You About Your Sleep: The Vagal Tone Connection — using heart rate variability as a window into autonomic sleep quality
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — the gut-vagus-brain axis and its role in autonomic regulation during sleep
- Why Is Your REM Sleep Fragmented? The Brainstem Switch That Controls Dream Sleep — brainstem cholinergic-monoaminergic switching and REM fragmentation
- Your Brain Makes Its Own Sleep Drug — And It’s More Sophisticated Than Valium — GABA-A receptor modulation via neurosteroids
- Alcohol Sedates You, Then Wakes You at 3AM — GABA-A rebound excitability after alcohol metabolism
- Why Is Your Rapid Eye Movement Sleep Fragmented? The Brainstem Switch That Controls It — the brainstem circuit that controls dream sleep and which medications suppress it
- Does Benadryl Degrade Your Sleep? How Anticholinergic Drugs Suppress Rapid Eye Movement Sleep — how anticholinergic medications block the muscarinic receptors REM requires
- What Your Overnight Heart Rate Variability Is Telling You About Your Sleep: The Vagal Tone Connection — using HRV as a window into parasympathetic recovery during sleep
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut bacteria, GABA production, and the vagal pathway to sleep regulation
- Do Antidepressants Suppress Rapid Eye Movement Sleep? — SSRIs, SNRIs, and tricyclics affect sleep architecture
- Can Vagus Nerve Stimulation Devices Improve Insomnia? — clinical trial evidence for taVNS devices
- Does Your Sleep Tracker Accuracy Matter? — what Oura, WHOOP, and Apple Watch get right and wrong
- Is Your Insomnia a Nervous System Problem? — how to tell if autonomic dysregulation is driving sleep disruption
References
Guimaraes, A. P., et al. (2024). GABA supplementation, increased heart-rate variability, emotional response, sleep efficiency and reduced depression in sedentary overweight women undergoing physical exercise: Placebo-controlled, randomized clinical trial. Journal of Dietary Supplements, 21(4), 512-526. https://pubmed.ncbi.nlm.nih.gov/38321713/
Kong, S. D. X., et al. (2023). Heart rate variability during slow wave sleep is linked to functional connectivity in the central autonomic network. Brain Communications, 5(3), fcad129. https://pubmed.ncbi.nlm.nih.gov/37234683/
Ma, Y., Mullington, J. M., Wayne, P. M., & Yeh, G. Y. (2024). Heart rate variability during sleep onset in patients with insomnia with or without comorbid sleep apnea. Sleep Medicine, 122, 92-98. https://pubmed.ncbi.nlm.nih.gov/39137665/
Oishi, Y., Saito, Y. C., & Sakurai, T. (2023). GABAergic modulation of sleep-wake states. Pharmacology & Therapeutics, 249, 108505. https://pubmed.ncbi.nlm.nih.gov/37541595/
Perry, J. C., et al. (2014). Interconnectivity of sympathetic and sleep networks is mediated through reduction of gamma aminobutyric acidergic inhibition in the paraventricular nucleus. Journal of Sleep Research, 23(2), 168-175. https://pubmed.ncbi.nlm.nih.gov/24283672/
Winsky-Sommerer, R. (2009). Role of GABA-A receptors in the physiology and pharmacology of sleep. European Journal of Neuroscience, 29(9), 1779-1794. https://pubmed.ncbi.nlm.nih.gov/19473233/
Xiang, T., et al. (2023). Impairment of GABA inhibition in insomnia disorders: Evidence from the peripheral blood system. Frontiers in Psychiatry, 14, 1134434. https://pubmed.ncbi.nlm.nih.gov/36846238/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
