Gut-brain content typically goes in one direction: gut bacteria affect the brain and sleep. The reverse — that poor sleep damages the gut microbiome — is less discussed but equally supported. A randomized crossover study restricted healthy young men to 2 hours of sleep per night for 3 nights and measured a 21% reduction in bacterial richness (Karl et al., 2023). Chronic insomnia sustains that reduction.
This article covers the evidence that sleep disruption damages the gut microbiome, the bacterial changes documented in human and rodent studies, the bidirectional causal evidence from Mendelian randomization, and how to break the reinforcing cycle. For the full autonomic framework, see the autonomic sleep disruption pillar. The gut-brain connection is one of several autonomic pathways affecting sleep — this article covers the sleep-to-gut direction of that pathway.
What Happens to Your Gut Microbiome When You Do Not Sleep?
Karl et al. (2023) enrolled 19 healthy young men (mean age 20, mean BMI 24.4) in a randomized crossover trial comparing three nights of adequate sleep (7-9 hours) against three nights of severe sleep restriction (2 hours per night). Using 16S rRNA gene sequencing, the researchers found that amplicon sequencing variant (ASV) richness — a measure of how many distinct bacterial taxa are present — was 21% lower during sleep restriction compared to adequate sleep (P = 0.03).
The reduction was in richness, not evenness. Shannon and Simpson diversity indices did not differ between conditions, meaning the number of distinct taxa decreased but the relative proportions of remaining species stayed stable. The first thing sleep deprivation does to the gut microbiome is remove taxa. The composition of what remains changes later.
One ASV within the Ruminococcaceae family showed differential abundance between conditions. Intestinal permeability — measured to test whether sleep restriction promotes gut barrier breakdown — did not change, dissociating acute richness loss from barrier impairment in healthy young men on a short timeline.
Supasitdikul et al. (2026) synthesized 20 studies — 4 human, 5 rat, 8 mouse, and 3 combined — to quantify the effect of sleep deprivation on gut microbiome composition across species. In rodent studies with larger sample sizes, Shannon diversity decreased with a standardized mean difference of -1.27 (95% CI: -2.20 to -0.34), and the Firmicutes-to-Bacteroidetes ratio increased with an SMD of +2.60 (95% CI: 1.61 to 3.59).
What does the Firmicutes-to-Bacteroidetes ratio change mean? An elevated ratio is associated with inflammatory conditions, metabolic disruption, and reduced production of short-chain fatty acids (SCFAs) — the microbial metabolites that feed colonocytes, maintain the gut barrier, and support vagal signaling to the brain. At the taxa level, sleep deprivation reduced Lactobacillaceae and Erysipelotrichaceae while increasing Ruminococcaceae and Lachnospiraceae. Lactobacillus abundance declined — a genus with documented roles in GABA production and vagal nerve communication.
Human studies showed nonsignificant trends in the same direction but were limited by small sample sizes and heterogeneous study designs. The rodent evidence is robust. The human evidence is consistent in direction but not yet sufficient for firm meta-analytic conclusions on its own.
What the Karl study adds is speed. Measurable richness loss occurred after 3 nights. This is not a consequence of months of chronic insomnia. It is detectable within 72 hours of sleep restriction in otherwise healthy people.
Is the Relationship Between Sleep and Gut Bacteria Bidirectional?
Shi et al. (2025) used genome-wide association study data to test whether the sleep-gut relationship is causal in both directions. Insomnia data came from 386,533 individuals. Microbiota data came from two independent cohorts: the MiBioGen alliance (18,340 individuals) and the Dutch Microbiome Project (8,208 individuals).
Mendelian randomization uses genetic variants as instruments. Because genetic variants are assigned at conception, they are not confounded by lifestyle, diet, or medication — making them stronger instruments for testing causation than observational studies can provide. If a genetic variant that predisposes to insomnia also predicts changes in gut bacteria, and this association survives tests for pleiotropy (the variant affecting the gut through some other pathway), the causal direction is supported.
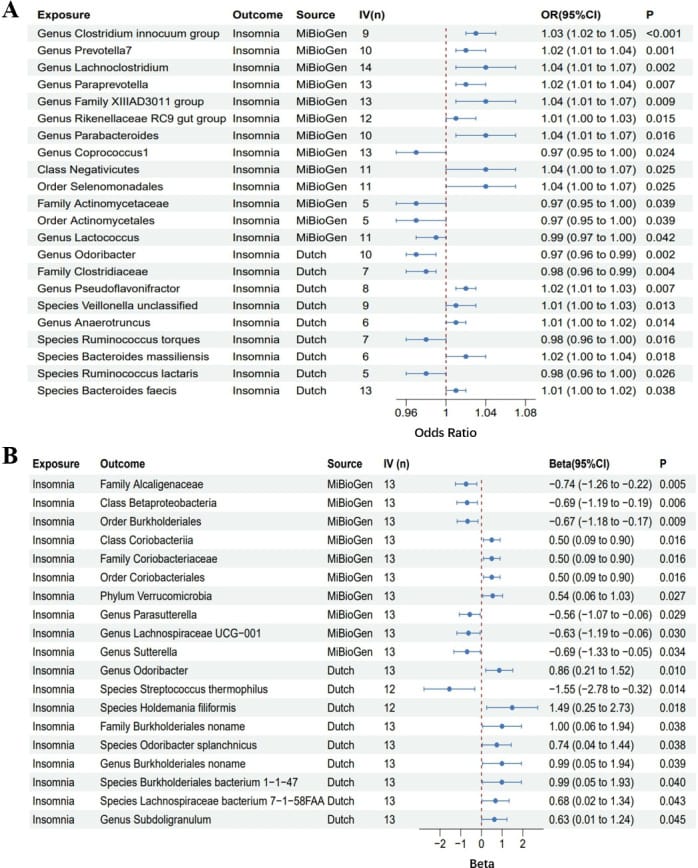
The reverse analysis — the direction this article focuses on — demonstrated that insomnia itself may causally reduce 7 taxa (OR: 0.21-0.57) and increase 12 taxa (OR: 1.65-4.43). This provides the strongest genetic evidence to date that poor sleep reshapes gut microbiome composition.
The forward analysis (gut affecting sleep) identified 14 gut bacterial taxa as potential causal contributors to insomnia risk, with odds ratios ranging from 1.01 to 1.04 per standard deviation increase in the taxon. Eight taxa showed protective causal effects with odds ratios of 0.97 to 0.99. The genus Odoribacter demonstrated a positive causal relationship with insomnia after Steiger filtering — a test that verifies the causal direction is correctly specified.
Cochran’s Q testing revealed no heterogeneity among the genetic instruments for the majority of taxa tested. MR-Egger and MR-PRESSO analyses found no horizontal pleiotropy, strengthening the causal inference in both directions.
Observational studies support this bidirectional architecture. Smith et al. (2019) measured gut microbiome composition alongside actigraphy-recorded sleep in 26 healthy young men and found that total microbiome diversity was positively correlated with sleep efficiency and inversely correlated with wake-after-sleep-onset. At the phylum level, both Bacteroidetes and Firmicutes richness showed positive associations with sleep efficiency and with interleukin-6 levels, suggesting the microbiome-sleep link may be mediated through immune pathways.
Holzhausen et al. (2024) extended this to a population-based sample of 720 people. Greater night-to-night sleep duration variability, higher wake-after-sleep-onset, lower sleep efficiency, and worse self-reported sleep quality were all independently associated with lower microbiome richness (Chao1 estimator) and diversity (Shannon and Inverse Simpson indices). Sleep consistency — low night-to-night variability — emerged as an underappreciated dimension of sleep health for microbiome maintenance.
Poor sleep reduces microbial diversity. Reduced diversity impairs SCFA production. Reduced SCFA production compromises vagal signaling. Compromised vagal signaling worsens sleep. Each night of disrupted sleep depletes the microbial populations that support the neural pathways involved in sleep — making the next night harder.
Does Circadian Disruption Affect the Gut Microbiome?
The evidence above comes from acute sleep deprivation — shortened sleep duration with maintained timing. Circadian disruption is a separate variable: sleep that occurs at the wrong time, regardless of duration. Shift workers and people with irregular sleep schedules experience both.
Grasa-Ciria et al. (2025) conducted a systematic review of gut microbiota alterations in adult shift workers, including 4 observational studies and 1 Mendelian randomization study. Observational studies reported reduced alpha-diversity in night shift workers compared to day workers. Pro-inflammatory genera were elevated in the shift-work group, including Escherichia/Shigella, Blautia, and Dialister — taxa previously linked to intestinal inflammation. These microbiota alterations correlated with gastrointestinal complaints and indicators of cardiometabolic disruption.
The Mendelian randomization component provided preliminary evidence supporting a causal relationship between circadian misalignment, gut dysbiosis, and increased cardiovascular risk — moving beyond association.
Why does changing sleep timing matter for gut bacteria? Because gut bacteria have circadian rhythms of their own. Mashaqi and Gozal (2020) reviewed the bidirectional relationship between gut dysbiosis and circadian rhythm misalignment. Both the central circadian clock (suprachiasmatic nucleus) and peripheral clocks — including those in the gut epithelium — can be affected by and can affect gut microbiota composition and metabolite production. Gut microbiota and their metabolites follow circadian patterns that can be disrupted when host sleep timing shifts.
The metabolic consequences are direct: nearly all metabolic pathways involved in carbohydrate, protein, and lipid processing are under circadian regulation. When the host clock and the microbial clock decouple, those metabolic pathways lose temporal coordination. The review also noted that intermittent hypoxia associated with obstructive sleep apnea alters the gut microbiome — demonstrating that sleep disorders can drive dysbiosis through multiple mechanisms, not circadian timing alone.
Morwani-Mangnani et al. (2022) added an age dimension to this picture. Their review of 11 human studies found that the effect of sleep disruption on the gut microbiome varies across the lifespan. In older adults, shorter sleep duration was associated with an increase in pro-inflammatory bacteria — consistent with the hypothesis that sleep loss accelerates inflammatory processes in aging populations. In younger adults, the effect on the Firmicutes-to-Bacteroidetes ratio was inconsistent across studies. Improvements in sleep quality in older adults were associated with increases in beneficial Verrucomicrobia and Lentisphaerae phyla, suggesting a restorative microbial response to better sleep.
The implication: the gut microbiome damage from sleep disruption may accumulate more consequentially with age. The same sleep disruption that produces a recoverable richness drop in a 20-year-old may produce a more persistent inflammatory composition change in a 60-year-old.
How Do You Break the Sleep-Gut Reinforcing Cycle?
The sleep-gut reinforcing cycle has two entry points: sleep and the microbiome. Addressing one side while the other remains disrupted is less effective because the intact half of the cycle continues driving degradation.
The sleep side. Addressing the autonomic drivers of poor sleep removes the upstream cause of microbiome damage. If poor sleep is driven by autonomic hyperarousal — elevated sympathetic activity at sleep onset — the approach addresses that mechanism. If it is driven by circadian misalignment, the approach addresses timing. The autonomic sleep disruption pillar covers the full framework. Consistency matters: Holzhausen et al. (2024) found that night-to-night sleep variability was independently associated with reduced microbiome diversity. Regular sleep timing supports microbial rhythms.
The microbiome side. Probiotic strains with sleep evidence provide research-guided microbial support. The genera identified in the Mendelian randomization study — including those with causal protective effects against insomnia — offer a starting point for strain selection. Dietary diversity and prebiotic fiber support the endogenous production of SCFAs, which maintain the gut barrier and serve as the primary fuel for colonocytes.
Li et al. (2018) reviewed metabolic, immune, and neural pathways connecting gut colonization to sleep regulation. The review identified three routes through which gut bacteria influence sleep: metabolic (SCFA production and tryptophan metabolism), immune (cytokine regulation and intestinal barrier integrity), and neural (vagal afferent signaling from gut to brainstem sleep centers). When the sleep-to-gut damage described in the sections above reduces the microbial populations that support these pathways, the gut-to-sleep return signaling degrades — which is why both sides of the cycle need to be addressed.
Recovery timeline. No study has directly measured microbiome recovery after insomnia resolution. The indirect evidence suggests recovery can be rapid. Karl et al. (2023) demonstrated that richness loss occurs within 3 nights of sleep restriction in healthy young men, and the population-based data from Holzhausen et al. (2024) show that people with consistent sleep patterns have higher diversity than those with irregular patterns — suggesting the microbiome responds to the current sleep pattern, not just the historical one. For the evidence-based timeline of probiotic effects on sleep, that article covers the expected recovery window.
The vagotomy evidence establishes that probiotics reach the brain through the vagus nerve — and that this pathway is required for their effects on mood and behavior. The same pathway supports the sleep-relevant signaling that is compromised when the microbiome is depleted by poor sleep. Restoring the microbial side of the pathway while improving sleep addresses both halves of the reinforcing cycle.
Gut microbiome disruption may not be the only factor affecting your sleep. Autonomic hyperarousal, metabolic disruptions, inflammatory processes, or hormonal changes may also be contributing. When multiple causes overlap, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
How Quickly Does Sleep Deprivation Affect the Gut Microbiome?
The Karl et al. (2023) finding is notable because the participants were healthy, young, and had no pre-existing gut conditions. The 21% richness reduction occurred in 3 nights, not weeks or months. This speed suggests the microbiome is responding to acute physiological changes associated with sleep loss — likely cortisol elevation, altered intestinal motility, and changes in mucosal immune function — rather than to cumulative dietary or behavioral changes.
The meta-analytic evidence from Supasitdikul et al. (2026) confirms that the direction of effect is consistent across both short-term and longer-term sleep deprivation studies: diversity decreases and the Firmicutes-to-Bacteroidetes ratio increases. The speed of onset in the Karl study suggests that even a few nights of poor sleep during an illness, a stressful period, or travel may produce measurable microbiome changes.
Can Improving Sleep Restore Gut Microbiome Diversity?
The gap in the evidence is direct: no study has taken people with insomnia, resolved their insomnia, and measured what happened to their microbiome. The indirect evidence from multiple directions supports the recovery hypothesis.
Holzhausen et al. (2024) found that people with consistent sleep patterns — low night-to-night variability in duration, high sleep efficiency, low wake-after-sleep-onset — had higher microbiome richness and diversity. This cross-sectional association cannot prove that improving sleep restores diversity, but it is consistent with the prediction.
The Mendelian randomization evidence from Shi et al. (2025) is stronger: if insomnia causally reduces the abundance of 7 taxa (OR: 0.21-0.57), resolving insomnia removes the causal driver of that reduction. The genetic evidence supports the direction of recovery even without a direct experimental trial.
Morwani-Mangnani et al. (2022) found that improvements in sleep quality in older adults were associated with increases in beneficial Verrucomicrobia and Lentisphaerae phyla — suggesting a restorative microbial response to better sleep even in aging populations where recovery might be expected to be slower.
Does the Sleep-Gut Connection Explain Why Insomnia Gets Worse Over Time?
The meta-analytic evidence from Supasitdikul et al. (2026) shows the sleep-to-gut direction: sleep deprivation reduces diversity and increases the Firmicutes-to-Bacteroidetes ratio. Li et al. (2018) mapped the gut-to-sleep pathways: metabolic (reduced SCFA production), immune (altered cytokine profiles and barrier integrity), and neural (compromised vagal signaling).
When both directions are active simultaneously, the sleep-gut relationship becomes self-sustaining. Night one of poor sleep reduces bacterial richness. Reduced richness impairs SCFA production. Reduced SCFAs compromise vagal signaling and increase intestinal permeability to inflammatory metabolites. Increased inflammation and reduced vagal tone make night two harder. Night two further reduces richness.
This may explain a pattern that many people with chronic insomnia recognize: insomnia that begins with an identifiable trigger (stress, illness, travel) persists after the trigger resolves. If the trigger initiated microbiome changes that now sustain the sleep disruption independently, removing the original trigger is insufficient. The microbiome-mediated feedback loop has become its own sustaining factor — which is why addressing both the sleep side and the gut side can be more effective than addressing either alone.
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Does Lion’s Mane Affect Your Sleep? Why It Helps Some People and Keeps Others Awake — lion’s mane’s dual mechanism: neurotrophic activity, cholinergic activity, and sleep architecture
- Why Do Cholinergic Supplements Give You Vivid Dreams? — the acetylcholine-REM pathway behind supplement-related vivid dreams
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — how post-viral autonomic disruption can affect sleep continuity
- Why Does Your Heart Race at Night After COVID? What the Vagus Nerve Has to Do With It — why post-COVID autonomic instability can show up as nighttime tachycardia
- Can POTS and Dysautonomia Cause Insomnia? Why Your Autonomic Nervous System Will Not Let You Rest — how orthostatic intolerance and autonomic instability can fragment sleep
- Can a Viral Infection Cause Chronic Insomnia? — how viral infections can disrupt autonomic regulation and sleep
- Why Will Your Nervous System Not Let You Sleep? Polyvagal Theory and Insomnia — how polyvagal states map to insomnia, hyperarousal, and unrefreshing sleep
- Why Does Trauma Cause Insomnia? How Your Nervous System Stays on Guard at Night — how trauma can keep sleep shaped by hyperarousal and vigilance
- Why Are You Sleeping 12 Hours and Still Exhausted? The Dorsal Vagal Shutdown Explained — the dorsal vagal pattern behind long sleep that still feels unrefreshing
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- How Do Probiotics Reach Your Brain? The Vagotomy Evidence — what vagotomy studies show about gut-brain communication
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
- Grasa-Ciria, D., Couto, S., Samatán, E., Martínez-Jarreta, B., Cenit, M. D. C., & Iguacel, I. (2025). Disrupted rhythms, disrupted microbes: A systematic review of shift work and gut microbiota alterations. Nutrients, 17(17), 2894. https://pubmed.ncbi.nlm.nih.gov/40944282/
- Holzhausen, E. A., Peppard, P. E., Sethi, A. K., Safdar, N., Malecki, K. C., Schultz, A. A., Deblois, C. L., & Hagen, E. W. (2024). Associations of gut microbiome richness and diversity with objective and subjective sleep measures in a population sample. Sleep, 47(3), zsad300. https://pubmed.ncbi.nlm.nih.gov/37988614/
- Karl, J. P., Whitney, C. C., Wilson, M. A., Fagnant, H. S., Radcliffe, P. N., Chakraborty, N., Campbell, R., Hoke, A., Gautam, A., Hammamieh, R., & Smith, T. J. (2023). Severe, short-term sleep restriction reduces gut microbiota community richness but does not alter intestinal permeability in healthy young men. Scientific Reports, 13(1), 213. https://pubmed.ncbi.nlm.nih.gov/36604516/
- Li, Y., Hao, Y., Fan, F., & Zhang, B. (2018). The role of microbiome in insomnia, circadian disturbance and depression. Frontiers in Psychiatry, 9, 669. https://pubmed.ncbi.nlm.nih.gov/30568608/
- Mashaqi, S., & Gozal, D. (2020). Circadian misalignment and the gut microbiome. A bidirectional relationship triggering inflammation and metabolic disorders — a literature review. Sleep Medicine, 72, 93-108. https://pubmed.ncbi.nlm.nih.gov/32559717/
- Morwani-Mangnani, J., Giannos, P., Belzer, C., Beekman, M., Eline Slagboom, P., & Prokopidis, K. (2022). Gut microbiome changes due to sleep disruption in older and younger individuals: A case for sarcopenia? Sleep, 45(12), zsac239. https://pubmed.ncbi.nlm.nih.gov/36183306/
- Shi, S., Liu, D., Baranova, A., Cao, H., & Zhang, F. (2025). Investigating bidirectional causal relationships between gut microbiota and insomnia. General Psychiatry, 38(4), e101855. https://pubmed.ncbi.nlm.nih.gov/40822499/
- Smith, R. P., Easson, C., Lyle, S. M., Kapoor, R., Donnelly, C. P., Davidson, E. J., Parikh, E., Lopez, J. V., & Tartar, J. L. (2019). Gut microbiome diversity is associated with sleep physiology in humans. PLoS One, 14(10), e0222394. https://pubmed.ncbi.nlm.nih.gov/31589627/
- Supasitdikul, T., Mazariegos, J. R. R., Nhat, N. N., Tung, Y.-T., Yang, D.-F., Lee, L.-J., Gunawan, S. P., & Chen, Y.-C. (2026). Sleep deprivation alters gut microbiome diversity and taxonomy: A systematic review and meta-analysis of human and rodent studies. Journal of Sleep Research, 35(2), e70125. https://pubmed.ncbi.nlm.nih.gov/40562421/
Written by Kat Fu, M.S., M.S. — Last reviewed: May 2026 — 9 references cited
