Men considering or already using testosterone replacement therapy often ask whether it will help or hurt their sleep. The answer depends on the underlying sleep condition — and the evidence points in different directions depending on the situation.
This article covers the evidence on how testosterone replacement therapy affects sleep quality, the interaction between testosterone replacement therapy and obstructive sleep apnea, and the role of body weight in mediating both. For the broader landscape of how hormones disrupt sleep in men, see Hormonal Sleep Disruption in Men.
Testosterone replacement therapy is one approach that targets testosterone-related sleep disruption. But other contributors to fragmented sleep — cortisol dysregulation, GABA deficiency, inflammatory cytokines, and circadian disruption — can each independently fragment sleep and may persist regardless of testosterone status.
Does Testosterone Replacement Therapy Improve Sleep in Men with Low Testosterone?
The best evidence for testosterone replacement therapy improving sleep comes from a 2018 controlled study by Shigehara and colleagues. The researchers screened 100 hypogonadal men for sleep disturbance and, after excluding those with obstructive sleep apnea, enrolled 48 participants — 24 received testosterone replacement therapy and 24 served as untreated controls over 12 months (Shigehara et al., 2018).
At baseline, hypogonadal men with sleep disturbance had worse scores across multiple validated measures: the SF-36 health survey, the Aging Males’ Symptoms scale (AMS), the International Prostate Symptom Score (IPSS), and the Sexual Health Inventory for Men (SHIM).
After 12 months, the testosterone replacement therapy group showed improvements in sleep disturbance, erectile function, sexual desire, and several quality-of-life domains compared to controls. The control group showed no change in any of these parameters over the same period. The authors proposed sleep disturbance as an indicator of severe hypogonadism — meaning that unexplained sleep complaints in men may point toward testosterone deficiency as a contributing cause.
The qualification matters: these men were screened to exclude obstructive sleep apnea. The positive result applies to men whose sleep problems stem from testosterone deficiency itself — not from obstructed breathing.
Population-level data supports this connection. A large U.S. claims database analysis by Agrawal and colleagues found insomnia associated with testosterone deficiency at an odds ratio of 1.74 (Agrawal et al., 2024). This means men with insomnia were 74% more likely to also have testosterone deficiency compared to matched controls without insomnia.
Testosterone replacement therapy addresses sleep disruption caused by testosterone deficiency. It does not address sleep disruption caused by other mechanisms — cortisol dysregulation, GABA deficiency, inflammation, or circadian disruption each require different approaches.
Can Testosterone Replacement Therapy Make Sleep Apnea Worse?
The concern about testosterone replacement therapy and sleep apnea is widespread — but the evidence is more nuanced than a blanket caution.
Hoyos and colleagues conducted the only randomized, double-blind, placebo-controlled trial directly testing testosterone replacement therapy in men with severe obstructive sleep apnea (Hoyos et al., 2012). They enrolled 67 obese men with an apnea-hypopnea index of 30 or more events per hour — all placed on a hypocaloric diet and assigned to either intramuscular testosterone undecanoate (1000 mg) or placebo at weeks 0, 6, and 12.
At the 7-week assessment, testosterone worsened the oxygen desaturation index (ODI) by 10.3 events per hour compared to placebo (p=0.03) and increased the percentage of sleep time with oxygen saturation below 90% by 6.1% (p=0.01).
By week 18, neither the ODI nor nocturnal hypoxemia differed between groups (ODI p=0.36; hypoxemia p=0.23). The worsening was time-limited.
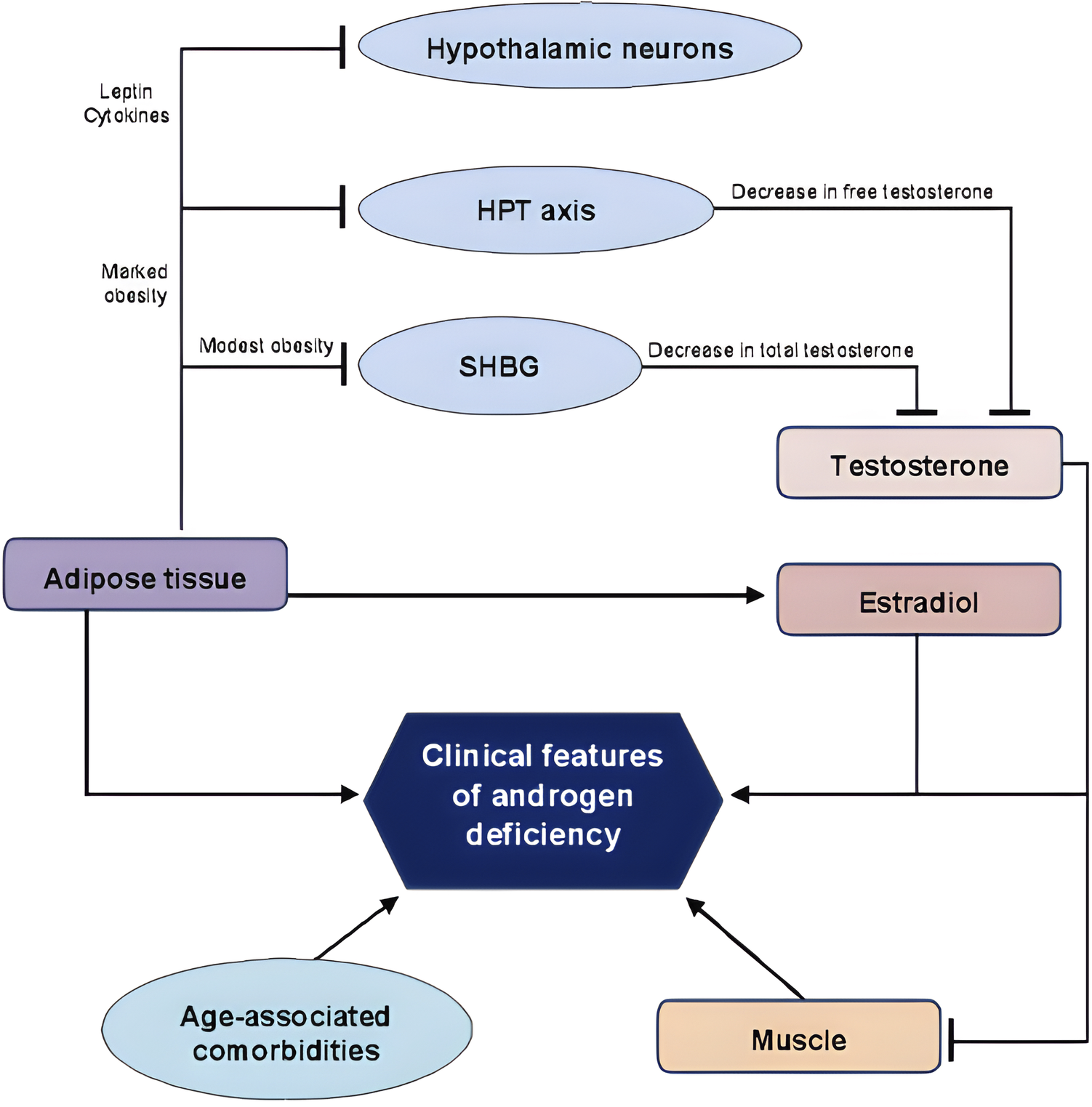
A 2023 review by Graziani and colleagues synthesized the likely mechanism: short-term testosterone replacement therapy may reduce contraction of upper airway dilator muscles and blunt the neural responses to hypoxia and hypercapnia — the reflexes that normally reopen the airway during apnea episodes (Graziani et al., 2023).
Over the longer term, testosterone replacement therapy can improve body composition by reducing adiposity and increasing respiratory muscle mass, which may indirectly benefit obstructive sleep apnea. This aligns with evidence that body weight is a primary driver of both testosterone decline and sleep-disordered breathing.
The first weeks after starting testosterone replacement therapy are the risk window, particularly in men with existing severe obstructive sleep apnea who are not using continuous positive airway pressure (CPAP). Monitoring during this period is recommended.
Does Sleep Apnea Lower Testosterone?
The relationship between sleep apnea and testosterone runs in both directions. Many men have both low testosterone and obstructive sleep apnea — and the question of which came first has a measurable answer.
Wang and colleagues conducted a meta-analysis of 24 studies including 1,389 men with obstructive sleep apnea and 845 male controls (Wang et al., 2023). The standardized mean difference in testosterone was -0.97 — a large effect size, meaning men with obstructive sleep apnea had testosterone levels nearly a full standard deviation lower than men without it. This effect was specific to men; no comparable difference was found in women.
The heterogeneity across studies was high (I-squared = 94.9%), with race, age, body mass index, and assay methods all contributing to variability between studies.
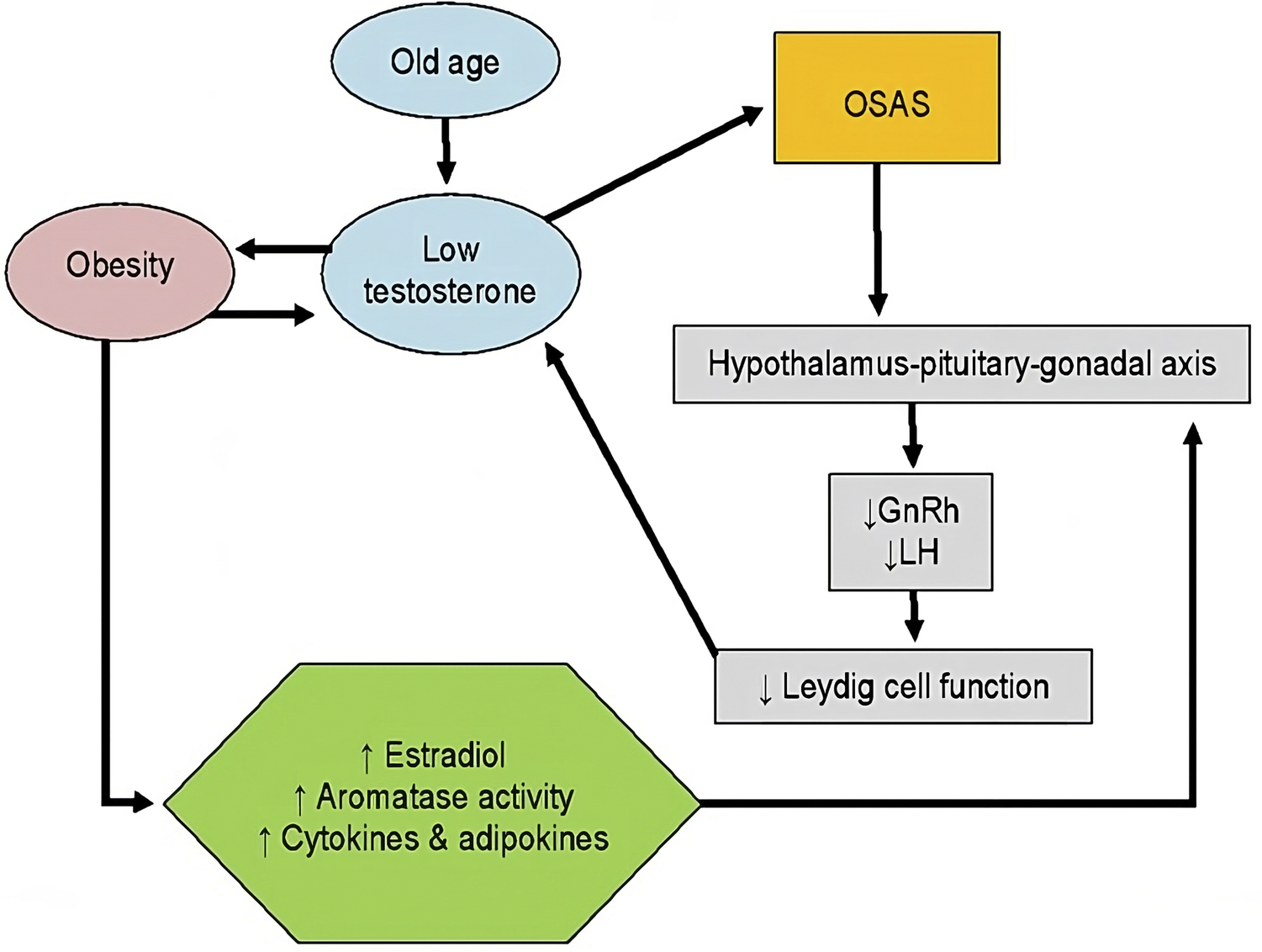
The mechanism runs through the hypothalamic-pituitary-gonadal (HPG) axis. Intermittent hypoxia — the repeated oxygen drops that occur during apnea episodes — suppresses gonadotropin-releasing hormone (GnRH) pulsatility. Less GnRH means less luteinizing hormone (LH), which means less Leydig cell stimulation in the testes, which means less testosterone production (Graziani et al., 2023). This is the same hypothalamic suppression pathway through which inflammatory cytokines reduce testosterone.
However, body weight complicates this picture. Barrett-Connor and colleagues studied 1,312 community-dwelling men aged 65 and older and found that lower testosterone was associated with lower sleep efficiency, more awakenings, less slow-wave sleep, and higher apnea-hypopnea indices (Barrett-Connor et al., 2008). But after adjusting for body mass index, these associations were attenuated or became non-significant. Adiposity — not testosterone itself — appeared to be the primary mediator of sleep-disordered breathing in these older men.
This has practical implications: losing weight may improve both testosterone levels and sleep apnea simultaneously because adiposity drives both. Testosterone replacement therapy addresses the testosterone side but may not address the underlying adiposity-obstructive sleep apnea connection. For more on how visceral fat suppresses testosterone and disrupts sleep, see Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep?.
Testosterone replacement therapy addresses one contributor to sleep disruption, but multiple causes often overlap. Cortisol dysregulation, GABA deficiency, inflammatory cytokines, and circadian disruption can each independently fragment sleep — and each may persist regardless of testosterone status. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
How Long Does It Take for Testosterone Replacement Therapy to Improve Sleep?
Two timelines from the research define what to expect. In the Shigehara study of hypogonadal men without obstructive sleep apnea, improvements in sleep disturbance, sexual function, and quality of life accumulated over 12 months of testosterone replacement therapy (Shigehara et al., 2018). These changes did not appear in the untreated control group, indicating that the improvements were attributable to testosterone replacement therapy rather than placebo effect or time.
In the Hoyos trial of men with severe obstructive sleep apnea, the trajectory was different: oxygen desaturation worsened at 7 weeks, then returned to baseline by 18 weeks (Hoyos et al., 2012). Sleep effects of testosterone replacement therapy develop over weeks to months, and the direction of change in the early weeks depends on whether obstructive sleep apnea is present.
Should Men with Sleep Apnea Avoid Testosterone Replacement Therapy?
The Graziani 2023 review recommends a personalized approach: polysomnographic evaluation before starting testosterone replacement therapy in high-risk men (obese men, those with metabolic conditions), and close monitoring in the first 7 weeks after initiation (Graziani et al., 2023). Men already using CPAP have ongoing airway support during the early adjustment period.
Long-term, testosterone replacement therapy may reduce adiposity and increase respiratory muscle mass — both of which can benefit obstructive sleep apnea indirectly. The evidence does not indicate that testosterone replacement therapy causes new-onset sleep apnea; the transient worsening in the Hoyos trial occurred in men who already had severe obstructive sleep apnea.
Can Improving Sleep Raise Testosterone Without Testosterone Replacement Therapy?
The nocturnal testosterone production cycle depends on sleep. Testosterone concentrations peak during sleep, and disrupted or shortened sleep reduces that peak.
Hernandez-Perez and colleagues analyzed NHANES data from 8,748 adults and found the sleep-testosterone relationship varies by age (Hernandez-Perez et al., 2024). In young men aged 20-40, sleeping six hours or fewer was associated with elevated testosterone (OR 3.62), possibly reflecting a compensatory hormonal response. In middle-aged men aged 41-64, extended sleep duration was associated with reduced testosterone (OR 2.03), indicating an age-dependent change in how sleep duration and testosterone relate.
Sleep improvement is a complementary approach — not a replacement for testosterone replacement therapy when testosterone deficiency is present, but a supporting step that may sustain testosterone production. For the full mechanism of how low testosterone directly fragments sleep architecture, see the dedicated article.
Are All Sleep Problems Linked to Low Testosterone?
The Agrawal 2024 claims database analysis quantified these graded associations across three types of sleep disorders in men aged 40-70 (Agrawal et al., 2024). Circadian rhythm disruption — where the internal clock is misaligned with the sleep-wake cycle — had the strongest link to testosterone deficiency. This is consistent with the known circadian patterning of testosterone production, which peaks during sleep and depends on intact circadian clock function.
Insomnia (OR 1.74) had a stronger association with testosterone deficiency than sleep apnea (OR 1.66), suggesting that sleep continuity and duration may be as hormonally consequential as the oxygen desaturation that characterizes apnea.
Testosterone deficiency is linked to multiple sleep disorder types, not only obstructive sleep apnea. Sleep disruption in men often involves multiple overlapping causes. For the broader framework, see Hormonal Sleep Disruption in Men.
Related Reading
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, growth hormone, and other hormones disrupt sleep in men
- Does Low Testosterone Cause Sleep Problems in Men? — How declining testosterone directly fragments sleep architecture in men
- Can a Cortisol Spike Wake You Up at 3am? — The cortisol-testosterone axis and nocturnal arousal patterns
- Does Growth Hormone Decline Affect Your Sleep After 40? — How age-related growth hormone loss disrupts slow-wave sleep
- Does Andropause Cause Insomnia? What Men Over 50 Need to Know — Compound hormonal aging and sleep disruption in men over 50
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep? — How visceral fat suppresses testosterone through aromatase and disrupts sleep
- Can Ultra-Processed Food Lower Testosterone and Disrupt Sleep? — How endocrine-disrupting chemicals in processed food suppress testosterone and degrade sleep
- Can Inflammation Suppress Testosterone and Disrupt Your Sleep? — How inflammatory cytokines suppress testosterone production at the brain and testicular level
- Can Low GABA Cause Waking Up at 3am? — How insufficient GABA maintains sleep through the night
- What Are the Signs of Low GABA at Night? — Recognizing GABA deficiency that disrupts sleep
- Do GABA Supplements Help You Stay Asleep Through the Night? — Evaluating GABA supplementation evidence for sleep maintenance
- Does GABA Affect Testosterone and Sleep in Men? — The GABA-testosterone connection in male sleep disruption
- How Do You Increase GABA Levels Naturally for Better Sleep? — Lifestyle and dietary approaches to support GABA function
References
Agrawal, P., Bourgeois, C., Engel, K., & Bhatt, N. (2024). Sleep disorders are associated with testosterone deficiency and erectile dysfunction — a U.S. claims database analysis. International Journal of Impotence Research, 36(2), 148-155. https://pubmed.ncbi.nlm.nih.gov/36473958/
Barrett-Connor, E., Dam, T.-T., Stone, K., Harrison, S. L., Redline, S., & Orwoll, E. (2008). The association of testosterone levels with overall sleep quality, sleep architecture, and sleep-disordered breathing. Journal of Clinical Endocrinology & Metabolism, 93(7), 2602-2609. https://pubmed.ncbi.nlm.nih.gov/18413429/
Graziani, A., Grande, G., & Ferlin, A. (2023). The complex relation between obstructive sleep apnoea syndrome, hypogonadism and testosterone replacement therapy. Frontiers in Reproductive Health, 5, 1219239. https://pubmed.ncbi.nlm.nih.gov/37881222/
Hernandez-Perez, J. G., Zamudio-Bernal, A. A., Gonzalez-Gallardo, S., Manjarrez-Gutierrez, G., & Lopez-Santiago, N. (2024). Association of sleep duration and quality with serum testosterone concentrations among men and women: NHANES 2011-2016. Andrology, 12(3), 587-597. https://pubmed.ncbi.nlm.nih.gov/37452666/
Hoyos, C. M., Killick, R., Yee, B. J., Marks, G. B., Grunstein, R. R., & Liu, P. Y. (2012). Effects of testosterone therapy on sleep and breathing in obese men with severe obstructive sleep apnoea: a randomized placebo-controlled trial. Clinical Endocrinology, 77(4), 599-607. https://pubmed.ncbi.nlm.nih.gov/22512435/
Shigehara, K., Konaka, H., Koh, E., Izumi, K., Kitagawa, Y., Mizokami, A., Keller, E. T., & Namiki, M. (2018). Sleep disturbance as a clinical sign for severe hypogonadism: efficacy of testosterone replacement therapy on sleep disturbance among hypogonadal men without obstructive sleep apnea. Aging Male, 21(2), 99-105. https://pubmed.ncbi.nlm.nih.gov/28920756/
Wang, H., Sun, Y., Wang, S., Qian, H., Pan, Y., Chen, G., & Zeng, Y. (2023). Obstructive sleep apnea and serum total testosterone: a system review and meta-analysis. Sleep & Breathing, 27(3), 789-797. https://pubmed.ncbi.nlm.nih.gov/35904664/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
