Over 40 million Americans take an antidepressant. Many report changes in dream intensity, sleep quality, or morning exhaustion that don’t match the expected benefit of the medication. REM sleep is when memory consolidation, emotional processing, and neural recovery are concentrated. When a medication reshapes that architecture, the downstream effects extend beyond subjective sleep quality to cognitive function and brain health over time.
This article covers how serotonergic antidepressants alter sleep architecture, which drug classes suppress REM the hardest and which preserve it, what REM rebound looks like on discontinuation, and what the research shows about long-term sleep quality on these medications. It does not cover the full autonomic cause overview — for that, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It. Medication-driven sleep disruption is one of several autonomic factors that reshape sleep; the pillar article covers the broader picture.
How Do Selective Serotonin Reuptake Inhibitors and Serotonin-Norepinephrine Reuptake Inhibitors Suppress Rapid Eye Movement Sleep?
The mechanism begins in the dorsal raphe nucleus, where serotonin-producing neurons are concentrated. During normal sleep, serotonin output decreases as the brain transitions toward REM — this decrease allows cholinergic neurons in the pedunculopontine tegmental nucleus (PPT) and laterodorsal tegmental nucleus (LDT) to activate and initiate REM episodes. SSRIs block serotonin reuptake transporters, increasing serotonin concentration in the synaptic cleft. That elevated serotonin inhibits the PPT/LDT cholinergic neurons that would otherwise generate REM. The result: delayed REM onset and shorter REM episodes throughout the night.
Wichniak et al. (2017) reviewed polysomnography data across multiple SSRI agents and found consistent REM suppression — reduced REM sleep percentage and prolonged REM latency — across the class. The magnitude varied by drug. Paroxetine and fluoxetine produced the strongest REM reduction. Sertraline and citalopram showed moderate effects. But the direction was the same across all SSRIs studied: less REM, later REM onset (Wichniak et al., 2017).
Wilson and Argyropoulos (2005) examined both acute and chronic effects. REM suppression appeared with the first dose and persisted with ongoing use. The degree of suppression did decrease over weeks — attributed to adaptive changes in serotonin autoreceptor sensitivity — but REM sleep percentage did not fully return to baseline during continued SSRI use (Wilson & Argyropoulos, 2005).
SNRIs add a second REM-suppressive pathway. Venlafaxine and duloxetine block both serotonin and norepinephrine reuptake. Norepinephrine, released from the locus coeruleus, also inhibits REM-generating circuits. Kluge et al. (2007) measured the effect of duloxetine in people with major depression using full-night polysomnography: REM sleep onset latency more than tripled (from 58.5 to 193.6 minutes), and total REM duration fell by approximately 46% (from 94.8 to 51.5 minutes). Duloxetine also increased stage 3 deep sleep from 21.0 to 37.4 minutes — a dissociation that distinguishes SNRIs from purely serotonergic agents (Kluge et al., 2007). The noradrenergic component may also increase sympathetic nervous system activity during sleep, contributing to more insomnia-type effects — difficulty falling asleep, nighttime awakenings — alongside REM suppression.
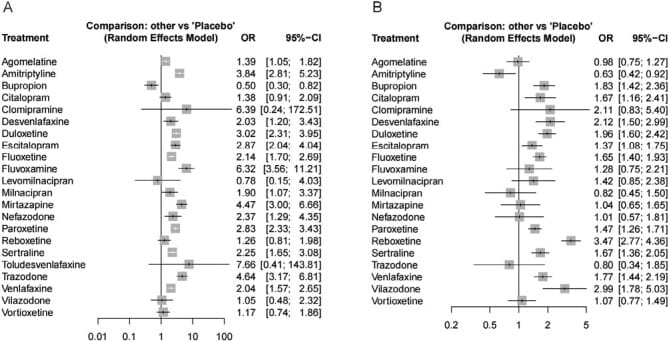
Which Antidepressants Are Worst for Sleep Architecture — and Which Preserve Rapid Eye Movement?
The Wichniak et al. (2017) review ranked antidepressants by magnitude of REM suppression across polysomnography studies. At the top: paroxetine, clomipramine, and fluoxetine — all producing marked reductions in REM sleep percentage and large increases in REM latency. In the moderate range: sertraline, citalopram, and venlafaxine. At the bottom — meaning the least REM disruption — bupropion and agomelatine, which showed minimal or no REM suppression.
Tricyclics suppress REM through a dual pathway that differs from SSRIs. In addition to serotonin reuptake inhibition, tricyclics block muscarinic acetylcholine receptors. Because acetylcholine is the primary neurotransmitter driving REM generation (via the PPT/LDT cholinergic neurons), this anticholinergic action adds a second REM-suppressive mechanism on top of the serotonergic one. Wilson and Argyropoulos (2005) noted that this dual mechanism explains why tricyclics like clomipramine can produce REM suppression as strong or stronger than SSRIs, despite generally lower serotonin reuptake selectivity. Tricyclics also affect sleep continuity differently — sedating tricyclics such as amitriptyline and doxepin tend to improve sleep onset and reduce nighttime awakenings through histamine H1 receptor blockade, even while suppressing REM (Wilson & Argyropoulos, 2005).
Mirtazapine occupies a separate category. It blocks serotonin 5-HT2 receptors and histamine H1 receptors, which increases deep sleep and may preserve or increase REM percentage. The histamine blockade produces sedation, which improves subjective sleep quality in many people. However, weight gain is a common side effect that limits its long-term use.
Bupropion works through dopamine and norepinephrine reuptake inhibition with no serotonergic action. Because the serotonergic pathway is the primary driver of REM suppression, bupropion largely spares REM architecture. Agomelatine acts as a melatonin receptor agonist and 5-HT2C antagonist — a pharmacological profile that supports both circadian rhythm alignment and REM preservation.
Zhou et al. (2023) conducted a network meta-analysis of 216 randomized controlled trials encompassing 64,696 participants and quantified insomnia and somnolence risk across 21 antidepressants. Among commonly prescribed SSRIs and SNRIs, sertraline, fluoxetine, escitalopram, and paroxetine all demonstrated insomnia odds ratios above placebo. Fluvoxamine carried the highest somnolence risk (OR 6.32). Sedating tricyclics such as amitriptyline showed protective effects against insomnia, reflecting their antihistaminergic properties — but this does not mean they preserve REM. An antidepressant can make you sleepier while still suppressing REM architecture underneath (Zhou et al., 2023).
The distinction matters: “best for sleep” and “best for the person” are not the same question. The choice involves depression efficacy, side-effect profile, individual pharmacogenomics, and the relative importance of sleep architecture versus sleep onset versus mood stability. REM suppression is one variable in that equation.
What Is Rapid Eye Movement Rebound After Stopping Antidepressants?
The neurobiology of REM rebound follows a predictable pattern. Chronic serotonergic suppression of REM-generating circuits leads to compensatory upregulation of cholinergic receptors in the PPT/LDT. When the suppressive serotonin input is removed — either by discontinuation or rapid dose reduction — those upregulated cholinergic neurons fire with less opposition than they had before the medication was started. The result is excessive REM: more REM episodes, longer REM duration, more vivid dream content.
Collins et al. (2024) provided direct neurochemical evidence for this rebound in a controlled study using mice given chronic paroxetine. Upon discontinuation, brain serotonin levels and serotonin metabolite (5-HIAA) concentrations in the hippocampus increased above control levels — the opposite of what occurs during ongoing use. Electrophysiological recordings confirmed that serotonin release, which had been suppressed during ongoing paroxetine exposure, rebounded above baseline levels after cessation. Dorsal raphe nucleus neurons showed increased activation in the early post-discontinuation window. Behaviorally, anxiety-like responses peaked at approximately day 2 post-cessation (Collins et al., 2024).
Sharp and Collins (2024) framed this in a broader review of SSRI discontinuation mechanisms: the rapid loss of inhibitory autoreceptor-mediated feedback that develops during sustained SSRI exposure allows serotonin neurons to enter a disinhibited, hyperactive state upon drug removal. This serotonergic overshoot is the neurochemical substrate for the increased REM pressure and dream intensity observed during antidepressant withdrawal (Sharp & Collins, 2024).
The timing of rebound correlates with the drug’s half-life. Wilson and Argyropoulos (2005) noted that paroxetine, with its short half-life, produces rapid-onset REM rebound within days. Fluoxetine, with a half-life extending beyond two weeks, produces a delayed and more gradual rebound. Feige et al. (2002) measured this directly in healthy volunteers: after three weeks of fluoxetine 40 mg/day, sleep continuity indices normalized within 2-4 days of discontinuation, but REM latency resolved slowly over the drug’s approximately 10-day elimination half-life, and REM fraction showed a rebound above pre-drug levels (Feige et al., 2002).
Antidepressants can also produce REM sleep without atonia (RSWA), a state where the normal muscle paralysis during REM is incomplete. This allows physical movement during dreams. When serotonergic antidepressants are discontinued, the combination of REM rebound and RSWA can produce the experience of vivid nightmares with physical acting-out that many people describe during withdrawal (Dauvilliers et al., 2018).
Gradual tapering is the standard approach to reduce rebound intensity. Slower dose reduction allows time for receptor re-equilibration — the cholinergic upregulation and serotonergic autoreceptor changes reverse more gradually, producing a smaller mismatch when the drug is fully discontinued.
Do Antidepressants Change Sleep Architecture Permanently?
The Wichniak et al. (2017) review examined longitudinal polysomnography data and found that REM percentage recovered after discontinuation in the studies that tracked recovery. The timeline varied — shorter for drugs with short half-lives, longer for drugs like fluoxetine — but the direction was consistently toward recovery rather than permanent change. However, recovery timeline data remains limited. Few studies follow people beyond the first several weeks after discontinuation (Wichniak et al., 2017).
Wilson and Argyropoulos (2005) documented persistent REM suppression during ongoing use — REM did not fully normalize even with months of continued use — but confirmed recovery trajectories after cessation. The partial tolerance that develops during use (less suppression at month 6 than at week 1) and the recovery after cessation suggest the brain retains the capacity to restore REM architecture (Wilson & Argyropoulos, 2005).
Feige et al. (2002) provided controlled data in healthy volunteers: after three weeks of fluoxetine, REM sleep fraction rebounded above baseline after discontinuation and then returned toward pre-drug levels as the drug cleared. The delta sleep ratio remained elevated for several days beyond drug clearance, but all measured sleep parameters trended toward baseline (Feige et al., 2002).
The masking problem complicates self-assessment. Depression itself disrupts sleep architecture — it increases REM density, shortens REM latency, and fragments sleep continuity. When an antidepressant suppresses REM and is then discontinued, the depression-related sleep disruption becomes visible again. A person may attribute their poor sleep to lasting drug effects when the underlying condition is the primary driver.
This matters for interpreting the experience of “my SSRI ruined my sleep permanently.” The explanation often involves three overlapping phenomena: REM rebound (short-term, weeks), return of depression-related sleep disruption (medium-term), and in some cases, autonomic dysregulation that predated the medication. Separating drug effect from underlying condition effect requires looking at the full timeline — what sleep was like before the antidepressant, during the medication, and at various points after discontinuation.
Antidepressant-related sleep disruption might not be the only factor affecting your sleep. Serotonergic REM suppression can compound with autonomic hyperarousal, GABA pathway impairment, circadian timing changes, or hormonal fluctuations. When multiple causes might be contributing, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can Selective Serotonin Reuptake Inhibitors Cause Insomnia?
SSRI-induced insomnia (difficulty falling asleep) is a distinct phenomenon from SSRI-induced REM suppression (altered sleep architecture once asleep). Both can occur simultaneously, but they arise from different mechanisms. The insomnia component is driven by serotonin’s activation of wake-promoting circuits; the REM suppression is driven by serotonin’s inhibition of cholinergic REM-generating circuits.
Evening dosing of activating SSRIs (fluoxetine, sertraline) tends to worsen insomnia; morning dosing is the standard adjustment. Wilson and Argyropoulos (2005) noted that activating SSRIs may initially increase sleep onset latency and arousals, while sedating agents can improve sleep continuity from the first dose — a distinction that influences timing and drug selection (Wilson & Argyropoulos, 2005). The Zhou et al. (2023) meta-analysis confirmed elevated insomnia odds ratios for sertraline, fluoxetine, escitalopram, and paroxetine compared to placebo across 64,696 participants (Zhou et al., 2023).
Why Do Some People Have Vivid Dreams on Antidepressants?
This within-night REM redistribution is distinct from the full REM rebound that occurs after discontinuation. During ongoing SSRI use, REM is suppressed early in the night when serotonergic tone is highest, but as serotonin levels fluctuate across sleep cycles, later REM episodes can emerge with greater intensity than they would without the medication. The compressed timing concentrates dream content into fewer, denser episodes.
Some people experience vivid dreams when starting an SSRI — before full serotonergic steady-state — and this often diminishes after 2-4 weeks as the brain adjusts. Wichniak et al. (2017) noted that decreased dream recall frequency during sustained SSRI use is a behavioral correlate of serotonergic REM suppression, suggesting that long-term users often report less dream recall, not more (Wichniak et al., 2017).
Why Do Selective Serotonin Reuptake Inhibitors Cause Rapid Eye Movement Sleep Without Muscle Paralysis?
During normal REM sleep, the sublaterodorsal nucleus triggers glycine and GABA release onto spinal motor neurons, producing the muscle paralysis (atonia) that prevents physical acting-out of dreams. Serotonin modulates this pathway — increased serotonergic tone in the ventral horn motor nuclei during REM can counteract the normal glycinergic and GABAergic inhibition, leaving motor neurons partially active.
Antidepressant-induced REM without atonia can mimic REM sleep behavior disorder (RBD), but the drug-induced form typically resolves after discontinuation. Biscarini et al. (2024) provided biomarker evidence supporting this distinction: only 30% of people with antidepressant-related RBD tested positive for dermal alpha-synuclein deposits, compared to 93% of those with RBD unrelated to antidepressant use (p = 0.00024) — suggesting that the antidepressant-related form is predominantly pharmacological rather than an early marker of neurodegenerative disease (Biscarini et al., 2024).
Do Serotonin-Norepinephrine Reuptake Inhibitors Affect Sleep Differently Than Selective Serotonin Reuptake Inhibitors?
The Wichniak et al. (2017) review compared sleep architecture profiles across SSRI and SNRI agents. Venlafaxine produced REM suppression comparable to mid-range SSRIs like sertraline, but with additional effects on sleep continuity that SSRIs did not produce — consistent with the noradrenergic component increasing arousal during sleep (Wichniak et al., 2017).
Kluge et al. (2007) demonstrated that duloxetine tripled REM latency and reduced REM duration by 46% while increasing stage 3 deep sleep — a dual effect distinguishing it from purely serotonergic agents where deep sleep changes are less pronounced (Kluge et al., 2007). The noradrenergic effects may make SNRIs more disruptive for people already in an autonomic hyperarousal state, where elevated sympathetic tone during sleep is already a problem. Adding norepinephrine reuptake inhibition on top of existing sympathetic overactivation can compound sleep onset difficulty and nighttime awakenings.
Related Reading:
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — how autonomic imbalance disrupts sleep architecture
- Can Your Nervous System Get Stuck in Fight or Flight and Ruin Your Sleep? — the self-reinforcing cortisol-sleep loop
- What Is Hyperarousal Insomnia? Why You’re Wired but Tired Every Night — autonomic mechanics behind the wired-but-tired state
- Why Won’t Your Brain Shut Off at Night? The Autonomic Connection — GABA receptor impairment and sympathetic overactivation
- Why Is Your Rapid Eye Movement Sleep Fragmented? The Brainstem Switch That Controls It — the brainstem circuit and cholinergic pathway
- Does Benadryl Destroy Your Sleep? How Anticholinergic Drugs Suppress REM — anticholinergic medications and REM suppression
- What Your Overnight HRV Is Telling You About Your Sleep: The Vagal Tone Connection — HRV as a window into parasympathetic recovery
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut-vagus-brain pathway
- Can Vagus Nerve Stimulation Devices Improve Insomnia? What the Research Shows — taVNS device evidence for insomnia
- Does Your Sleep Tracker Accuracy Matter? What Oura, WHOOP, and Apple Watch Get Right and Wrong About Sleep Stages — wearable validation data
- Is Your Insomnia a Nervous System Problem? How to Tell the Difference — identifying whether your insomnia has an autonomic component
- Can Vagus Nerve Stimulation Devices Improve Insomnia? — clinical trial evidence for taVNS devices
- Does Your Sleep Tracker Accuracy Matter? — what Oura, WHOOP, and Apple Watch get right and wrong
- Is Your Insomnia a Nervous System Problem? — how to tell if autonomic dysregulation is driving sleep disruption
References
Biscarini, F., et al. (2024). Biomarkers of neurodegeneration in isolated and antidepressant-related rapid eye movement sleep behavior disorder. European Journal of Neurology, 31(6), e16260. https://pubmed.ncbi.nlm.nih.gov/38409939/
Collins, H. M., et al. (2024). Rebound activation of 5-HT neurons following SSRI discontinuation. Neuropsychopharmacology, 49(10), 1580-1589. https://pubmed.ncbi.nlm.nih.gov/38609530/
Dauvilliers, Y., et al. (2018). REM sleep behaviour disorder. Nature Reviews Disease Primers, 4, 19. https://pubmed.ncbi.nlm.nih.gov/30166532/
Feige, B., et al. (2002). Fluoxetine and sleep EEG: effects of a single dose, subchronic treatment, and discontinuation in healthy subjects. Neuropsychopharmacology, 26(2), 246-258. https://pubmed.ncbi.nlm.nih.gov/11790520/
Kluge, M., Schüssler, P., & Steiger, A. (2007). Duloxetine increases stage 3 sleep and suppresses rapid eye movement (REM) sleep in patients with major depression. European Neuropsychopharmacology, 17(8), 527-531. https://pubmed.ncbi.nlm.nih.gov/17337164/
Sharp, T., & Collins, H. (2024). Mechanisms of SSRI therapy and discontinuation. Current Topics in Behavioral Neuroscience, 66, 21-47. https://pubmed.ncbi.nlm.nih.gov/37955823/
Wichniak, A., et al. (2017). Effects of antidepressants on sleep. Current Psychiatry Reports, 19(9), 63. https://pubmed.ncbi.nlm.nih.gov/28791566/
Wilson, S., & Argyropoulos, S. (2005). Antidepressants and sleep: a qualitative review of the literature. Drugs, 65(7), 927-947. https://pubmed.ncbi.nlm.nih.gov/15892588/
Zhou, S., et al. (2023). Adverse effects of 21 antidepressants on sleep during acute-phase treatment in major depressive disorder: a systemic review and dose-effect network meta-analysis. SLEEP, 46(10), zsad177. https://pubmed.ncbi.nlm.nih.gov/37422714/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 9 references cited
