The gut-brain axis includes one pathway with direct relevance to sleep: gut bacteria producing GABA, the vagus nerve carrying that input to the brain, and central GABA receptor expression changing in response. GABA is the neurotransmitter that enables the parasympathetic transition sleep requires — it reduces neural excitability, dampens arousal, and facilitates the transition from wakefulness to sleep.
This article covers how gut bacteria produce GABA, why the vagus nerve is the required communication pathway, what human probiotic trials show for sleep outcomes, and what happens when this gut-vagus-brain pathway is disrupted. It does not cover gut inflammation, intestinal permeability, or LPS translocation — those mechanisms are covered separately. For the full autonomic overview, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It.
The gut-vagus-GABA pathway is one of several autonomic mechanisms affecting sleep. The Autonomic Sleep Disruption overview covers the broader picture, including cortisol-driven hyperarousal, parasympathetic insufficiency, and cholinergic REM regulation.
Can Gut Bacteria Produce Gamma-Aminobutyric Acid (GABA) That Affects the Brain?
GABA (gamma-aminobutyric acid) is the primary inhibitory neurotransmitter in the central nervous system. It reduces neural excitability, lowers arousal, and is required for the parasympathetic dominance that enables sleep onset and sleep maintenance. GABA does not come from the brain alone — a measurable portion is produced in the gut.
Belelli et al. (2025), writing in Brain, mapped all identified GABA communication networks across the brain-gut-microbiome axis. GABA-containing neurons make up 5-8% of the enteric nervous system — the dense network of neurons embedded in the gut wall. Bacterial genera including Bifidobacterium and Lactobacillus are documented as quantitatively relevant microbial GABA sources that influence both enteric and central GABAergic tone. These bacteria produce GABA through glutamic acid decarboxylase (GAD) enzymes, converting glutamate — an excitatory amino acid — into GABA.
The Belelli review identified three routes by which gut-derived GABA reaches the brain: vagal afferent transmission (sensory nerve fibers running from the gut to the brainstem), microbiota-mediated changes in central GABA receptor expression, and absorption into circulation. Of these, vagal afferent transmission has the strongest causal evidence — demonstrated by the Bravo et al. (2011) vagotomy experiment described in the next section.
The review also documented sex-dependent differences in GABAergic activity throughout the gut-brain axis, providing a mechanistic explanation for the higher prevalence of insomnia and anxiety in females compared to males. Gene expression analysis revealed overlap between GABA receptor subunit profiles expressed in the brain and gut, with several gut-specific subunit combinations still understudied.
A 2024 human crossover trial showed that GABA-producing probiotic strains colonize the gut and produce measurable downstream effects. Casertano et al. (2024) tested a formulation of Levilactobacillus brevis P30021 and Lactiplantibacillus plantarum P30025 in 87 healthy adults over 12 weeks. Fecal microbiota analysis showed successful probiotic colonization, with increased relative abundance of Lactiplantibacillus (p = 0.009) and Levilactobacillus (p = 0.004). Fecal GABA concentrations did not correlate with psychological outcome measures — suggesting the mechanism acts through vagal or circulatory pathways downstream of gut production rather than through local intestinal GABA levels alone.
Why Is the Vagus Nerve the Required Pathway?
The vagus nerve is the longest cranial nerve in the body, running from the brainstem to the abdomen. It is composed of approximately 80% afferent (gut-to-brain) fibers and 20% efferent (brain-to-gut) fibers, giving it a predominantly sensory surveillance role (Bonaz et al., 2018). Those afferent fibers sample microbial metabolites — including GABA, short-chain fatty acids, and neurotransmitter precursors — at the intestinal wall and relay that information to the nucleus tractus solitarius in the brainstem.
Bravo et al. (2011) conducted the experiment that demonstrated the vagus nerve as the required pathway — not an optional or supplementary one. Chronic oral administration of Lactobacillus rhamnosus JB-1 to mice produced region-dependent alterations in GABA receptor mRNA expression throughout the brain. GABA(B1b) receptor mRNA increased in cingulate and prelimbic cortical regions and decreased in hippocampal subfields — a pattern that mirrors the receptor changes associated with reduced anxiety. The probiotic also reduced stress-induced plasma corticosterone (the rodent equivalent of cortisol), indicating suppression of hypothalamic-pituitary-adrenal axis reactivity. Treated mice showed reduced anxiety-like behavior in the elevated plus maze and reduced depression-like behavior in the forced swim test.
The vagotomy control is what made this study causal. A second group of mice received the same probiotic after surgical vagotomy — severing the vagus nerve. In these mice, every neurochemical and behavioral effect was abolished. GABA receptor expression did not change. Corticosterone suppression did not occur. Behavior did not improve. The probiotic bacteria were still alive in the gut, still producing GABA — but without the vagus nerve to carry the message, nothing reached the brain.
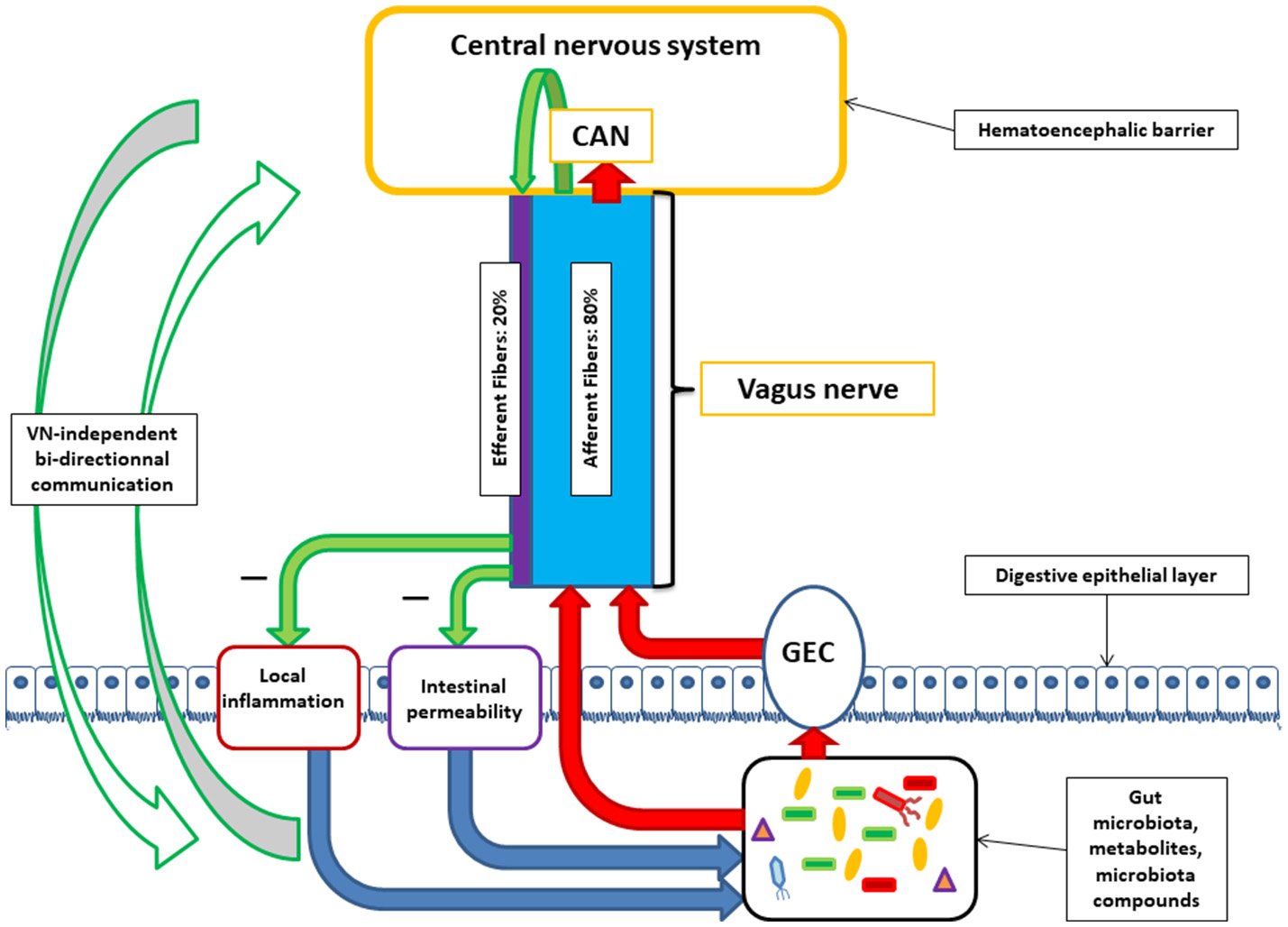
Bonaz et al. (2018) contextualized why this matters beyond a single mouse experiment. The efferent vagal fibers activate the cholinergic anti-inflammatory pathway — releasing acetylcholine that suppresses pro-inflammatory cytokines — which reduces intestinal permeability and supports a microbiota-permissive environment. Stress inhibits vagal tone while activating sympathetic outflow, increasing gut permeability and disrupting microbial composition. Reduced vagal tone has been measured in people with irritable bowel syndrome and inflammatory bowel disease — conditions characterized by dysbiosis, elevated intestinal permeability, and comorbid anxiety and sleep disturbance.
For sleep, the implication is that GABA produced by gut bacteria requires an intact, functioning vagus nerve to modulate brain GABA receptor expression. Anything that reduces vagal tone — chronic stress, inflammation, sympathetic dominance — may reduce the brain’s ability to receive this input.
Do Gamma-Aminobutyric Acid (GABA)-Producing Probiotics Improve Sleep in Humans?
Patterson et al. (2024) enrolled 89 healthy adults aged 18-45 in a randomized, double-blind, placebo-controlled trial evaluating Bifidobacterium longum 1714 over eight weeks. At the four-week interim assessment, the probiotic group showed improvements in subjective sleep quality (p < 0.05) and reduced daytime dysfunction due to sleepiness (p < 0.05) compared to placebo — suggesting rapid onset of gut-brain axis modulation. By week eight, the probiotic group demonstrated improved social functioning (p < 0.05) and energy and vitality (p < 0.05) relative to controls.
Actigraphy-based objective sleep monitoring detected no differences between groups. The improvements were in subjective perception rather than movement-derived sleep architecture metrics — a dissociation that appears consistently across the probiotic-sleep literature. B. longum 1714 is a strain with documented gut-brain axis activity, including modulation of vagal afferent activity and cortisol responses to stress.
Lan et al. (2023) conducted a separate RCT enrolling 40 adults with stress-induced insomnia. Participants received Bifidobacterium breve CCFM1025 daily for four weeks. Pittsburgh Sleep Quality Index (PSQI) scores in the probiotic group declined from a baseline mean of 11.60 to 7.75 (p = 0.0007), while the placebo group showed no statistically significant change (10.10 to 8.65, p = 0.4316). Salivary cortisol, plasma cortisol, and adrenocorticotropic hormone (ACTH) levels all declined more in the probiotic group, providing objective neuroendocrine evidence of HPA axis downregulation alongside the sleep improvements.
Liu et al. (2025) pooled data from six randomized controlled trials encompassing 424 people with insomnia in a meta-analysis. Probiotic supplementation reduced PSQI scores by a mean difference of 2.10 points compared to controls (95% CI: -3.86 to -0.34, p = 0.02) — given that PSQI scores of 5 or higher define poor sleep quality, a 2.10-point reduction crosses from poor into normal range for some participants. Hamilton Depression Rating Scale scores declined by 7.72 points in probiotic groups compared to controls (95% CI: -14.55 to -0.89, p = 0.03), reflecting the bidirectional insomnia-depression relationship.
Objective sleep architecture parameters — total sleep time, sleep efficiency, and sleep latency — did not reach statistical significance across the pooled studies in the meta-analysis. This suggests probiotics affect perceived sleep quality and mood states rather than polysomnographic sleep structure. The proposed mechanisms include normalization of gut bacterial composition, enhanced GABA and serotonin production by gut bacteria, reduced intestinal inflammation, and downstream HPA axis dampening.
What Happens to Sleep When the Gut-Vagus-Brain Pathway Is Disrupted?
The gut-vagus-brain pathway depends on two conditions: a gut microbiome that produces adequate GABA, and a vagus nerve with sufficient tone to carry that input. Disruption at either point can reduce central GABA receptor modulation and compromise the parasympathetic conditions sleep requires.
Bonaz et al. (2018) documented that stress inhibits vagal tone while activating sympathetic outflow. The efferent vagal pathway normally suppresses pro-inflammatory cytokines through the cholinergic anti-inflammatory pathway. When vagal tone drops, intestinal permeability can increase, microbial composition can change, and the capacity of gut bacteria to communicate with the brain through the vagus nerve declines. In practice, stress reduces vagal tone, reduced vagal tone disrupts the gut environment, and the disrupted gut environment further reduces the GABA input reaching the brain.
Jiang et al. (2025) demonstrated the bidirectional nature of this disruption. Sleep deprivation disrupted intestinal barrier integrity and increased inflammatory markers in the gut. GABA-producing postbiotics from Levilactobacillus brevis reversed these effects — restoring intestinal barrier integrity, reducing inflammatory markers, and suppressing neuroinflammatory gene expression in the brain. This study provides evidence that the gut-sleep relationship runs in both directions: disrupted sleep harms the gut, and restoring gut GABA production can reverse some of the damage sleep deprivation causes.
The Casertano et al. (2024) crossover trial adds a relevant detail. Probiotic supplementation reduced rumination scores (p = 0.006) — a measure of repetitive negative thought cycling that is a recognized driver of sleep-onset difficulty and nocturnal awakenings. Rumination is a form of cognitive hyperarousal, and its reduction through a gut-targeted approach suggests the gut-vagus-brain pathway may modulate sleep not only through GABA receptor effects but also through downstream cognitive processes.
The Lan et al. (2023) cortisol data support this: the decline in salivary cortisol, plasma cortisol, and ACTH in the probiotic group indicates that gut-brain axis modulation can dampen HPA axis reactivity — the same cortisol elevation that drives the stress-insomnia loop. A disrupted gut-vagus pathway may therefore contribute to insomnia through multiple downstream effects: reduced central GABA receptor expression, sustained cognitive arousal, and inadequate HPA axis suppression.
The gut-vagus-GABA pathway may not be the only autonomic mechanism affecting your sleep. Parasympathetic insufficiency, cortisol-driven hyperarousal, GABA receptor impairment, or medication-induced REM suppression might also be contributing. When multiple causes overlap, identifying which ones might be active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Which Probiotic Strains Produce Gamma-Aminobutyric Acid (GABA)?
GABA production in the gut is enzyme-dependent — bacteria that carry the glutamic acid decarboxylase (GAD) gene convert glutamate into GABA. Multiple species within the Lactobacillus and Bifidobacterium genera carry this gene, but not all strains within a species produce GABA at the same rate or with the same downstream effects.
Lactobacillus rhamnosus JB-1 is the strain with the strongest mechanistic evidence for vagal GABA transmission — it is the strain used in the Bravo et al. (2011) vagotomy study that demonstrated the vagus nerve as the required pathway. Bifidobacterium longum 1714 improved subjective sleep quality in healthy adults at four weeks (Patterson et al., 2024). Bifidobacterium breve CCFM1025 reduced PSQI scores from 11.60 to 7.75 and lowered cortisol in adults with stress-induced insomnia (Lan et al., 2023). Levilactobacillus brevis P30021 and Lactiplantibacillus plantarum P30025 reduced rumination in a 12-week crossover trial (Casertano et al., 2024).
Strain specificity matters. The probiotic literature shows that effects are strain-dependent — a Lactobacillus product that does not contain a GAD-active strain may not produce GABA at the concentrations needed to influence vagal transmission.
Can Addressing Gut Health Resolve Insomnia?
The meta-analysis by Liu et al. (2025) found that probiotics improved subjective sleep quality (PSQI mean difference of -2.10) but did not change objective sleep architecture parameters — total sleep time, sleep efficiency, and sleep latency did not reach significance across pooled trials. This pattern — subjective improvement without objective architecture changes — suggests the gut-vagus-GABA pathway modulates the conditions that support sleep (reducing arousal, dampening HPA axis reactivity, lowering rumination) rather than restructuring sleep stages.
Insomnia typically involves multiple contributing factors — autonomic imbalance, hormonal changes, circadian disruption, inflammatory processes. The gut-vagus-GABA pathway is one contributor within that picture. For someone whose insomnia has a strong stress-gut-arousal component, restoring gut GABA production through targeted probiotic strains may improve sleep quality. For someone whose insomnia is driven primarily by cortisol dysregulation or circadian misalignment, gut-targeted strategies may offer partial benefit rather than full resolution.
Can Sleep Deprivation Damage the Gut?
Jiang et al. (2025) documented that sleep deprivation produces measurable harm to the gut. Intestinal barrier integrity — the tight junctions between cells lining the intestinal wall — was disrupted, and inflammatory markers increased. The GABA-producing postbiotics reversed both effects: intestinal barrier integrity was restored, inflammatory markers declined, and neuroinflammatory gene expression in the brain was suppressed.
This bidirectionality means that insomnia and gut disruption can reinforce each other. Poor sleep compromises the gut environment, a compromised gut environment reduces GABA production and vagal communication, reduced GABA and vagal communication impair the parasympathetic conditions for sleep, and impaired sleep further compromises the gut. Breaking this cycle at the gut level — through dietary changes, targeted probiotic supplementation, or reducing factors that disrupt microbial composition — may help interrupt the reinforcement.
Does the Gut Produce Enough Gamma-Aminobutyric Acid (GABA) to Affect Sleep?
The question is not whether the gut produces enough GABA to flood the brain with an inhibitory neurotransmitter — that is not how the pathway works. The Bravo et al. (2011) vagotomy study demonstrated that gut bacteria alter GABA receptor expression in the brain rather than delivering GABA molecules to neural tissue. The probiotic changed how many GABA receptors were expressed in the cortex, hippocampus, amygdala, and locus coeruleus — changing the brain’s sensitivity to GABA, not the amount of GABA present.
Belelli et al. (2025) described this as a multi-route communication pathway: vagal afferent fibers detect gut-produced GABA and relay the information centrally, microbial metabolites enter portal circulation and influence GABA-glutamate balance, and sustained microbial GABA production modulates central receptor gene expression over time. The 5-8% GABA-neuron composition of the enteric nervous system, combined with bacterial GABA production, provides a continuous source of vagal input that is sufficient to modulate brain receptor expression — as the Bravo vagotomy study proved by showing that removing the vagal connection eliminated the brain effects entirely.
Related Reading:
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — how autonomic imbalance fragments sleep architecture and what drives it, including vagal tone, GABA, and neuroinflammation pathways
- Can Your Nervous System Get Stuck in Fight or Flight and Ruin Your Sleep? — the self-reinforcing cortisol-sleep loop and how chronic stress locks the autonomic nervous system into sustained sympathetic activation
- What Is Hyperarousal Insomnia? Why You’re Wired but Tired Every Night — how hyperarousal differs from general insomnia, cortical and cognitive markers, and why “tired but wired” reflects measurable brain activity
- Why Won’t Your Brain Shut Off at Night? The Autonomic Connection — why the brain stays active at bedtime, the role of GABA receptor impairment in autonomic sleep regulation
- Why Is Your Rapid Eye Movement Sleep Fragmented? The Brainstem Switch That Controls It — how the brainstem flip-flop mechanism regulates REM-NREM transitions and what disrupts it
- Does Benadryl Destroy Your Sleep? How Anticholinergic Drugs Suppress REM Sleep — how anticholinergic medications suppress REM sleep by blocking acetylcholine, and why sedation is not the same as restorative sleep
- What Your Overnight HRV Is Telling You About Your Sleep: The Vagal Tone Connection — what heart rate variability during sleep reflects about parasympathetic recovery and how to interpret overnight HRV trends
- Does Benadryl Degrade Your Sleep? How Anticholinergic Drugs Suppress Rapid Eye Movement Sleep — how anticholinergic medications block the muscarinic receptors REM requires
- What Your Overnight Heart Rate Variability Is Telling You About Your Sleep: The Vagal Tone Connection — using HRV as a window into parasympathetic recovery during sleep
- Do Antidepressants Suppress Rapid Eye Movement Sleep? — SSRIs, SNRIs, and tricyclics affect sleep architecture
- Can Vagus Nerve Stimulation Devices Improve Insomnia? — clinical trial evidence for taVNS devices
- Does Your Sleep Tracker Accuracy Matter? — what Oura, WHOOP, and Apple Watch get right and wrong
- Is Your Insomnia a Nervous System Problem? — how to tell if autonomic dysregulation is driving sleep disruption
References
Belelli, D., Bhatt, D. K., Butler, E., Duguid, I. C., Bhatt, S. R., & Lambert, J. J. (2025). From bugs to brain: unravelling the GABA signalling networks in the brain-gut-microbiome axis. Brain, 148(5), 1479-1498. https://pubmed.ncbi.nlm.nih.gov/39716883/
Bonaz, B., Bazin, T., & Pellissier, S. (2018). The vagus nerve at the interface of the microbiota-gut-brain axis. Frontiers in Neuroscience, 12, 49. https://pubmed.ncbi.nlm.nih.gov/29467611/
Bravo, J. A., Forsythe, P., Chew, M. V., Escaravage, E., Savignac, H. M., Dinan, T. G., Bienenstock, J., & Cryan, J. F. (2011). Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proceedings of the National Academy of Sciences USA, 108(38), 16050-16055. https://pubmed.ncbi.nlm.nih.gov/21876150/
Casertano, M., Fogliano, V., & Ercolini, D. (2024). GABA-producing lactobacilli boost cognitive reactivity to negative mood without improving cognitive performance: A human double-blind placebo-controlled cross-over study. Brain, Behavior, and Immunity, 122, 324-336. https://pubmed.ncbi.nlm.nih.gov/39163908/
Jiang, Y., Li, S., Zhu, Y., Liu, Y., Zhang, M., & Wang, J. (2025). GABA-producing postbiotics from Levilactobacillus brevis reverse sleep deprivation-induced intestinal and neuroinflammatory damage. Frontiers in Microbiology, 16, 1563908. https://pubmed.ncbi.nlm.nih.gov/40613302/
Lan, Y., Lu, J., Zhang, T., Guo, Y., Liao, Z., Jia, C., & Chen, W. (2023). Bifidobacterium breve CCFM1025 improves sleep quality via regulating the activity of the HPA axis: A randomized clinical trial. Nutrients, 15(21), 4700. https://pubmed.ncbi.nlm.nih.gov/37960353/
Liu, Y., Wang, L., Zhang, S., & Hu, X. (2025). Impact of probiotics on sleep quality and mood states in patients with insomnia: A systematic review and meta-analysis. Frontiers in Microbiology, 16, 1596478. https://pubmed.ncbi.nlm.nih.gov/40740336/
Patterson, E., Griffin, S. M., Ibarra, A., Ellsiepen, E., & Hellhammer, J. (2024). Bifidobacterium longum 1714 improves sleep quality and aspects of well-being in healthy adults: A randomized, double-blind, placebo-controlled clinical trial. Scientific Reports, 14(1), 3037. https://pubmed.ncbi.nlm.nih.gov/38355674/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 8 references cited
