Growth hormone peaks during deep sleep, but not everyone gets the same pulse. People with excess visceral fat — the fat surrounding abdominal organs — produce less growth hormone during sleep, even when their total body weight is similar to someone who produces normal amounts. This metabolic pattern is rooted in fat distribution rather than body weight or age.
This article covers how visceral adiposity suppresses growth hormone secretion through elevated free fatty acids, heightened somatostatin tone, and reduced ghrelin activity — and how sleep restriction drives further visceral fat accumulation, completing a self-reinforcing cycle. It does not cover the age-related GH change covered in the hormonal men cluster.
Visceral fat disrupting the growth hormone-sleep axis is one of several metabolic mechanisms that fragment sleep. The full picture is in the Metabolic Sleep Disruption pillar.
When Does Growth Hormone Peak During Sleep and What Controls the Pulse?
A 2025 study in Cell used optogenetic and calcium imaging techniques on hypothalamic neurons to identify the mechanism: during NREM sleep, somatostatin (SST) neurons — which normally suppress growth hormone — reduce their firing rate, while growth hormone-releasing hormone (GHRH) neurons increase theirs. This creates a narrow window where the pituitary gland can release growth hormone without inhibition (Ding et al., 2025).
During REM sleep, both GHRH and SST neurons fire simultaneously, producing a lower-amplitude GH output. The large pulse — the one that accounts for the majority of the day’s growth hormone — depends on NREM-specific somatostatin withdrawal.
Growth hormone also has a self-limiting feedback effect: after the GH pulse, growth hormone activates locus coeruleus neurons that promote wakefulness. Each GH pulse carries its own termination mechanism (Ding et al., 2025).
In a study of 149 healthy men aged 16 to 83, nocturnal GH secretion was statistically associated with the amount of slow-wave sleep (P < 0.001). SWS dropped from 18.9% of total sleep time in early adulthood (ages 16-25) to 3.4% by midlife (ages 36-50), with GH secretion declining by 372 micrograms per decade during that same window (Van Cauter et al., 2000).
This NREM-dependent circuit is the target that visceral fat disrupts. By amplifying somatostatin tone, visceral adiposity prevents the SST withdrawal needed for the NREM growth hormone pulse.
How Does Visceral Fat Suppress Growth Hormone Secretion?
The clearest direct evidence comes from a controlled study of 24 premenopausal women divided into three groups: viscerally obese, small visceral fat area, and normal-weight controls. Women with large visceral fat area showed a 4-fold reduction in mean plasma growth hormone compared to both comparison groups. The women with small visceral fat area — despite having higher total body fat than lean controls — maintained GH levels equivalent to lean controls (Pijl et al., 2001).
This study points to the topography of fat deposition as the determinant of GH suppression. GH half-life and volume of distribution were identical across all three groups, ruling out altered clearance as an explanation. The suppression was driven by diminished basal and pulsatile secretion — reduced growth hormone output from the pituitary.
A study of 42 healthy women (20 premenopausal, 22 postmenopausal) isolated visceral fat as an important predictor. Abdominal visceral fat mass alone explained 22% of the variability in GHRH-stimulated growth hormone secretion (P = 0.002), after controlling for menstrual status and age. The mechanism appears to involve enhanced somatostatin inhibitory tone: visceral fat blunts the pituitary’s response to GHRH (which requires somatostatin withdrawal) but does not impair its response to GHRP-2 (which bypasses the somatostatin gate). This dissociation points to somatostatin amplification as the primary pathway (Veldhuis et al., 2009).
A third mechanism comes through ghrelin. Ghrelin functions both as the body’s own GH-releasing peptide and as a slow-wave sleep promoter. In obesity, baseline acylated ghrelin levels are chronically lower than in lean individuals. This reduces both the SWS-promoting and GH-releasing arms of ghrelin activity simultaneously (Ditmer et al., 2026).
These peripheral suppression pathways — elevated free fatty acids, hyperinsulinemia, heightened somatostatin tone, and reduced ghrelin — all converge on the same hypothalamic GHRH/SST circuit mapped by Ding et al. (2025). Visceral fat prevents the somatostatin withdrawal that NREM sleep normally produces, blocking the growth hormone pulse at its source.
Does Poor Sleep Drive Visceral Fat Accumulation?
The intracellular mechanism was identified in a 2024 study. Sleep deprivation suppresses SIRT1, an NAD+-dependent deacetylase enzyme. SIRT1 normally deacetylates and activates FOXO1, a transcription factor that upregulates ATGL (adipose triglyceride lipase) — the rate-limiting enzyme for breaking down stored fat. When sleep deprivation suppresses SIRT1, this entire chain stalls: FOXO1 remains inactive, ATGL expression drops, and triglycerides accumulate in visceral white adipose tissue because stored fat cannot be mobilized even without caloric surplus (Wang et al., 2024).
Resveratrol, a pharmacological SIRT1 activator, restored the lipolytic pathway and reversed the fat accumulation in sleep-deprived mice — providing mechanistic evidence that SIRT1 suppression is the causal link between sleep loss and visceral fat storage (Wang et al., 2024).
At the population level, NHANES data from 14,021 U.S. adults showed a dose-response relationship between visceral adiposity and sleep disorders. Participants in the highest visceral adiposity index (VAI) quartile had 21% greater odds of sleep disorders compared to the lowest quartile (OR = 1.21; 95% CI 1.03-1.41). The relationship was linear — each unit increase in VAI was associated with a 3% higher risk of sleep disorders in the fully adjusted model (OR = 1.03; 95% CI 1.00-1.05) (Liu et al., 2025).
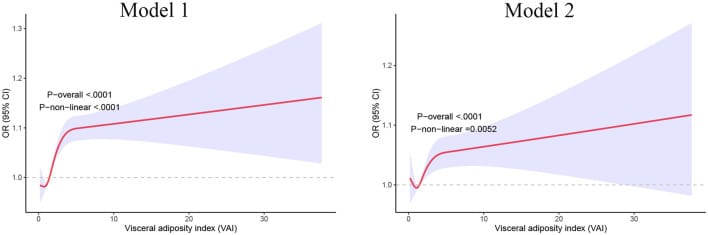
The proposed biological mechanism: visceral adipose tissue releases pro-inflammatory cytokines — TNF-alpha and IL-1beta — that follow circadian rhythms peaking at 01:00-02:00h and are regulators of slow-wave sleep architecture. Elevated cytokine load from high visceral fat disrupts these overnight sleep-regulating processes (Liu et al., 2025).
The cycle maps directly: visceral fat suppresses GH through somatostatin amplification and ghrelin suppression (Pijl et al., 2001; Veldhuis et al., 2009). Reduced GH and fragmented SWS impair the SIRT1/FOXO1/ATGL lipolytic pathway (Wang et al., 2024). Impaired lipolysis drives more visceral fat accumulation. More visceral fat further suppresses GH. Each fragmented night reinforces the cycle.
The SWS and GH decline in men accelerates during the fourth decade — the same window when visceral fat accumulation also accelerates (Van Cauter et al., 2000). The cycle may begin during this period, with metabolic and sleep deterioration reinforcing each other from midlife onward.
Does Cortisol Block Growth Hormone Release?
In 149 healthy men, evening cortisol rose by 19.3 nmol/L per decade beginning after age 50 (P < 0.001). The chronologies of SWS decline and cortisol rise are distinct: SWS declines between ages 16 and 50, while the cortisol rise begins after age 50. This suggests the two processes are regulated independently — cortisol compounds an existing GH deficit rather than initiating it (Van Cauter et al., 2000).
Cortisol elevation promotes wakefulness and suppresses NREM depth, shrinking the somatostatin-withdrawal window that the GH pulse requires. This is the same circuit that visceral fat disrupts peripherally through elevated free fatty acids and hyperinsulinemia — cortisol disrupts it centrally by preventing the deep NREM state from being reached or sustained (Ding et al., 2025).
Chronic insomnia compounds ghrelin suppression. People with chronic insomnia have approximately 30% lower nocturnal ghrelin than controls. Lower ghrelin reduces both SWS promotion and GH-release drive — stress-driven insomnia suppresses the ghrelin bridge between sleep and growth hormone (Ditmer et al., 2026).
In people with high visceral adiposity, the inflammatory cytokines TNF-alpha and IL-1beta peak at 01:00-02:00h — overlapping with the cortisol nadir window. In high-VAI individuals, cytokine-driven inflammation may prevent cortisol from reaching its normal overnight low, further compressing the window available for GH secretion (Liu et al., 2025).
The combination creates compounded suppression: visceral fat amplifies somatostatin tone peripherally, cortisol suppresses NREM depth centrally, and ghrelin suppression from both obesity and chronic sleep disruption weakens the remaining GH-release drive. Each pathway reduces GH independently, but in someone with high visceral fat and elevated evening cortisol, the effects stack.
Visceral fat, cortisol elevation, insulin resistance, and reduced ghrelin activity might each be contributing to growth hormone suppression during sleep — and they rarely appear in isolation. The same metabolic environment that reduces your GH pulses might also be fragmenting your deep sleep through several of these pathways at once. Find out which causes might be driving your 3am wakeups
Frequently Asked Questions
How Do You Maximize Growth Hormone During Sleep?
Factors that support SWS depth and growth hormone secretion based on the research covered in this article: reducing visceral adiposity, managing insulin resistance, regular exercise (which both reduces visceral fat and increases SWS depth), and avoiding late-night meals that elevate insulin during the sleep window. Sleep-fragmenting conditions like sleep apnea directly reduce the NREM time available for GH secretion — addressing those conditions removes a mechanical barrier to the GH pulse.
The full set of metabolic factors affecting sleep is covered in the Metabolic Sleep Disruption pillar.
What Happens to Growth Hormone if You Wake Up at 3am?
In the hypothalamic circuit, GH itself activates locus coeruleus neurons that promote wakefulness — so the GH pulse carries a self-limiting feedback effect (Ding et al., 2025). If visceral fat is driving the sleep fragmentation, each disrupted night further suppresses GH, which over time contributes to more visceral fat accumulation through the SIRT1/FOXO1/ATGL pathway described above.
Does Losing Weight Restore Growth Hormone Secretion?
While reducing visceral fat improves GH output, the suppression may not be fully reversible with weight loss alone. One interpretation: reduced GH secretion may be partly a primary trait predisposing to visceral fat storage, not solely a consequence of it. The Pijl et al. study measured a single time point post-weight-loss, and longer-term recovery data is limited.
Does Visceral Fat Affect Sleep Even Without Weight Gain?
Fat distribution matters more than total weight for the growth hormone-sleep connection. Waist circumference or the visceral adiposity index — which integrates waist circumference, BMI, and lipid parameters — may be more informative than BMI alone for assessing metabolic risk to sleep and GH secretion.
Related Reading
- Metabolic Sleep Disruption
- Can a Blood Sugar Drop Wake You Up at 3am?
- Why Do You Wake Up Hungry at 3am?
- Why Does Cortisol Spike at 3am and Wake You Up?
- Can a Calorie Deficit Cause Insomnia?
- Adrenal Fatigue and 3am Waking
- Insulin Resistance and Insomnia
- Prediabetes and Sleep Problems
- CGM, Nocturnal Glucose, and Sleep
- Intermittent Fasting, Blood Sugar, and Sleep
- Does Keto Cause 3am Waking?
- Dawn Phenomenon in Non-Diabetics
- Fatty Liver and Sleep Problems
- Gut Dysbiosis, Blood Sugar, and Sleep
- Late-Night Eating, Blood Sugar, and Sleep
References
Ding, X., Hwang, F. J., Silverman, D., Zhong, P., Li, B., Ma, C., Lu, L., Jiang, G., Zhang, Z., Huang, X., Tu, X., Tian, Z. M., Ding, J., & Dan, Y. (2025). Neuroendocrine circuit for sleep-dependent growth hormone release. Cell, 188(18), 4968-4979.e12. https://pubmed.ncbi.nlm.nih.gov/40562026/
Ditmer, M., Wojtera, A., Gabryelska, A., Turkiewicz, S., Białasiewicz, P., Strzelecki, D., & Sochal, M. (2026). Ghrelin’s role in sleep and sleep deprivation: a narrative review. Frontiers in Psychiatry, 17, 1744781. https://pubmed.ncbi.nlm.nih.gov/41696469/
Liu, C., Qiu, L., Wang, T., Ye, Z., Wu, S., Li, D., Lin, H., & Jin, Y. (2025). Association between visceral adiposity index and sleep disorders among the U.S. adults: a cross-sectional study. Frontiers in Neurology, 16, 1540182. https://pubmed.ncbi.nlm.nih.gov/40417111/
Pijl, H., Langendonk, J. G., Burggraaf, J., Frölich, M., Cohen, A. F., Veldhuis, J. D., & Meinders, A. E. (2001). Altered neuroregulation of GH secretion in viscerally obese premenopausal women. The Journal of Clinical Endocrinology and Metabolism, 86(11), 5509-5515. https://pubmed.ncbi.nlm.nih.gov/11701729/
Van Cauter, E., Leproult, R., & Plat, L. (2000). Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA, 284(7), 861-868. https://pubmed.ncbi.nlm.nih.gov/10938176/
Veldhuis, J. D., Hudson, S. B., Erickson, D., Bailey, J. N., Reynolds, G. A., & Bowers, C. Y. (2009). Relative effects of estrogen, age, and visceral fat on pulsatile growth hormone secretion in healthy women. American Journal of Physiology. Endocrinology and Metabolism, 297(2), E367-E374. https://pubmed.ncbi.nlm.nih.gov/19470834/
Wang, W., Liu, K., Xu, H., Zhang, C., Zhang, Y., Ding, M., Xing, C., Huang, X., Wen, Q., Lu, C., & Song, L. (2024). Sleep deprivation induced fat accumulation in the visceral white adipose tissue by suppressing SIRT1/FOXO1/ATGL pathway activation. Journal of Physiology and Biochemistry, 80(3), 561-572. https://pubmed.ncbi.nlm.nih.gov/38856814/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 7 references cited
