The concept of “post-infection sleep syndrome” was proposed in the journal SLEEP in 2026 (Sun, Mullington, & Thomas, 2026), using long COVID as the paradigm case. The term is recent, but post-viral insomnia itself is not new. It has been documented after the original SARS coronavirus, Epstein-Barr virus, influenza, and other infections for decades. What the 2026 proposal formalized is the clinical recognition that post-infectious sleep disruption is a distinct syndrome involving central nervous system and autonomic pathways.
This article covers how viral infections disrupt autonomic sleep regulation, the objective sleep evidence across multiple viruses (not only COVID), the alpha-EEG sleep anomaly that characterizes post-viral fatigue, and the role of EBV reactivation. For the full autonomic model, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It. Post-viral autonomic damage is one of several causes covered in that pillar; this article goes deeper on the infection-specific pathway.
How Do Viruses Damage Autonomic Nerve Function?
The autonomic damage is not COVID-specific. Milovanovic et al. (2025) enrolled 440 participants — 210 with chronic fatigue syndrome of unknown etiology, 137 with post-COVID chronic fatigue, and 91 healthy controls. Both post-viral fatigue groups showed reduced baroreceptor sensitivity and impaired heart rate variability compared to controls (p < 0.05). Extreme blood pressure variations during head-up tilt testing occurred in 45-47% of both groups, and positive tilt test results indicating syncope susceptibility were found in approximately 71% of both groups (Milovanovic et al., 2025).
Moldofsky and Patcai (2011) documented the same pattern after the original SARS coronavirus — 22 Toronto subjects (21 of whom were healthcare workers) who remained unable to return to work 13-36 months after infection. Polysomnography revealed sleep instability and the alpha-EEG anomaly previously associated with fibromyalgia and ME/CFS (Moldofsky & Patcai, 2011).
Three damage pathways emerge across the literature:
- Direct viral neural infection. SARS-CoV-2 can infect vagal tissue through ACE2 receptors expressed on vagal neurons, producing direct injury to the nerve fibers that carry parasympathetic input from the brainstem to the heart, lungs, and gut.
- Autoimmune-mediated nerve injury. Post-viral immune activation can produce antibodies that attack small nerve fibers — a condition called small fiber neuropathy — damaging the autonomic fibers responsible for heart rate regulation and sleep-wake transitions.
- Chronic neuroinflammation. McCarthy (2022) proposed TGF-beta dysregulation as a molecular mechanism linking coronavirus infections to persistent circadian and sleep disruption. The 2003 SARS outbreak produced ME/CFS-like syndromes in 27-40% of survivors, with circadian and sleep features consistent with the pattern now documented after COVID (McCarthy, 2022).
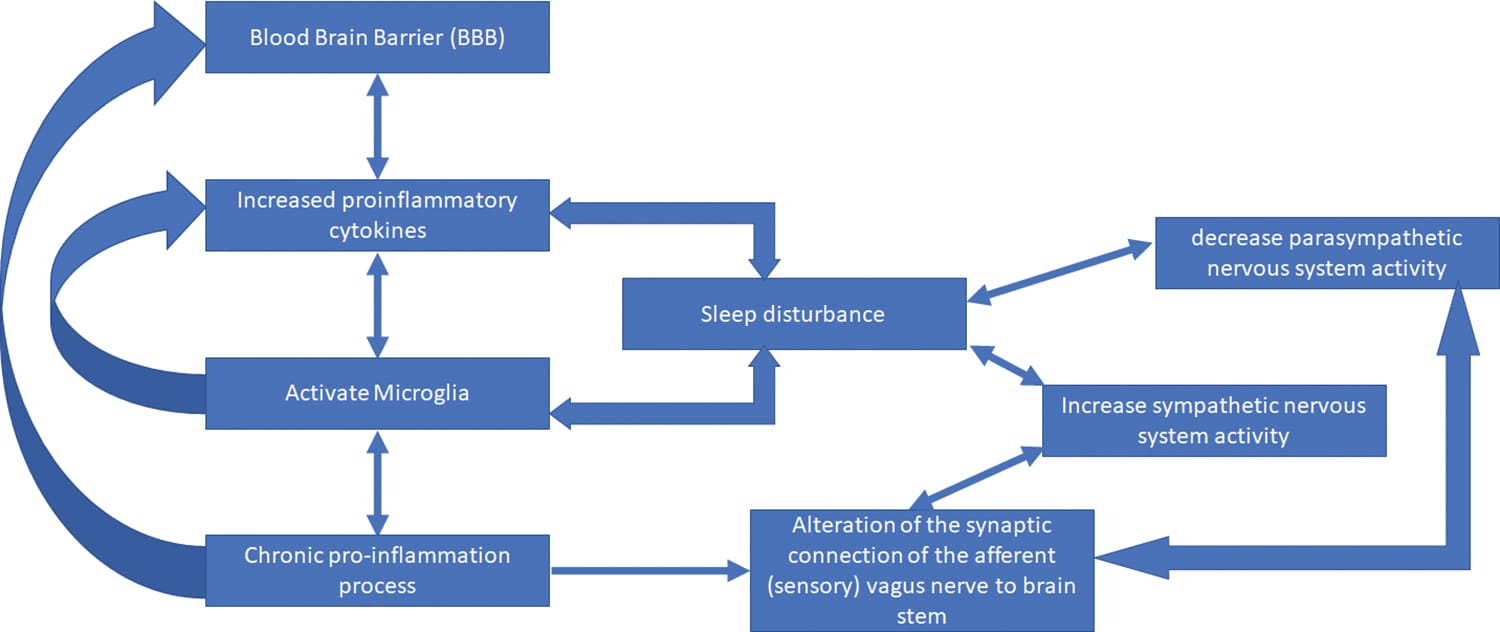
All three pathways converge on the same outcome: the parasympathetic tone required for sleep onset, NREM maintenance, and overnight cardiac regulation is impaired. How long COVID damages the vagus nerve and disrupts sleep covers the COVID-specific vagal damage mechanism in detail.
What Does Post-Viral Sleep Architecture Look Like on Polysomnography?
Mohamed et al. (2023) synthesized 24 case-control studies examining objective sleep parameters (18 using polysomnography, 6 using actigraphy) in ME/CFS. Across 20 adult studies, adult ME/CFS participants had 4.5% lower sleep efficiency than controls, with reduced stage 2 NREM, elevated stage 3 deep sleep, and prolonged REM latency. Adolescent ME/CFS participants showed a similar 4.5% reduction in a smaller sample, though this finding did not reach statistical significance (p = 0.25) (Mohamed et al., 2023).
That pattern does not match primary insomnia or depression. In primary insomnia, sleep architecture is relatively preserved. In depression, REM latency is shortened and deep sleep is reduced. In post-viral fatigue, the architecture itself is altered in a way that points to autonomic and neuroinflammatory mechanisms.
What Is the Alpha Electroencephalogram Sleep Anomaly in Post-Viral Fatigue?
Whelton, Salit, and Moldofsky (1992) documented one of the more distinctive polysomnographic findings in post-viral fatigue: the alpha-EEG anomaly. Their study examined 14 ME/CFS participants and 12 healthy controls. All 14 ME/CFS participants displayed a prominent alpha-EEG NREM anomaly (p < 0.001) — an intrusion of alpha-frequency brain activity (7.5-11.0 Hz, activity associated with relaxed wakefulness) into restorative slow-wave sleep stages (Whelton et al., 1992).
In practical terms: the sleep stages register as NREM, but the brain maintains a partial arousal state. Alpha waves intrude into delta sleep, the deepest restorative stage. Sleep looks adequate on a tracker but does not produce the restoration that deep sleep should deliver. This is the electrophysiological basis for “I slept eight hours but feel like I did not sleep.”
The alpha-EEG anomaly has since been documented across ME/CFS, fibromyalgia, post-SARS fatigue (Moldofsky & Patcai, 2011), and post-COVID fatigue.
Does Epstein-Barr Virus Reactivation After COVID Compound Sleep Disruption?
Palomo et al. (2026) analyzed 873 serum samples from 40 ME/CFS participants and 378 from 16 healthy controls. ME/CFS participants showed elevated dUTPase IgG antibodies to EBV, HHV-6, and varicella zoster virus (p < 0.001). 72.5% showed simultaneous antibodies to multiple herpesviruses, compared to 31% of controls. Antibody levels correlated with fatigue severity, and longitudinal sampling confirmed chronic reactivation rather than residual antibodies (Palomo et al., 2026).
For someone with a history of EBV infection (the majority of adults worldwide), COVID can produce a dual viral burden. COVID damages the vagus nerve directly through ACE2-mediated infection of vagal tissue. Reactivated EBV drives persistent immune activation, tryptophan depletion, and neuroinflammation. Both pathways converge on sleep disruption and compound each other.
Kadl et al. (2025) documented the scale: among 280 people with long COVID evaluated using the Insomnia Severity Index, insomnia was present in 50% at the initial visit and 42% at follow-up — both prevalent and persistent rather than self-resolving (Kadl et al., 2025). Waking up with a racing heart after COVID covers the nighttime autonomic experience in detail.
Are There Different Types of Post-Viral Sleep Disruption?
Coelho et al. (2024) evaluated 42 long COVID participants using diagnostic sleep testing. Five mechanistically distinct sleep disorder types emerged: obstructive sleep apnea, chronic insomnia disorder, primary hypersomnia (including narcolepsy type 2), REM behavior disorder, and circadian phase delay (Coelho et al., 2024).
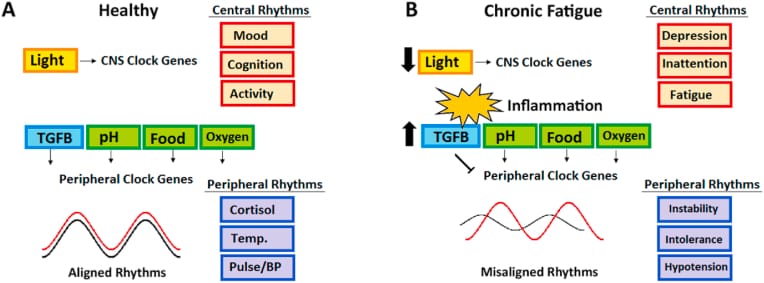
Rauwerda et al. (2024) compared insomnia phenotypes between post-COVID and ME/CFS populations directly. Insomnia was present in 64% of post-COVID participants. Post-COVID participants had shorter subjective sleep duration than ME/CFS participants (p = 0.003) — a difference suggesting that these insomnia phenotypes, while overlapping, are not identical (Rauwerda et al., 2024).
Mooren et al. (2023) identified the autonomic signature underlying several of these types. Their 24-hour HRV study found impaired parasympathetic activity during nighttime hours in post-COVID participants, with sympathetic overactivation exceeding measures in people with established cardiac disease. People hospitalized during acute infection showed more pronounced damage — suggesting that initial infection severity influences the degree of long-term autonomic impairment (Mooren et al., 2023). POTS, dysautonomia, and sleep after COVID covers the condition-specific autonomic pathway.
Post-viral autonomic impairment may not be the only factor affecting your sleep. GABA receptor changes, metabolic disruptions, inflammatory processes, or circadian misalignment may also be contributing. When multiple causes overlap, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups –>
Frequently Asked Questions
How Long Does Post-Viral Insomnia Last?
The Moldofsky 2011 post-SARS study is the longest follow-up with objective polysomnographic data: all 22 participants showed persistent sleep architecture disruption 13-36 months after infection (Moldofsky & Patcai, 2011). The Kadl 2025 data showed insomnia prevalence decreasing from 50% to 42% between visits — partial recovery in some cases, persistence in the majority (Kadl et al., 2025).
Can the Flu Cause Chronic Insomnia?
The broader post-viral fatigue literature has documented chronic fatigue conditions with circadian and autonomic features following multiple viral infections. McCarthy (2022) focused on how TGF-beta dysregulation after coronavirus infections disrupts circadian rhythms, a mechanism likely shared across viral triggers. The Milovanovic et al. (2025) ME/CFS cohort (n=210) was classified as chronic fatigue syndrome of unknown etiology, and their autonomic impairment profiles were comparable to the post-COVID group (Milovanovic et al., 2025).
What Is the Difference Between Post-Viral Insomnia and Depression-Related Insomnia?
This distinction matters because both conditions can look similar on a sleep questionnaire but differ on polysomnography. In post-viral insomnia, REM latency is prolonged and the NREM stages show alpha intrusion. In depression, REM latency is shortened, with increased REM density and reduced deep sleep. These patterns point to different neurochemical mechanisms: autonomic and neuroinflammatory in post-viral insomnia, serotonergic and cholinergic in depression.
The Mohamed 2023 meta-analysis confirmed that the post-viral ME/CFS sleep architecture pattern is statistically distinct from depression across the included studies (Mohamed et al., 2023).
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Does Lion’s Mane Affect Your Sleep? Why It Helps Some People and Keeps Others Awake — lion’s mane’s dual mechanism: neurotrophic activity, cholinergic activity, and sleep architecture
- Why Do Cholinergic Supplements Give You Vivid Dreams? — the acetylcholine-REM pathway behind supplement-related vivid dreams
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — how post-viral autonomic disruption can affect sleep continuity
- Why Does Your Heart Race at Night After COVID? What the Vagus Nerve Has to Do With It — why post-COVID autonomic instability can show up as nighttime tachycardia
- Can POTS and Dysautonomia Cause Insomnia? Why Your Autonomic Nervous System Will Not Let You Rest — how orthostatic intolerance and autonomic instability can fragment sleep
- Why Will Your Nervous System Not Let You Sleep? Polyvagal Theory and Insomnia — how polyvagal states map to insomnia, hyperarousal, and unrefreshing sleep
- Why Does Trauma Cause Insomnia? How Your Nervous System Stays on Guard at Night — how trauma can keep sleep shaped by hyperarousal and vigilance
- Why Are You Sleeping 12 Hours and Still Exhausted? The Dorsal Vagal Shutdown Explained — the dorsal vagal pattern behind long sleep that still feels unrefreshing
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- How Do Probiotics Reach Your Brain? The Vagotomy Evidence — what vagotomy studies show about gut-brain communication
- Does Poor Sleep Damage Your Gut Microbiome? — the relationship between sleep fragmentation, microbiome disruption, and inflammatory load
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
Coelho, F. M. S., Czuma, R., Ticotsky, A., Maley, J., Mullington, J. M., & Thomas, R. J. (2024). Sleep disorder syndromes of post-acute sequelae of SARS-CoV-2 (PASC) / Long Covid. Sleep Medicine, 123, 37-41. https://pubmed.ncbi.nlm.nih.gov/39236463/
Kadl, A., Davis, E. M., Oliver, S. F., Lazoff, S. A., Popovich, J., Atya, A. A. E., Enfield, K. B., & Quigg, M. (2025). Prevalence and associations of insomnia after COVID-19 infection. Journal of Clinical Sleep Medicine, 21(2), 383-391. https://pubmed.ncbi.nlm.nih.gov/39436395/
McCarthy, M. J. (2022). Circadian rhythm disruption in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Implications for the post-acute sequelae of COVID-19. Brain, Behavior, & Immunity – Health, 20, 100412. https://pubmed.ncbi.nlm.nih.gov/35465246/
Mohamed, A. Z., Andersen, T., Radovic, S., Del Fante, P., Kwiatek, R., Calhoun, V., Bhuta, S., Hermens, D. F., Lagopoulos, J., & Shan, Z. Y. (2023). Objective sleep measures in chronic fatigue syndrome: A systematic review and meta-analysis. Sleep Medicine Reviews, 69, 101771. https://pubmed.ncbi.nlm.nih.gov/36948138/
Moldofsky, H., & Patcai, J. (2011). Chronic widespread musculoskeletal pain, fatigue, depression and disordered sleep in chronic post-SARS syndrome; a case-controlled study. BMC Neurology, 11, 37. https://pubmed.ncbi.nlm.nih.gov/21435231/
Milovanovic, B., Markovic, N., Petrovic, M., Zugic, V., Ostojic, M., Rankovic-Nicic, L., & Bojic, M. (2025). Assessment of Autonomic Nervous System Function in Patients with Chronic Fatigue Syndrome and Post-COVID-19 Syndrome Presenting with Recurrent Syncope. Journal of Clinical Medicine, 14(3), 811. https://pubmed.ncbi.nlm.nih.gov/39941481/
Mooren, F. C., Bockelmann, I., Waranski, M., Kotewitsch, M., Teschler, M., Schafer, H., & Schmitz, B. (2023). Autonomic dysregulation in long-term patients suffering from Post-COVID-19 Syndrome assessed by heart rate variability. Scientific Reports, 13(1), 15814. https://pubmed.ncbi.nlm.nih.gov/37739977/
Palomo, I. M., Cox, B., Williams, M. V., & Ariza, M. E. (2026). Chronic Reactivation of Persistent Human Herpesviruses EBV, HHV-6 and VZV and Heightened Anti-dUTPase IgG Antibodies Are a Recurrent Hallmark in Post-Infectious ME/CFS and is Associated With Fatigue. Journal of Medical Virology, 98(1), e70769. https://pubmed.ncbi.nlm.nih.gov/41451845/
Rauwerda, N. L., Kuut, T. A., Braamse, A. M. J., Csorba, I., Nieuwkerk, P., van Straten, A., & Knoop, H. (2024). Insomnia and sleep characteristics in post COVID-19 fatigue: A cross-sectional case-controlled study. Journal of Psychosomatic Research, 177, 111522. https://pubmed.ncbi.nlm.nih.gov/38113796/
Sun, H., Mullington, J. M., & Thomas, R. J. (2026). Post-infection sleep syndrome: long COVID as an example. SLEEP, 49(3), zsaf366. https://pubmed.ncbi.nlm.nih.gov/41239971/
Whelton, C. L., Salit, I., & Moldofsky, H. (1992). Sleep, Epstein-Barr virus infection, musculoskeletal pain, and depressive symptoms in chronic fatigue syndrome. The Journal of Rheumatology, 19(6), 939-943. https://pubmed.ncbi.nlm.nih.gov/1328633/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 11 references cited
