Vivid dreams are among the frequently reported effects of cholinergic supplements. People in lucid dreaming communities seek this effect on purpose. People taking alpha-GPC or lion’s mane for cognition are often caught off guard by it. Both groups tend to lack the underlying explanation.
This article covers the acetylcholine-REM mechanism, which supplements produce this effect and through what pathways, and whether vivid dreams indicate healthy or impaired sleep. It does not cover the full autonomic sleep framework — for that, see How Does Autonomic Nervous System Dysfunction Disrupt Sleep?. Cholinergic tone is one of several autonomic factors controlling sleep architecture, and the pillar article covers the broader picture.
Why Does Acetylcholine Control How Vivid Your Dreams Are?
The acetylcholine cycle across sleep stages is not flat. It follows a pattern that mirrors the NREM-REM alternation. In a 2001 microdialysis study in cats, Vazquez and Baghdoyan measured basal forebrain acetylcholine release across precisely defined sleep and wake states. Acetylcholine output during REM sleep (0.77 +/- 0.07 pmol/10 min) was approximately 33% higher than during waking (0.58 +/- 0.03 pmol/10 min). During NREM, it dropped to more than 40% below waking levels (0.34 +/- 0.01 pmol/10 min) (Vazquez & Baghdoyan, 2001). While this was measured in cats, the same directional pattern — high acetylcholine during REM, low during NREM — is consistent with human pharmacological data.
That NREM trough matters. Acetylcholine needs to drop to near-zero during deep sleep for NREM-specific processes — including memory consolidation — to function. It then surges during REM to drive the cortical activation that produces dreaming. This alternation between low and high cholinergic tone is what makes the sleep cycle work.
The receptor side supports this. A 2018 study by Niwa and colleagues used CRISPR gene editing to knock out muscarinic acetylcholine receptors in mice. Removing both Chrm1 (M1) and Chrm3 (M3) receptors virtually abolished REM sleep. Neither receptor alone was sufficient — both were genetically required for REM generation (Niwa et al., 2018). This is a mouse model, so direct receptor-level evidence in humans remains limited. But the finding aligns with human pharmacological evidence: cholinergic input is not incidental to REM — it is a requirement at the receptor level.
In healthy humans, the same relationship holds. Hohagen and colleagues (1993) administered an oral muscarinic agonist (SDZ 210-086) to 12 healthy male volunteers and measured polysomnographic sleep parameters. At the 1.0 mg dose, the drug shortened REM latency, increased total REM duration, and increased the percentage of REM sleep within total sleep time — while decreasing slow-wave sleep. An additional finding: REM density (number of eye movements per unit of REM time) decreased even as total REM expanded, indicating a dissociation between tonic and phasic REM components under cholinergic stimulation (Hohagen et al., 1993).
The takeaway: vivid dreams on cholinergic supplements are amplified REM activity. When you increase acetylcholine availability — whether through a precursor or an enzyme inhibitor — you are intensifying the neurotransmitter that drives REM onset, duration, and cortical activation during dreaming.
Which Supplements Raise Acetylcholine and Affect Dream Quality?
Do Acetylcholine Precursors Affect Dreams?
Alpha-GPC contains approximately 40% choline by weight and crosses the blood-brain barrier with high efficiency, making it one of the faster routes to elevated brain acetylcholine. A 2025 review of alpha-GPC’s mechanisms documented its capacity for CNS penetration and its role as a direct choline donor for acetylcholine synthesis and phosphatidylcholine membrane biosynthesis in neurons (Li et al., 2025). A 2024 randomized crossover trial in healthy males found that a single 630 mg dose of alpha-GPC improved selective attention and cognitive flexibility on the Stroop test (mean improvement 13.0 vs. 5.2 for placebo, p = 0.013), consistent with acute cholinergic enhancement (Kerksick, 2024).
CDP-choline (citicoline) follows a dual pathway — it supplies choline for acetylcholine synthesis but also influences dopamine and norepinephrine metabolism. This dual action can produce a different subjective profile than alpha-GPC alone.
Choline bitartrate has lower blood-brain barrier penetration than alpha-GPC. It raises peripheral choline levels but delivers less choline to the brain, producing milder cognitive and dream effects at equivalent doses.
How Do Acetylcholinesterase Inhibitors Affect Dream Vividness?
Galantamine has the strongest controlled evidence for dream effects among cholinergic supplements. In a double-blind, placebo-controlled crossover study of 121 participants, LaBerge and colleagues (2018) found that galantamine produced dose-dependent increases in lucid dreaming: 14% at placebo, 27% at 4 mg (odds ratio 2.29), and 42% at 8 mg (odds ratio 4.46). Galantamine also increased dream recall, sensory vividness, bizarreness, and narrative complexity. Galantamine inhibits acetylcholinesterase, increasing the acetylcholine available in the synapse during REM (LaBerge et al., 2018). It is also a positive allosteric modulator of nicotinic acetylcholine receptors, which may further amplify cholinergic transmission.
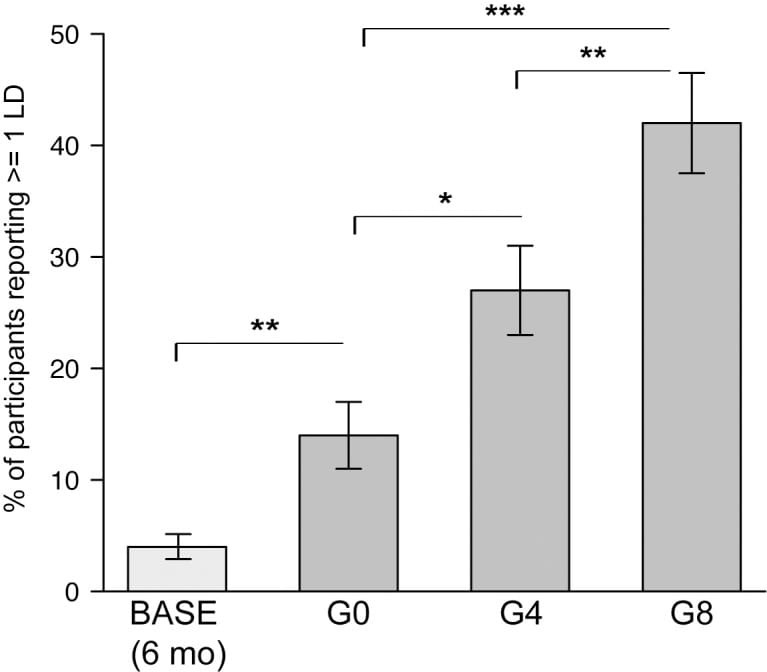
Huperzine A inhibits acetylcholinesterase with a long half-life, which may extend its effects across multiple sleep cycles. The 2024 review by Oldoni and colleagues identified huperzine A alongside galantamine as a primary candidate for pharmacological lucid dream induction, noting that acetylcholinesterase inhibitors can increase dream lucidity, recall, bizarreness, and visual vividness with generally low side-effect profiles (Oldoni et al., 2024).
Lion’s mane contains compounds (erinacerin A and hericenone B) that have been identified as potential acetylcholinesterase inhibitors in computational modeling studies. This is one proposed reason some people report vivid dreams on lion’s mane supplements — the cholinergic mechanism may overlap with galantamine’s, though the potency would differ.
Why Do Inhibitors Produce Stronger Dream Effects Than Precursors?
Precursors add raw material. Inhibitors amplify whatever cholinergic activity is already happening. During REM, when endogenous acetylcholine release is at its peak, an acetylcholinesterase inhibitor preserves more of that acetylcholine in the synapse — producing a more targeted amplification of REM-stage neurotransmission than a precursor does.
Combination risk: Stacking a precursor (alpha-GPC) with an inhibitor (galantamine or huperzine A) compounds the cholinergic load. More choline available for synthesis, combined with slower breakdown, can produce intense dream effects and may alter the NREM-REM balance more than either approach alone.
Are Vivid Dreams a Sign of Good or Bad Sleep Quality?
The answer depends on what is driving the vividness.
A 2025 computational modeling study by Satchell and colleagues examined how acetylcholine’s differential levels across NREM and REM produce complementary memory functions. During NREM, when acetylcholine is low, reduced inhibition of excitatory networks enables synchronized bursting that recruits additional neurons into memory engrams — broadening encoding. During REM, when acetylcholine is high, increased inhibition restricts excitatory firing, pruning engram populations and sharpening memory representations. The model predicts that NREM-REM cycling is functionally optimal because it alternates between these two neurochemical states in sequence (Satchell et al., 2025). This is a computational model, not a direct measurement in sleeping humans — but it generates testable predictions consistent with what pharmacological studies show.
This matters for supplement users because exogenous acetylcholine keeps levels elevated during NREM — the phase when acetylcholine should be low. If the NREM trough is blunted, the consolidation phase that depends on low cholinergic tone may be impaired. Even though REM may become more vivid, the overall sleep benefit can be reduced because both phases need to function in sequence.
Gott and colleagues (2024) developed a related framework linking acetylcholine to metacognitive awareness during sleep. Their review examined how acetylcholine regulates fluctuations in consciousness during sleep, including the role of cholinergic activity in lucid dreaming (Gott et al., 2024). This suggests that heightened dream vividness may reflect elevated cholinergic tone during REM — which tells you something about neurotransmitter availability but not necessarily about whether the broader sleep architecture is intact.
The practical indicator: if vivid dreams are accompanied by daytime fatigue, fragmented sleep, or frequent awakenings, the cholinergic input may be impairing architecture rather than enhancing it. Vivid dreams without those accompanying effects are less concerning. The distinction is whether the elevated cholinergic tone is limited to REM or is bleeding into NREM stages where it does not belong.
How Does the Brain Switch Between Deep Sleep and Dream Sleep?
The NREM-REM alternation is driven by two competing neuron populations in the brainstem. Cholinergic neurons in the pedunculopontine and laterodorsal tegmental nuclei promote REM sleep. Aminergic neurons — releasing serotonin and norepinephrine — promote wakefulness and NREM. GABA-mediated inhibition between these populations creates a reciprocal relationship: when one group is active, the other is suppressed.
Across a typical night, this reciprocal inhibition produces approximately 90-minute cycles. Early in the night, NREM dominance is strongest — deep slow-wave sleep occupies larger portions of each cycle. As the night progresses, the balance tilts toward REM. The first REM period may last 10 minutes; the final one before waking can last 30-60 minutes.
Exogenous acetylcholine tips this balance. When a cholinergic supplement raises acetylcholine availability, it strengthens the REM-promoting side of the reciprocal circuit. The Hohagen et al. (1993) data showed this directly: the muscarinic agonist shortened REM latency and expanded total REM duration in healthy humans. The Vazquez and Baghdoyan (2001) microdialysis data provides the mechanistic basis — acetylcholine release during REM already exceeds waking levels, so adding more amplifies a neurotransmitter that is already at its peak.
For a detailed discussion of the brainstem cholinergic-aminergic switch and how REM fragmentation occurs from the endogenous side, see Why Is Your REM Sleep Fragmented? The Brainstem Switch That Controls Dream Sleep.
Timing matters. When a supplement is taken relative to sleep onset determines which cycles are affected. A dose taken at bedtime raises acetylcholine during the first NREM-dominant cycles, potentially shortening deep sleep in the first half of the night. A dose taken after 4-5 hours of sleep — as in the LaBerge galantamine study — targets the REM-dominant second half, when the supplement’s cholinergic effects align with the brain’s natural acetylcholine surge.
Cholinergic supplement effects on REM architecture might not be the only factor affecting your sleep. Autonomic hyperarousal, GABA changes, metabolic disruptions, or inflammatory processes might also be contributing. When multiple causes overlap, identifying which ones might be active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can Nootropics Permanently Change Your Dreams?
The LaBerge et al. (2018) galantamine study measured effects acutely — within a single night after a single dose. No long-term follow-up data has shown persistent changes in dream architecture after discontinuation. The cholinergic enhancement is pharmacologically time-limited: galantamine’s half-life is approximately 7 hours, huperzine A’s is approximately 10-14 hours, and alpha-GPC’s cholinergic effects are tied to acute choline availability. Once the supplement clears, acetylcholine metabolism returns to its endogenous rhythm.
Why Do Vivid Dreams Happen in the Early Morning Hours?
The Vazquez and Baghdoyan (2001) microdialysis data established that acetylcholine release is highest during REM and lowest during NREM. Because later sleep cycles contain proportionally more REM and less deep sleep, the cholinergic surge in the final third of the night is more sustained. A supplement that raises acetylcholine availability compounds this natural pattern — the exogenous increase is added on top of an endogenous peak, producing vivid dreams in the early morning hours when REM is already at its longest.
Do Choline Supplements Cause Nightmares?
The LaBerge et al. (2018) study measured emotional valence alongside dream vividness. Lucid dreams showed higher emotional positivity, greater sense of control, and more self-reflection compared to non-lucid dreams. But the key word is “on average” — individual responses varied. Increasing cholinergic tone amplifies the sensory and emotional intensity of whatever the dream contains. For people whose dream content tends toward anxiety or stress-related themes, that amplification can manifest as more vivid nightmares. The mechanism is the same whether the emotional content is positive or negative: elevated acetylcholine during REM intensifies perceptual and emotional processing.
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Does Lion’s Mane Affect Your Sleep? Why It Helps Some People and Keeps Others Awake — lion’s mane’s dual mechanism: neurotrophic activity, cholinergic activity, and sleep architecture
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — how post-viral autonomic disruption can affect sleep continuity
- Why Does Your Heart Race at Night After COVID? What the Vagus Nerve Has to Do With It — why post-COVID autonomic instability can show up as nighttime tachycardia
- Can POTS and Dysautonomia Cause Insomnia? Why Your Autonomic Nervous System Will Not Let You Rest — how orthostatic intolerance and autonomic instability can fragment sleep
- Can a Viral Infection Cause Chronic Insomnia? — how viral infections can disrupt autonomic regulation and sleep
- Why Will Your Nervous System Not Let You Sleep? Polyvagal Theory and Insomnia — how polyvagal states map to insomnia, hyperarousal, and unrefreshing sleep
- Why Does Trauma Cause Insomnia? How Your Nervous System Stays on Guard at Night — how trauma can keep sleep shaped by hyperarousal and vigilance
- Why Are You Sleeping 12 Hours and Still Exhausted? The Dorsal Vagal Shutdown Explained — the dorsal vagal pattern behind long sleep that still feels unrefreshing
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- How Do Probiotics Reach Your Brain? The Vagotomy Evidence — what vagotomy studies show about gut-brain communication
- Does Poor Sleep Damage Your Gut Microbiome? — the relationship between sleep fragmentation, microbiome disruption, and inflammatory load
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
- Vazquez, J., & Baghdoyan, H. A. (2001). Basal forebrain acetylcholine release during REM sleep is significantly greater than during waking. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology, 280(2), R598–R601. https://pubmed.ncbi.nlm.nih.gov/11208592/
- Niwa, Y., Kanda, G. N., Yamada, R. G., Shi, S., Sunagawa, G. A., Ukai-Tadenuma, M., Fujishima, H., Matsumoto, N., Masumoto, K., Nagano, M., Kasukawa, T., Galloway, J., Perrin, D., Shigeyoshi, Y., Ukai, H., Kiyonari, H., Sumiyama, K., & Ueda, H. R. (2018). Muscarinic acetylcholine receptors Chrm1 and Chrm3 are essential for REM sleep. Cell Reports, 24(9), 2231–2247.e7. https://pubmed.ncbi.nlm.nih.gov/30157420/
- Hohagen, F., Riemann, D., Spiegel, R., Holzhauer, M., & Berger, M. (1993). Influence of the cholinergic agonist SDZ 210-086 on sleep in healthy subjects. Neuropsychopharmacology, 9(3), 225–232. https://pubmed.ncbi.nlm.nih.gov/8280346/
- LaBerge, S., LaMarca, K., & Baird, B. (2018). Pre-sleep treatment with galantamine stimulates lucid dreaming: A double-blind, placebo-controlled, crossover study. PLoS ONE, 13(8), e0201246. https://pubmed.ncbi.nlm.nih.gov/30089135/
- Oldoni, A. A., Bacchi, A. D., Mendes, F. R., Tiba, P. A., & Mota-Rolim, S. (2024). Neuropsychopharmacological induction of (lucid) dreams: A narrative review. Brain Sciences, 14(5), 426. https://pubmed.ncbi.nlm.nih.gov/38790404/
- Gott, J. A., Stucker, S., Kanske, P., Haaker, J., & Dresler, M. (2024). Acetylcholine and metacognition during sleep. Consciousness and Cognition, 117, 103608. https://pubmed.ncbi.nlm.nih.gov/38042119/
- Satchell, M., Butel-Fry, E., Noureddine, Z., Simmons, A., Ognjanovski, N., Aton, S. J., & Zochowski, M. R. (2025). Cholinergic modulation of neural networks supports sequential and complementary roles for NREM and REM states in memory consolidation. PLoS Computational Biology, 21(6), e1013097. https://pubmed.ncbi.nlm.nih.gov/40526789/
- Kerksick, C. M. (2024). Acute alpha-glycerylphosphorylcholine supplementation enhances cognitive performance in healthy men. Nutrients, 16(23), 4240. https://pubmed.ncbi.nlm.nih.gov/39683633/
- Li, J., Zhang, J., Wang, Y., Yang, Y., Su, Y., Gu, L., & Chang, C. (2025). L-Alpha-glycerylphosphorylcholine (L-a-GPC): A comprehensive review of its preparation techniques and versatile biological effects. Journal of Food Science, 90(6), e70338. https://pubmed.ncbi.nlm.nih.gov/40556032/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 9 references cited
