“My doctor says it’s anxiety, but it feels physical.” That disconnect drives millions of searches. When insomnia persists despite addressing stress, sleep hygiene, and psychological factors, the question becomes whether the nervous system itself is the problem — and the distinction has implications for brain health, longevity, and which approach will help.
This article covers the evidence that insomnia can be a nervous system condition, how to distinguish autonomic-driven insomnia from other types, what measurable markers indicate nervous system involvement, and where to go next. It does not cover the full autonomic overview — for that, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It. Autonomic dysregulation is one of several causes of sleep disruption; the parent article covers the broader picture.
What Is the Difference Between Psychological Insomnia and Nervous System Insomnia?
Psychophysiological insomnia follows a conditioned arousal model. The bed becomes a cue for wakefulness. Racing thoughts at bedtime and difficulty falling asleep develop through repeated pairing of the sleep environment with frustration and alertness. Autonomic markers — heart rate, cortisol — may be within normal range during the daytime.
Nervous system insomnia follows the hyperarousal model. Riemann et al. (2010) proposed that people with chronic insomnia have 24-hour hyperarousal: elevated cortisol, increased whole-body metabolic rate, higher heart rate, and EEG fast-frequency activity during NREM sleep. This is not a nighttime-only phenomenon — the overactivation persists through waking hours as well.
Dressle and Riemann (2023) updated the evidence base for this model. Their review confirmed that multiple physiological measurements — HPA axis output, autonomic nervous system activity, and EEG spectral power — show persistent overactivation in people with chronic insomnia. The term “psychophysiological insomnia” may itself be misleading, because the condition has measurable physical correlates that extend beyond conditioned behavioral patterns.
The older classification of “primary vs. secondary insomnia” — where insomnia was viewed as a byproduct of another condition — is also increasingly questioned. Current evidence suggests insomnia itself drives autonomic changes in a bidirectional relationship: the nervous system overactivation worsens sleep, and the disrupted sleep amplifies the overactivation (Rösler et al., 2024). This makes the primary/secondary distinction less useful for identifying causes.
Why this distinction matters in practice: CBT-I (cognitive behavioral therapy for insomnia) addresses the conditioned arousal component — the learned association between bed and wakefulness. When insomnia is also driven by autonomic overactivation, addressing the nervous system component directly — through vagal tone improvement, HRV-responsive practices, or stress-axis modulation — may be needed alongside behavioral approaches.
What Does the Research Show About Nervous System Overactivation in Chronic Insomnia?
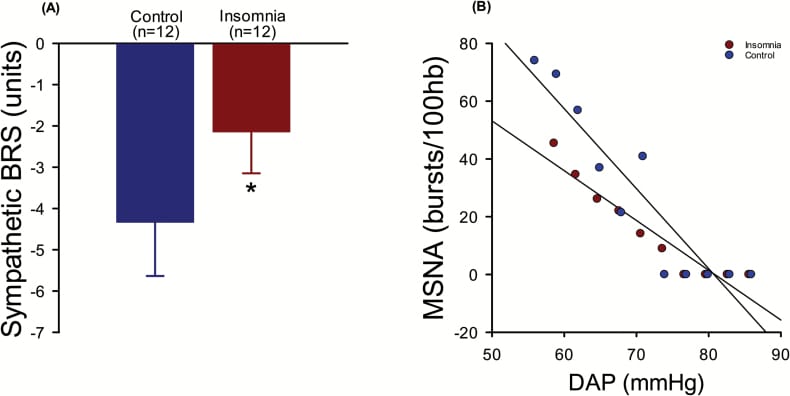
A direct nerve-level measurement comes from Carter et al. (2018), who used microneurography — microelectrodes inserted into the peroneal nerve — to measure sympathetic nerve firing directly. People with chronic insomnia showed elevated muscle sympathetic nerve activity (MSNA) compared to matched controls. This is not inferred from heart rate or cortisol — it is measured at the nerve level. The study also found that sympathetic baroreflex sensitivity was lower in the insomnia group, meaning the cardiovascular regulation mechanism that should buffer against excessive sympathetic activation was impaired.
EEG data adds a cortical dimension. Riemann et al. (2010) documented that people with insomnia show increased beta and gamma power during NREM sleep — the brain generates waking-type electrical activity while the person is technically asleep. Jang et al. (2024) extended this finding to daytime waking EEG: elevated beta and gamma power during a 5-minute resting-state recording predicted insomnia severity independently of anxiety and depression scores (incremental R² = 0.041, p = 0.018). This means the cortical hyperarousal is distinguishable from the hyperarousal associated with emotional distress.
At sleep onset, the autonomic transition into sleep is measurably impaired. Ma et al. (2024) studied 328 people and found that those with insomnia had higher heart rate (p = 0.004), lower overall heart rate variability (p = 0.003), and reduced parasympathetic variation (p < 0.001) during the sleep-onset period, independent of sleep apnea. The normal transition from sympathetic to parasympathetic dominance that enables sleep initiation is blunted.
The 24-hour pattern ties these findings together. Dressle and Riemann (2023) confirmed that cortisol output, metabolic rate, and autonomic markers are elevated throughout the day in people with chronic insomnia — not just at night. Rösler et al. (2024), tracking 207 participants over 9 consecutive days, found that the overnight accumulation of physiological and emotional arousal was larger in people with insomnia than in controls. Poor sleep did not reset the arousal — it amplified it into the next day.
How Can You Tell if Your Insomnia Is Caused by Your Nervous System?
Pattern 1: Tired but wired. Physical fatigue with mental alertness at bedtime. Homeostatic sleep drive — the accumulation of adenosine during wakefulness — produces the sensation of tiredness. But the autonomic nervous system controls the transition into sleep. When sympathetic tone remains elevated — heart rate up, cortisol up, muscle tension sustained — the transition cannot complete even when sleep pressure is high. The Carter et al. (2018) data on elevated sympathetic nerve firing provides the physiological basis: the nervous system is maintaining an arousal state that overrides sleep pressure.
Pattern 2: Wearable data. Consumer wearables (Oura, WHOOP, Apple Watch) can reveal autonomic patterns during sleep. Di Marco et al. (2024) studied 3,165 people with polysomnography and found that individuals with insomnia had a higher probability of waking from every sleep stage — N1, N2, N3, and REM — reflecting global instability in sleep state maintenance. Their novel machine learning metric (WESI — Wake Electroencephalographic Similarity Index) confirmed that insomnia involves persistent intrusion of wake-state brain activity throughout the sleep period. On a consumer level, this pattern shows up as elevated resting heart rate during sleep, low overnight HRV, and absence of the normal heart rate dip in the first half of the night.
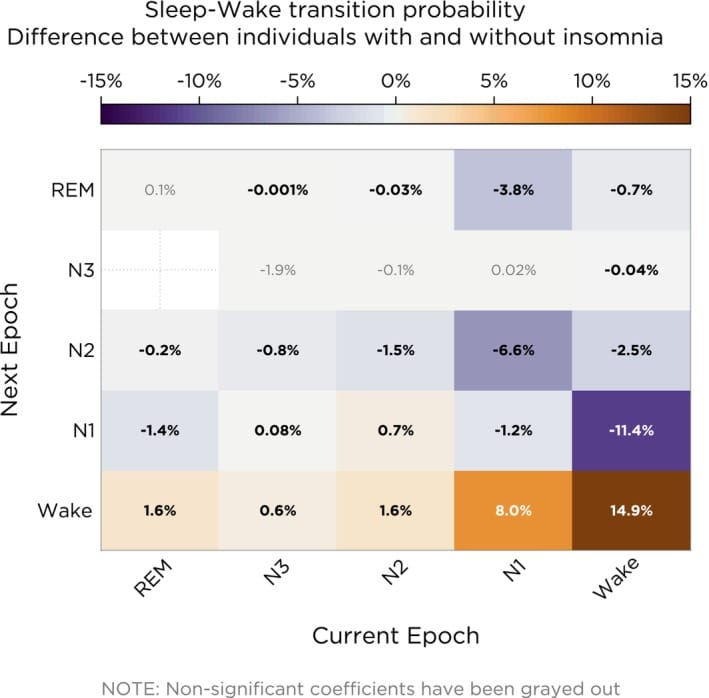
Pattern 3: Normal sleep duration, non-restorative sleep. Some people with autonomic-driven insomnia log 7-8 hours of sleep yet wake feeling unrested. The EEG hyperarousal evidence explains why — the brain generates waking-type electrical activity during NREM, producing sleep that looks adequate by duration but does not provide full recovery. Di Marco et al. (2024) showed that the WESI score — measuring how much sleep EEG resembles wakefulness — was higher across all sleep stages in people with insomnia compared to controls.
Pattern 4: Insomnia that does not respond to sleep hygiene. Standard recommendations — dark room, consistent schedule, no screens before bed, cool temperature — address sleep environment and behavioral cues. When these changes produce little improvement, the issue may be upstream: in the nervous system’s arousal state, not in the behavioral habits surrounding sleep.
This section is not a formal evaluation. It identifies patterns consistent with nervous system involvement that can inform what to explore next. For people with dysautonomia or POTS, the connection to autonomic function is often already recognized — roughly 90% of people with POTS report sleep disruption, and the autonomic mechanism is well understood in that population.
What Medical Conditions Produce Insomnia Through Nervous System Mechanisms?
Dysautonomia and POTS. Autonomic instability directly disrupts the sympathetic-parasympathetic balance needed for sleep. Roughly 90% of people with POTS report sleep disruption. The autonomic nervous system cannot execute the transition from daytime sympathetic dominance to nighttime parasympathetic dominance that sleep initiation requires. Nano et al. (2017), in their review of 34 studies, found that cardiovascular autonomic patterns in insomnia vary by phenotype — and that grouping heterogeneous insomnia presentations together obscures measurable physiological differences. Conditions with known autonomic instability (like POTS) represent one end of that phenotypic spectrum.
Post-traumatic stress disorder. Sustained sympathetic activation from the trauma response maps directly to the hyperarousal model. The Dressle and Riemann (2023) evidence showing elevated cortisol and autonomic markers in chronic insomnia applies to PTSD-driven insomnia as well — the nervous system maintains a sustained arousal state that prevents the autonomic transition into sleep.
The “neurologist or psychiatrist?” question. The appropriate evaluation depends on the suspected mechanism. A neurologist is appropriate when autonomic indicators extend beyond insomnia — heart rate instability, temperature dysregulation, orthostatic intolerance — suggesting a broader autonomic condition. A psychiatrist is appropriate when insomnia coexists with mood disorders, PTSD, or anxiety that requires pharmacological management. A sleep specialist (for polysomnography and actigraphy) is the starting referral for persistent insomnia that has not been formally evaluated. These are not mutually exclusive — the autonomic mechanism may coexist with psychological factors, and evaluation by more than one type of specialist may be appropriate.
When to pursue further evaluation: persistent insomnia lasting longer than 3 months that does not respond to CBT-I or sleep hygiene changes; presence of other autonomic indicators (heart rate variability abnormalities, orthostatic intolerance, temperature dysregulation); or a history of conditions known to affect the autonomic nervous system.
Autonomic-driven insomnia rarely exists in isolation. Nervous system overactivation might compound with metabolic changes, hormonal fluctuations, inflammatory processes, or circadian disruption. Many people have more than one cause contributing to their sleep disruption. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Is Insomnia a Mental Illness or a Physical Condition?
The classification of insomnia has evolved. Older models categorized insomnia as a byproduct of another condition — depression, anxiety, pain. The ICSD-3 (International Classification of Sleep Disorders, third edition) now recognizes insomnia as a disorder in its own right, with cognitive, behavioral, and physiological components that interact. The psychological-vs-physical framing is increasingly viewed as a false dichotomy: a person can have conditioned arousal (psychological) and elevated sympathetic nerve activity (physical) simultaneously. Riemann et al. (2010) documented this overlap — cortical, autonomic, and neuroendocrine dimensions are co-occurring but conceptually distinct contributors to chronic insomnia.
What Is Hyperarousal Insomnia?
Hyperarousal insomnia is distinct from situational insomnia (caused by acute stress, jet lag, or a temporary disruption) and from sleep-state misperception (where polysomnography records normal sleep but the person experiences it as poor). In hyperarousal insomnia, the overactivation is a trait — it does not resolve by removing a single stressor. Dressle and Riemann (2023) reviewed evidence across multiple measurement domains (HPA axis, autonomic nervous system, EEG) and found persistent overactivation patterns that remained stable over time. Rösler et al. (2024), tracking participants over 9 days, confirmed that people with insomnia scored higher on a composite hyperarousal factor than controls at every measurement point across all 9 days. For a deeper examination of the wired-but-tired mechanism, see What Is Hyperarousal Insomnia? Why You’re Wired but Tired Every Night.
Why Can You Not Sleep Even When You Are Exhausted?
Sleep regulation involves two main processes: homeostatic sleep pressure (Process S), which builds with time awake, and the circadian rhythm (Process C), which determines the timing window for sleep. Autonomic arousal can override both. The Carter et al. (2018) microneurography data showed that people with insomnia had elevated sympathetic nerve firing even during seated rest — meaning the nervous system maintains arousal that competes with sleep pressure. This is why exhaustion and sleeplessness can coexist: the drive to sleep is present, but the nervous system’s arousal state prevents the transition.
Should You See a Neurologist or Psychiatrist for Insomnia?
A practical framework: (1) persistent insomnia alone — a sleep specialist for polysomnography and structured evaluation is the starting point. (2) Insomnia with autonomic indicators — heart rate instability, orthostatic intolerance, temperature dysregulation — a neurological evaluation for autonomic conditions is appropriate. (3) Insomnia coexisting with mood disorders, anxiety, or PTSD — a psychiatrist and/or sleep specialist may be appropriate. (4) Multiple overlapping factors — a multidisciplinary approach may be needed. CBT-I has demonstrated efficacy across insomnia subtypes (Dressle & Riemann, 2023) and can be pursued alongside specialist evaluation rather than deferred.
Related Reading:
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — how autonomic imbalance disrupts sleep architecture
- Can Your Nervous System Get Stuck in Fight or Flight and Ruin Your Sleep? — the self-reinforcing cortisol-sleep loop
- What Is Hyperarousal Insomnia? Why You’re Wired but Tired Every Night — autonomic mechanics behind the wired-but-tired state
- Why Won’t Your Brain Shut Off at Night? The Autonomic Connection — GABA receptor impairment and sympathetic overactivation
- Why Is Your Rapid Eye Movement Sleep Fragmented? The Brainstem Switch That Controls It — the brainstem circuit and cholinergic pathway
- Does Benadryl Destroy Your Sleep? How Anticholinergic Drugs Suppress Rapid Eye Movement Sleep — anticholinergic medications and REM suppression
- What Your Overnight Heart Rate Variability Is Telling You About Your Sleep: The Vagal Tone Connection — HRV as a window into parasympathetic recovery
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut-vagus-brain pathway
- Do Antidepressants Suppress Rapid Eye Movement Sleep? What SSRIs, SNRIs, and Tricyclics Do to Sleep Architecture — serotonergic REM suppression from medications
- Can Vagus Nerve Stimulation Devices Improve Insomnia? What the Research Shows — taVNS device evidence for insomnia
- Does Your Sleep Tracker Accuracy Matter? What Oura, WHOOP, and Apple Watch Get Right and Wrong About Sleep Stages — wearable validation data
- Do Antidepressants Suppress Rapid Eye Movement Sleep? — SSRIs, SNRIs, and tricyclics affect sleep architecture
- Can Vagus Nerve Stimulation Devices Improve Insomnia? — clinical trial evidence for taVNS devices
- Does Your Sleep Tracker Accuracy Matter? — what Oura, WHOOP, and Apple Watch get right and wrong
References
Bonnet, M. H., & Arand, D. L. (2010). Hyperarousal and insomnia: state of the science. Sleep Medicine Reviews, 14(1), 9-15. https://pubmed.ncbi.nlm.nih.gov/19640748/
Carter, J. R., Grimaldi, D., Fonkoue, I. T., Medalie, L., Mokhlesi, B., & Van Cauter, E. (2018). Assessment of sympathetic neural activity in chronic insomnia: evidence for elevated cardiovascular risk. Sleep, 41(6), zsy048. https://pubmed.ncbi.nlm.nih.gov/29522186/
Di Marco, T., Scammell, T. E., Sadeghi, K., Datta, A. N., Little, D., Tjiptarto, N., … & Dauvilliers, Y. (2024). Hyperarousal features in the sleep architecture of individuals with and without insomnia. Journal of Sleep Research, 34(1), e14256. https://pubmed.ncbi.nlm.nih.gov/38853521/
Dressle, R. J., & Riemann, D. (2023). Hyperarousal in insomnia disorder: Current evidence and potential mechanisms. Journal of Sleep Research, 32(6), e13928. https://pubmed.ncbi.nlm.nih.gov/37183177/
Jang, G., et al. (2024). Hyperarousal-state of insomnia disorder in wake-resting state quantitative electroencephalography. Clinical Psychopharmacology and Neuroscience, 22(1), 83-92. https://pubmed.ncbi.nlm.nih.gov/38247416/
Ma, Y., Mullington, J. M., Wayne, P. M., & Yeh, G. Y. (2024). Heart rate variability during sleep onset in patients with insomnia with or without comorbid sleep apnea. Sleep Medicine, 122, 92-98. https://pubmed.ncbi.nlm.nih.gov/39137665/
Nano, M. M., Fonseca, P., Vullings, R., & Aarts, R. M. (2017). Measures of cardiovascular autonomic activity in insomnia disorder: A systematic review. PLOS ONE, 12(10), e0186716. https://pubmed.ncbi.nlm.nih.gov/29059210/
Riemann, D., Spiegelhalder, K., Feige, B., Voderholzer, U., Berger, M., Perlis, M., & Nissen, C. (2010). The hyperarousal model of insomnia: a review of the concept and its evidence. Sleep Medicine Reviews, 14(1), 19-31. https://pubmed.ncbi.nlm.nih.gov/19481481/
Rösler, L., van Kesteren, E.-J., Leerssen, J., van der Lande, G., Lakbila-Kamal, O., Foster-Dingley, J. C., … & van Someren, E. J. W. (2024). Hyperarousal dynamics reveal an overnight increase boosted by insomnia. Journal of Psychiatric Research, 179, 317-325. https://pubmed.ncbi.nlm.nih.gov/39341067/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 9 references cited
