Chronic stress does more than make it harder to fall asleep. It can physically alter the way the autonomic nervous system regulates itself, locking the body into sustained sympathetic activation — elevated heart rate, elevated cortisol, suppressed parasympathetic recovery — that fragments sleep architecture and degrades brain health over time.
This article covers the self-reinforcing cortisol-sleep loop, how each bad night amplifies the next, why chronic insomnia is a 24-hour disorder (not a nighttime problem), and what wearable data can reveal about autonomic imbalance during sleep. It does not cover hyperarousal as a standalone condition or broader autonomic mechanisms. For the full overview, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It.
Autonomic imbalance is one of several causes of disrupted sleep and 3am wakeups. The parent article covers the broader picture, including vagal tone, GABA, and neuroinflammation pathways.
Does Chronic Stress Change the Way the Hypothalamic-Pituitary-Adrenal Axis Regulates Itself?
The hypothalamic-pituitary-adrenal axis — the HPA axis — is the primary hormonal stress response cascade. When a stressor registers, the hypothalamus releases corticotropin-releasing hormone (CRH), which triggers the pituitary to release adrenocorticotropic hormone (ACTH), which triggers the adrenal glands to release cortisol. Under normal conditions, elevated cortisol feeds back to the hypothalamus and pituitary through glucocorticoid receptors and suppresses further CRH and ACTH release. The stressor passes, cortisol drops, and the cascade resets.
Under chronic stress, this feedback mechanism degrades. Glucocorticoid receptors in the hippocampus and hypothalamus become resistant — they stop responding to cortisol at normal concentrations (Gutierrez Nunez et al., 2025). The result is that cortisol production continues even after the stressor has resolved. It is a self-sustaining hormonal state where the negative feedback loop has physically stopped functioning at the receptor level.
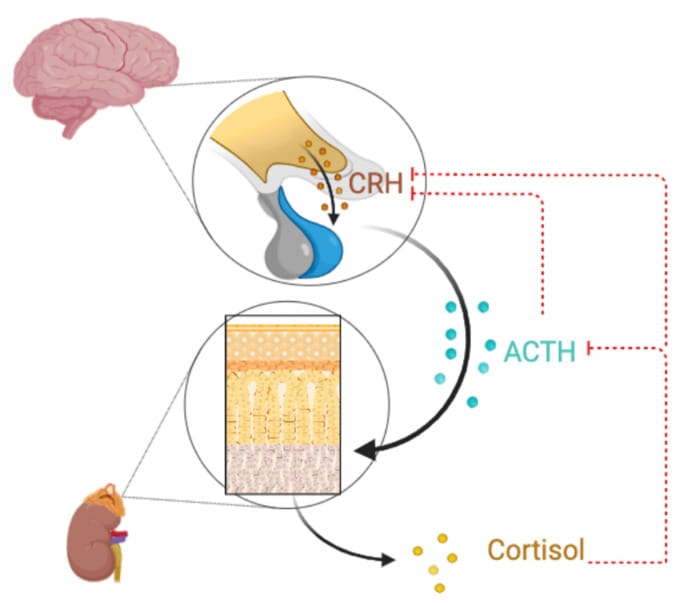
A meta-analysis of 20 case-control studies (449 medication-free people with insomnia compared with 357 healthy controls) found that people with chronic insomnia show a standardized mean difference (SMD) of 0.50 in cortisol elevation compared to controls (Dressle et al., 2022). Blood-based cortisol measurement yielded a larger effect size (SMD = 0.67), suggesting venous sampling captures HPA axis changes more sensitively than salivary or urinary measures. The cortisol elevation retained a normal circadian rhythm but was elevated across the full 24-hour period — consistent with persistent hyperarousal rather than a circadian timing problem.
A 2007 review of chronic insomnia and the stress response found that people with insomnia secrete more cortisol concentrated in the evening and the first half of the night, and those with the worst objective sleep fragmentation secrete the highest amounts (Basta et al., 2007). The HPA axis elevation does not exist in isolation — it accompanies elevated catecholamines (norepinephrine, dopamine metabolites), increased metabolic rate, and heightened core body temperature. These are markers of chronic sympathetic activation that persist well beyond the sleep window.
The implication: once glucocorticoid receptor resistance develops, the cortisol-sleep loop becomes self-sustaining. Elevated cortisol at night fragments sleep. Fragmented sleep further activates the HPA axis. The stressor can be long gone, but the hormonal state it created continues.
Does Each Bad Night Make the Next Night Worse?
People who describe insomnia as a “vicious cycle” are describing something measurable. A 2024 ecological momentary assessment study followed 169 people with insomnia and 38 controls over 9 consecutive days, collecting daily data on sleep quality and hyperarousal (Rosler et al., 2024). The results quantified the night-by-night positive feedback loop: a worse night of sleep produced higher morning hyperarousal, and higher morning hyperarousal predicted worse sleep the following night.
The overnight increase in hyperarousal was stronger in people with insomnia than in healthy controls. This study captured the self-reinforcing loop day by day over consecutive nights, confirming that the cycle escalates rather than stabilizes on its own.
PET imaging research adds a neurological dimension. Brain glucose metabolism — a proxy for neural activity — remains elevated during NREM sleep in people with insomnia (Bonnet & Arand, 2010). During NREM, brain activity should decline as the cortex disengages from waking-level processing. In people with chronic insomnia, this disengagement does not occur. The brain continues at waking-like metabolic rates even during stages that should be restorative, meaning NREM sleep is structurally present but functionally compromised.
Together, these findings explain why one bad night can cascade. Elevated cortisol from poor sleep raises morning arousal. Elevated arousal sustains sympathetic activation through the day. Elevated sympathetic activation at bedtime prevents parasympathetic engagement — the drop in heart rate, blood pressure, and cortisol that normally accompanies sleep onset. The next night starts from a higher baseline of arousal, not a reset. Over 9 days, the Rosler data show this baseline ratcheting upward in people with insomnia, while controls return to their baseline.
Is Insomnia a 24-Hour Disorder or a Nighttime Problem?
The assumption that insomnia is a nighttime problem — something that happens when you get into bed — is not supported by the physiology. Chronic insomnia involves elevated sympathetic activation, elevated cortisol, and elevated inflammatory markers across the full 24-hour cycle.
Basta et al. (2007) measured plasma ACTH, cortisol, catecholamines, IL-6, and TNF-alpha in people with insomnia and controls across 24 hours. People with insomnia showed elevated cortisol concentrated in the evening and first half of the night, elevated 24-hour catecholamines (norepinephrine and dopamine metabolites), and elevated pro-inflammatory cytokines IL-6 and TNF-alpha. These markers were not confined to the sleep window. The immune activation and sympathetic tone persisted through the daytime hours.
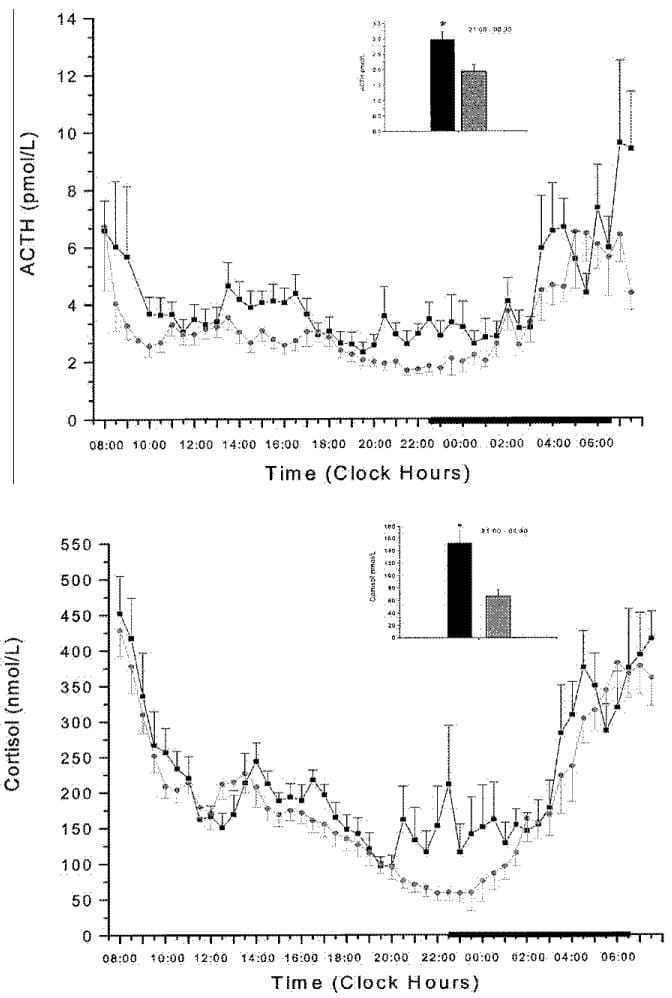
A 2023 study extended this into an objective, measurable daytime marker. McCall et al. (2023) measured the pupillary light reflex — the speed at which the pupil constricts in response to light — in people with and without insomnia during afternoon hours (3-5pm). Pupillary constriction velocity is controlled by the autonomic nervous system: faster constriction reflects parasympathetic dominance, slower constriction reflects sympathetic dominance. In a sample of adults seeking mental health care, pupillary light reflex velocity correlated with insomnia severity at r = -0.929 — meaning the worse the insomnia, the slower the pupil responded to light, reflecting sustained sympathetic dominance hours before bedtime.
PET imaging data from Bonnet and Arand (2010) showed elevated brain glucose metabolism during NREM sleep in people with insomnia, confirming that even during sleep stages where neural activity should decline, the brain remains in a waking-like metabolic state.
This reframes how insomnia works. The sleep disruption at night is a downstream effect of autonomic and hormonal activation that runs around the clock. Addressing the nighttime sleep window alone — sleep hygiene adjustments, bedtime routines — does not address the 24-hour physiological state driving the disruption.
Can a Wearable Device Detect Whether Your Nervous System Is Stuck?
Wix-Ramos et al. (2024) used wearable biosensors alongside polysomnography (PSG) in 32 people with chronic insomnia and 19 healthy controls. The wearable sensors measured heart rate, electrodermal activity (skin conductance, which reflects sympathetic nervous system activation), and peripheral skin temperature throughout the night.
The insomnia group showed elevated heart rate and heightened electrodermal activity across all sleep stages — not during wakefulness or light sleep alone, but during deep sleep and REM as well. Peripheral skin temperature did not decline during sleep in the insomnia group. This matters because the parasympathetic nervous system normally drives vasodilation (blood vessel widening) in the hands and feet at sleep onset, which releases heat and drops core body temperature. The absence of this temperature decline indicates the parasympathetic vasodilation response was not engaging — the thermoregulatory cooling that accompanies normal sleep onset was not occurring.
For wearable users, this translates to measurable patterns. Overnight heart rate variability (HRV) reflects the balance between sympathetic and parasympathetic activity. A pattern of low overnight HRV, elevated resting heart rate during sleep, and absent or blunted heart rate dip during the night may reflect the same autonomic imbalance the Wix-Ramos study documented with research-grade sensors.
The McCall et al. (2023) pupillary light reflex data adds another dimension: autonomic imbalance is detectable during waking hours, not only during sleep. The correlation of r = -0.929 between pupillary constriction velocity and insomnia severity suggests that daytime autonomic testing could provide a complementary measure to overnight wearable data.
Consumer wearables do not measure electrodermal activity or peripheral skin temperature with the precision of research-grade sensors. But the heart rate and HRV data they do capture can reflect the underlying autonomic pattern — and tracking these metrics over weeks provides a longitudinal picture of whether autonomic balance is changing in response to stress reduction, breathing practices, or other changes.
Sustained sympathetic activation from cortisol-driven HPA axis imbalance rarely exists in isolation. Cortisol elevation might overlap with metabolic disruptions, hormonal changes, or inflammatory processes that compound sleep fragmentation. When multiple causes are active at the same time, addressing only one may not resolve the pattern.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
How Long Does It Take for the Cortisol-Sleep Loop to Resolve?
The cortisol-sleep loop does not reverse quickly. Glucocorticoid receptor resistance developed over months of chronic stress does not reverse in days. Laborde et al. (2019) found that 30 days of slow-paced breathing (at 6 breaths per minute) produced improvements in cardiac vagal activity and subjective sleep quality compared to a control group. This provides a timeframe reference — 30 days of consistent practice for initial measurable changes in vagal tone — but the full resolution of cortisol-sleep feedback likely requires weeks to months of sustained autonomic nervous system regulation depending on how long the imbalance has been active and how many contributing factors are involved.
Is Stress Insomnia Different from Regular Insomnia?
The distinction between “stress insomnia” and “chronic insomnia” is less clean than it appears. Dressle and Riemann (2023) reviewed evidence across physiological, cortical, and cognitive-emotional hyperarousal domains and found that neuroendocrine markers — HPA axis activity and cortisol — provided the strongest replicable physiological marker of insomnia. Cognitive-emotional hyperarousal (sleep-related worry, rumination, intrusive thoughts) showed the broadest evidence base as both a trigger and a maintaining factor.
The practical distinction that matters: if insomnia that started during a stressful period persists after that period ends, the autonomic nervous system may have transitioned from a reactive state (responding to an active stressor) to a self-sustaining one (glucocorticoid receptor resistance, conditioned arousal, elevated baseline sympathetic tone). The stressor was the trigger, but the autonomic and hormonal changes it produced are now maintaining the insomnia independently.
Can Your Nervous System Get Permanently Stuck in Fight or Flight?
Glucocorticoid receptor sensitivity can be restored. Hippocampal feedback on the HPA axis can recover. Parasympathetic tone can improve. But these changes require consistent, sustained effort — not a single supplement or a week of relaxation. The Gutierrez Nunez et al. (2025) review documents the molecular mechanisms maintaining glucocorticoid receptor resistance (receptor downregulation, impaired nuclear translocation of glucocorticoid receptors, epigenetic changes to GR gene expression) and notes that these are responsive to interventions that reduce chronic stress load and inflammation over time.
The longer the imbalance has been active, the more entrenched the molecular changes become — but “entrenched” does not mean “irreversible.”
Does Cortisol Testing Help Identify This?
Cortisol follows a circadian rhythm: it peaks 30-45 minutes after waking (the cortisol awakening response), declines through the afternoon, and reaches its lowest point around midnight. A single measurement — morning blood draw, random salivary sample — captures one point on a curve that varies across 24 hours.
The Dressle et al. (2022) meta-analysis found that people with insomnia show globally elevated cortisol that retains a normal circadian shape but is elevated across the full cycle. Blood-based cortisol measurement (SMD = 0.67) captured this more sensitively than salivary or urinary measures (overall SMD = 0.50). A multi-point salivary cortisol collection — typically at waking, 30 minutes post-waking, afternoon, and evening — provides the curve shape needed to identify whether the diurnal rhythm is flattened, evening cortisol is elevated, or the cortisol awakening response is blunted. These patterns can indicate HPA axis imbalance consistent with the chronic stress-insomnia loop described above.
Related Reading:
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — how autonomic imbalance fragments sleep architecture and what drives it, including vagal tone, GABA, and neuroinflammation pathways
- What Is Hyperarousal Insomnia? Why You’re Wired but Tired Every Night — how hyperarousal differs from general insomnia, cortical and cognitive markers, and why “tired but wired” reflects measurable brain activity
- Why Won’t Your Brain Shut Off at Night? The Autonomic Nervous System Connection — why the brain stays active at bedtime, the role of sympathetic-parasympathetic balance in sleep onset, and what disrupts it
- Does Benadryl Destroy Your Sleep? How Anticholinergic Drugs Suppress REM Sleep — how anticholinergic medications suppress REM sleep by blocking acetylcholine, and why sedation is not the same as restorative sleep
- What Your Overnight HRV Is Telling You About Your Sleep: The Vagal Tone Connection — what heart rate variability during sleep reflects about parasympathetic recovery and how to interpret overnight HRV trends
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — the gut-brain axis, vagal afferent communication, and how gut inflammation can drive autonomic sleep disruption
- Why Is Your REM Sleep Fragmented? The Brainstem Switch That Controls Dream Sleep — how the brainstem flip-flop mechanism regulates REM-NREM transitions and what disrupts it
- Is It Just Stress? 4 Reasons You’re Waking at 3 A.M. — four common causes of 3am waking beyond stress, including blood sugar, cortisol, and hormonal changes
- Alcohol Sedates You, Then Wakes You at 3AM — how alcohol disrupts sleep architecture in the second half of the night through rebound sympathetic activation and REM suppression
- Why Won’t Your Brain Shut Off at Night? The Autonomic Connection — how GABA receptor impairment drives the sympathetic overactivation that prevents sleep
- Why Is Your Rapid Eye Movement Sleep Fragmented? The Brainstem Switch That Controls It — the brainstem circuit that controls dream sleep and which medications suppress it
- Does Benadryl Degrade Your Sleep? How Anticholinergic Drugs Suppress Rapid Eye Movement Sleep — how anticholinergic medications block the muscarinic receptors REM requires
- What Your Overnight Heart Rate Variability Is Telling You About Your Sleep: The Vagal Tone Connection — using HRV as a window into parasympathetic recovery during sleep
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut bacteria, GABA production, and the vagal pathway to sleep regulation
- Do Antidepressants Suppress Rapid Eye Movement Sleep? — SSRIs, SNRIs, and tricyclics affect sleep architecture
- Can Vagus Nerve Stimulation Devices Improve Insomnia? — clinical trial evidence for taVNS devices
- Does Your Sleep Tracker Accuracy Matter? — what Oura, WHOOP, and Apple Watch get right and wrong
- Is Your Insomnia a Nervous System Problem? — how to tell if autonomic dysregulation is driving sleep disruption
References
Basta, M., Chrousos, G. P., Vela-Bueno, A., & Vgontzas, A. N. (2007). Chronic insomnia and the stress response. Sleep Medicine Clinics, 2(2), 279-291. https://pubmed.ncbi.nlm.nih.gov/18071579/
Bonnet, M. H., & Arand, D. L. (2010). Hyperarousal and insomnia: State of the science. Sleep Medicine Reviews, 14(1), 9-15. https://pubmed.ncbi.nlm.nih.gov/19640748/
Dressle, R. J., Feige, B., Spiegelhalder, K., Schmucker, C., Benz, F., Mey, N. C., & Riemann, D. (2022). HPA axis activity in patients with chronic insomnia: A systematic review and meta-analysis of case-control studies. Sleep Medicine Reviews, 62, 101588. https://pubmed.ncbi.nlm.nih.gov/35091194/
Dressle, R. J., & Riemann, D. (2023). Hyperarousal in insomnia disorder: Current evidence and potential mechanisms. Journal of Sleep Research, 32(6), e13928. https://pubmed.ncbi.nlm.nih.gov/37183177/
Gutierrez Nunez, S., Peixoto Rabelo, S., Subotic, N., Caruso, J. W., & Knezevic, N. N. (2025). Chronic stress and autoimmunity: The role of HPA axis and cortisol dysregulation. International Journal of Molecular Sciences, 26(20), 9994. https://pubmed.ncbi.nlm.nih.gov/41155288/
Laborde, S., Hosang, T., Mosley, E., & Dosseville, F. (2019). Influence of a 30-day slow-paced breathing intervention compared to social media use on subjective sleep quality and cardiac vagal activity. Journal of Clinical Medicine, 8(2), 193. https://pubmed.ncbi.nlm.nih.gov/30736268/
McCall, W. V., Boggs, N., Letton, A., Memoracion-Perdue, A., & Nuckols, C. (2023). Daytime autonomic nervous system functions differ among adults with and without insomnia symptoms. Journal of Clinical Sleep Medicine, 19(10), 1749-1756. https://pubmed.ncbi.nlm.nih.gov/37421322/
Rosler, L., Gee, B., Espie, C. A., & Kyle, S. D. (2024). Hyperarousal dynamics reveal an overnight increase boosted by insomnia. Journal of Psychiatric Research, 179, 288-295. https://pubmed.ncbi.nlm.nih.gov/39341067/
Wix-Ramos, R., Gálvez-Goicuría, J., Verona-Almeida, M., Ayala, J. L., López-Viñas, L., Rocío-Martín, E., Luque-Cárdenas, C., Quintas, S., Gago-Veiga, A., & Pagán, J. (2024). Monitoring differences in the function of the autonomic nervous system in patients with chronic insomnia using a wearable device. Sleep Medicine, 115, 122-130. https://pubmed.ncbi.nlm.nih.gov/38359591/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 9 references cited
