If you have been addressing gut health and sleep separately, the molecular evidence suggests they share more machinery than is commonly recognized. The bacteria in your colon produce metabolites that affect clock gene transcription in your intestinal lining, your liver, your kidneys — and through the vagus nerve, your central clock in the hypothalamus. When those bacteria are depleted, clock gene expression dampens. When the clock is disrupted, those bacteria decline.
This article covers three routes by which gut microbes influence circadian timing, the probiotic trial data on sleep quality, and the bidirectional feedback that connects circadian disruption to microbial depletion. For the broader context of how circadian disruption fragments sleep after age 40, see the circadian sleep disruption pillar.
?-
How Do Gut Bacteria Set the Timing of Your Peripheral Clocks?
The molecular clock in each cell runs on a transcription-translation feedback loop: CLOCK and BMAL1 proteins bind together, activate PER and CRY genes, and the resulting PER and CRY proteins feed back to suppress their own production — cycling approximately every 24 hours. This loop runs in every nucleated cell, but what entrains it differs by tissue. In the gut epithelium, bacterial metabolites are a primary entrainment input.
Fawad et al. (2022) demonstrated this in intestinal enteroids and organoids (murine and human). Butyrate and other SCFAs inhibited HDAC3, which altered the acetylation landscape near clock gene promoters and produced concentration-dependent phase shifts in PER2 expression — with murine enteroids showing median phase delays of 6.2 hours and human organoids showing phase advances of approximately 5 hours. The effect was dose-dependent and specific to HDAC3: conditional genetic deletion of HDAC3 abolished the circadian phase shift that SCFAs produced, confirming the enzyme as the mechanistic link (Fawad et al., 2022).
A 2025 systematic review by dos Santos and Vasylyshyn extended this finding across tissues and clock genes. Their review of preclinical and clinical studies found that SCFAs modulate expression of PER1, PER2, BMAL1, CRY1, and CRY2 in intestinal, hepatic, and renal tissue — not one clock gene in one organ, but the full set of core clock components across multiple peripheral clocks (dos Santos & Vasylyshyn, 2025).
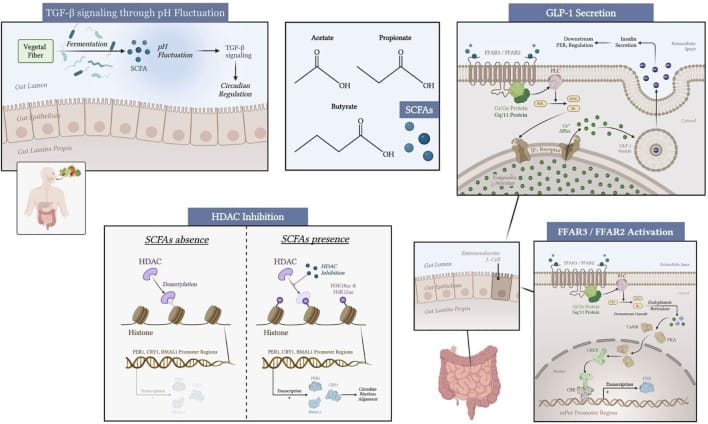
The practical implication: the bacteria that produce butyrate — primarily Firmicutes species like Faecalibacterium prausnitzii, Roseburia intestinalis, and Eubacterium rectale — are not “good gut bacteria” in a vague sense. They are upstream of the molecular clock in your peripheral organs. When these populations decline — from antibiotic use, low-fiber diets, or circadian disruption itself — the SCFA input that entrains peripheral clocks weakens.
?-
How Does Your Gut Communicate With Your Central Clock Through the Vagus Nerve?
The current literature describes at least three pathways through which gut microbes may influence circadian timing in the brain (Tofani, Clarke, & Cryan, 2025):
Route 1 — SCFAs and HDAC inhibition. This is the peripheral clock pathway described above. Butyrate, propionate, and acetate entrain intestinal, hepatic, and renal clocks. SCFAs also enter the bloodstream and can cross the blood-brain barrier at low concentrations, providing a secondary input to central clock regions.
Route 2 — Tryptophan to serotonin to melatonin. Gut bacteria metabolize dietary tryptophan, influencing how much tryptophan is available for serotonin synthesis in the gut and the brain. Since serotonin is the biochemical precursor to melatonin — the hormone that entrains the central clock and promotes sleep onset — microbial tryptophan metabolism influences melatonin production in the pineal gland. For more on how melatonin interacts with wake-promoting orexin neurons, see How Does Melatonin Suppress Orexin to Enable Sleep Onset?.
Route 3 — Vagal afferents. The vagus nerve carries sensory information from the gut to the brainstem, which projects to the hypothalamus. Microbial metabolites and cytokines activate vagal afferent fibers, providing a neural (not hormonal, not metabolic) communication channel between the gut microbiome and the suprachiasmatic nucleus (SCN) — the central circadian pacemaker.
In a separate study, Tofani et al. (2025) demonstrated that when the gut microbiota was depleted in mice, brain transcriptome and metabolome in stress-responding pathways were disrupted across the hippocampus and amygdala, and glucocorticoid rhythms — the HPA axis output that follows circadian timing — lost their normal daily patterning. Through fecal microbiota transplantation, Lactobacillus reuteri emerged as a candidate strain that may influence glucocorticoid secretion patterns — suggesting that individual bacterial species may influence circadian hormone rhythmicity through the gut-brain axis (Tofani et al., 2025). This was demonstrated in microbiota-depleted mice (germ-free and antibiotic-treated), so the translation to humans with an intact microbiome requires further study.
?-
Can Probiotics Improve Sleep Quality?
Santi et al. (2023) conducted a meta-analysis of randomized controlled trials examining the relationship between gut microbiome composition and sleep quality. The probiotic arm of the analysis found a measurable improvement in PSQI scores — the standard validated instrument for subjective sleep quality — in the probiotic groups compared to placebo. The pooled effect was a standardized mean difference of -0.40 (95% CI: -0.79 to -0.02), favoring the probiotic group (Santi et al., 2023).
Ito et al. (2024) confirmed this direction in a larger meta-analysis of 15 RCTs, confirming the PSQI improvement and strengthening the evidence that the effect is not an artifact of any single trial design (Ito et al., 2024).
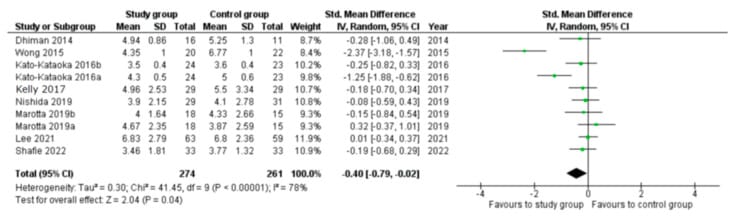
A few caveats matter here. The trials used different probiotic formulations — different species, different doses, different durations. The effect size was modest, consistent with what you would expect from addressing one input among many. And PSQI measures subjective sleep quality, not polysomnographic sleep architecture — so the data do not tell us whether probiotics change deep sleep duration, REM proportion, or sleep continuity.
What the data do support: targeting gut microbial composition can influence self-reported sleep quality in a direction consistent with the SCFA-clock gene mechanism described above. The question is not whether probiotics “fix” sleep — it is whether restoring microbial populations that produce clock-entraining metabolites contributes to better circadian function as one input among several.
?-
Does Poor Sleep Damage Your Gut Microbiome?
A 2025 review in Chronobiology International examined the gut microbiome consequences of rotating night schedules — a human model of chronic circadian disruption. Night-schedule workers showed reduced Firmicutes populations and altered Firmicutes-to-Bacteroidetes ratios compared to day workers. The depleted species included the butyrate producers (Faecalibacterium, Roseburia) that are upstream of the HDAC3-PER2 clock-entrainment pathway (Touitou, Perlemuter, & Touitou, 2025).
Disrupted circadian rhythms alter the timing and composition of bile acid secretion, mucus production, and intestinal motility — all of which shape the microbial environment. When these host-side rhythms lose their 24-hour patterning, the microbiome composition changes because the ecological niche changes. The depleted beneficial taxa then produce fewer SCFAs, less tryptophan is directed toward serotonin and melatonin synthesis, and vagal afferent input weakens — degrading all three routes of gut-to-brain circadian communication.
This bidirectional feedback has consequences for anyone whose sleep has been fragmented for months or years. The initial cause may have been stress, hormonal changes, medication effects (beta blockers, for instance, suppress melatonin and disrupt circadian timing), or inflammation affecting clock gene expression. Once the gut microbiome is depleted as a secondary consequence of circadian disruption, it can become an independent contributing factor — sustaining the sleep problem even after the original cause is addressed. Gut-derived metabolic changes can also affect the orexin wake-drive, which responds to metabolic and autonomic inputs — see Can Chronic Stress Hijack Your Orexin Wake Signal? for how stress and autonomic inputs compound.
?-
Does the Gut Microbiome Have Its Own Circadian Clock?
The rhythmicity of the gut microbiome is driven by the host. When you eat at consistent times, your liver releases bile acids on a predictable schedule, your gut motility follows a daily pattern, and the pH and nutrient availability in the colon oscillates. These host-driven cycles create a 24-hour oscillation in which bacterial species are dominant at which times — and which metabolites they produce, and in what quantities (Tofani, Clarke, & Cryan, 2025).
SCFA production — particularly butyrate — follows this daily oscillation, peaking in the hours after meals and declining during fasting periods. The clock-entraining input that SCFAs provide to peripheral organs is itself rhythmic, reinforcing circadian timing in a feedforward manner. When meal timing is erratic or when circadian disruption alters bile acid and motility rhythms, the daily oscillation in microbial metabolite production flattens — and the entraining input weakens.
?-
Which Probiotic Strains Have Been Tested for Sleep?
From the probiotic RCTs included in Santi et al. (2023) and Ito et al. (2024), the formulations that improved PSQI scores were multi-strain products containing various Lactobacillus (L. rhamnosus, L. acidophilus, L. casei) and Bifidobacterium (B. longum, B. bifidum, B. lactis) species. No single-strain trial has demonstrated a larger effect than a multi-strain formulation, and no trial has compared strains against each other for sleep endpoints.
The Lactobacillus reuteri finding from Tofani et al. (2025) — where this species was identified as a candidate that may influence glucocorticoid secretion in microbiota-depleted mice — is a mechanistic proof of concept that an individual strain can influence circadian hormone rhythms. But it was demonstrated in microbiota-depleted mice (germ-free and antibiotic-treated), not humans with an intact microbiome, so the translation to probiotic recommendations requires further study (Tofani et al., 2025).
What the current evidence supports: rather than searching for a single “best sleep probiotic,” the priority is maintaining the ecological conditions that support butyrate-producing populations — adequate dietary fiber, consistent meal timing, and minimizing unnecessary antibiotic exposure.
?-
Does Butyrate Supplementation Improve Sleep?
The mechanistic case is the strongest of any single microbial metabolite. Fawad et al. (2022) showed a dose-dependent, HDAC3-mediated effect on PER2 circadian phase in intestinal enteroids and organoids — a well-controlled model that reflects intestinal epithelial function (Fawad et al., 2022). dos Santos and Vasylyshyn (2025) found SCFA modulation of the full panel of core clock genes across multiple tissues (dos Santos & Vasylyshyn, 2025).
But butyrate supplementation faces a pharmacokinetic challenge: oral butyrate is largely absorbed in the upper gastrointestinal tract and metabolized before reaching the colon, where the SCFA-clock gene interaction occurs. Colonic butyrate levels depend primarily on bacterial fermentation of dietary fiber in situ, not on oral supplementation. This is why the fiber-to-microbiome-to-butyrate pathway may matter more than taking butyrate as a supplement — and why the composition of your gut bacteria, rather than a supplement, is the rate-limiting factor.
?-
Frequently Asked Questions
Is Leaky Gut Related to Sleep Problems?
How Long Does It Take Probiotics to Affect Sleep?
Does Meal Timing Affect Your Gut’s Influence on Sleep?
?-
Gut-circadian interactions may compound with hormonal, metabolic, autonomic, or inflammatory causes of sleep disruption. In adults over 40, multiple causes often overlap. Identifying which causes might be contributing is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
?-
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Does Your Circadian Clock Control When Insulin Works? Prediabetes, the Dawn Phenomenon, and Sleep — how circadian insulin timing, glucose rhythm, and dawn physiology affect sleep continuity
- Can Chronic Inflammation Disrupt Your Circadian Clock Genes? — how cytokines and NF-kB signaling suppress central and peripheral clock genes
- Can Your Body Temperature Keep You From Falling Asleep? — how thermoregulation, vasodilation, and temperature rhythm affect sleep onset and depth
- Why Are You Still Exhausted After Months on CPAP? — how intermittent hypoxia can suppress clock genes even after breathing normalizes
- Do Beta Blockers Suppress Melatonin? What Blood Pressure Medication Does to Your Body Clock — how beta blockers, melatonin suppression, and medication timing affect circadian sleep
References
dos Santos, A. A., & Vasylyshyn, A. (2025). The modulatory role of short-chain fatty acids on peripheral circadian gene expression: A systematic review. Frontiers in Physiology, 16, 1595057. https://pubmed.ncbi.nlm.nih.gov/40727446/
Fawad, J. A., Luzader, D. H., Hanson, G. F., Moutinho, T. J., Jr., McKinney, C. A., Mitchell, P. G., Brown-Steinke, K., Kumar, A., Park, M., Lee, S., Bolick, D. T., Medlock, G. L., Zhao, J. Y., Rosselot, A. E., Chou, C. J., Eshleman, E. M., Alenghat, T., Hong, C. I., Papin, J. A., & Moore, S. R. (2022). Histone deacetylase inhibition by gut microbe-generated short-chain fatty acids entrains intestinal epithelial circadian rhythms. Gastroenterology, 163(5), 1377-1390.e11. https://pubmed.ncbi.nlm.nih.gov/35934064/
Ito, H., Tomura, Y., Kitagawa, Y., Nakashima, T., Kobanawa, S., Uki, K., Oshida, J., Kodama, T., Fukui, S., & Kobayashi, D. (2024). Effects of probiotics on sleep parameters: A systematic review and meta-analysis. Clinical Nutrition ESPEN, 63, 623-630. https://pubmed.ncbi.nlm.nih.gov/39094854/
Santi, D., Debbi, V., Costantino, F., Spaggiari, G., Simoni, M., Greco, C., & Casarini, L. (2023). Microbiota composition and probiotics supplementations on sleep quality — a systematic review and meta-analysis. Clocks & Sleep, 5(4), 770-792. https://pubmed.ncbi.nlm.nih.gov/38131749/
Tofani, G. S., Clarke, G., & Cryan, J. F. (2025). I “Gut” Rhythm: the microbiota as a modulator of the stress response and circadian rhythms. The FEBS Journal, 292(6), 1454-1479. https://pubmed.ncbi.nlm.nih.gov/39841560/
Tofani, G. S., Leigh, S. J., Bastiaanssen, T. F. S., Gheorghe, C. E., Wilmes, L., Sen, P., Clarke, G., & Cryan, J. F. (2025). Gut microbiota regulates stress responsivity via the circadian system. Cell Metabolism, 37(1), 138-153.e5. https://pubmed.ncbi.nlm.nih.gov/39504963/
Touitou, Y., Perlemuter, G., & Touitou, C. (2025). Shift work, gut dysbiosis, and circadian misalignment: The combined impact of nighttime light exposure, nutrients, and microbiota rhythmicity. Chronobiology International, 42(10), 1275-1290. https://pubmed.ncbi.nlm.nih.gov/40772701/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 7 references cited
