Melatonin is the molecule widely associated with sleep. Orexin (also called hypocretin) is the molecule that keeps the brain awake. What fewer people realize is that melatonin actively suppresses orexin — making them two halves of a single regulatory balance.
This article covers how melatonin inhibits orexin neurons at the receptor level, why this matters for people who take melatonin supplements without results, and how aging degrades both sides of the balance. For orexin basics, see What Is Orexin and Why Does It Keep You Awake?.
The melatonin-orexin opposition is one component of broader circadian regulation. Both molecules answer to the suprachiasmatic nucleus (the brain’s master clock), and both change with age. For the full circadian picture, see the circadian pillar.
How Does Melatonin Inhibit Orexin Neurons?
Sharma, Sahota, and Thakkar (2018) tested melatonin’s direct effect on orexin neurons using microinfusion — delivering melatonin directly into the perifornical lateral hypothalamus (the brain region where orexin neurons are concentrated) in mice. They measured orexin neuron activity using c-Fos, a protein marker that indicates recent neuron firing.
The results were unambiguous. Melatonin microinfusion reduced c-Fos expression in orexin neurons by 66%, meaning roughly two-thirds of the orexin neurons that would normally be active were suppressed. The researchers confirmed that this effect required MT1 receptors: when they blocked MT1 with a selective antagonist, melatonin’s suppressive effect on orexin neurons disappeared. When they blocked MT2 receptors, the suppression continued unchanged.
This receptor specificity matters. Melatonin has two receptor subtypes — MT1 and MT2 — and they do different things. Gobbi and Comai (2019) mapped the functional division: MT1 receptors are present on orexin neurons and govern REM sleep regulation alongside orexin inhibition. MT2 receptors promote NREM sleep through a separate pathway involving GABAergic neurons in the reticular thalamus — and MT2 receptors are absent on orexin neurons.
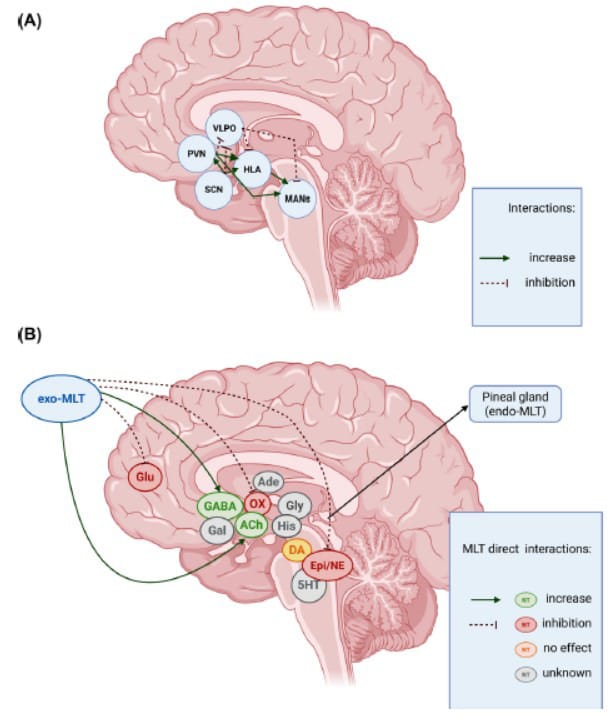
In plain language: when melatonin rises in the evening, it binds MT1 receptors on orexin neurons and reduces their firing rate. This removes the wake-promoting force that orexin exerts on downstream arousal centers — the locus coeruleus, dorsal raphe, and tuberomammillary nucleus. Melatonin does not force sleep by sedating the brain. It opens the window for sleep by suppressing the molecule that maintains wakefulness.
This distinction has a practical implication. If the reason someone cannot fall asleep is that their melatonin timing is delayed (from late light exposure, for example), then correctly timed melatonin addresses the root problem: orexin neurons remain active past the intended bedtime because the melatonin rise is arriving late. But if the reason orexin neurons are overactive has nothing to do with melatonin timing — if stress hormones or circadian disruption are driving orexin independently — then supplemental melatonin is working against a force it was not designed to overcome.
Caveat: the MT1-orexin inhibition data comes from mouse studies (Sharma et al., 2018). No equivalent human study has confirmed the same receptor-specific mechanism in human orexin neurons, though MT1 receptor expression in the human hypothalamus is established. The mouse findings are consistent with human sleep pharmacology data, but direct receptor-level translation has not been completed.
Why Does Melatonin Supplementation Underperform When Orexin Is Chronically Elevated?
Melatonin is one of the widely purchased sleep supplements in the world. For many users, it does not work. The typical response is to increase the dose — from 1 mg to 3 mg, then 5 mg, then 10 mg. This rarely solves the problem, and the reason traces back to the melatonin-orexin balance.
Palagini et al. (2023) reviewed how the orexin pathway integrates with the hypothalamic-pituitary-adrenal (HPA) axis — the body’s primary stress response pathway. Orexin neurons in the lateral hypothalamus receive direct input from corticotropin-releasing hormone (CRH) neurons. When someone experiences chronic stress, elevated CRH activates orexin neurons independently of circadian timing. The orexin drive stays engaged not because the circadian clock is promoting wakefulness, but because the stress axis is driving it.
This creates a situation where the push-pull is structurally unbalanced. Melatonin, even at correct timing, provides a fixed magnitude of orexin suppression via MT1 receptors. But when orexin neurons are receiving additional excitatory input from CRH, from norepinephrine, and from cortisol-mediated feedback loops, the melatonin-mediated inhibition cannot match the drive. The wake side of the balance overpowers the sleep side.
Zisapel (2018) reinforced this from the pharmacology side: melatonin supplementation produces measurable improvements in sleep onset latency and sleep quality when the underlying problem is melatonin — delayed timing, low endogenous production, or weak circadian amplitude. When the underlying problem is orexin overactivity driven by stress, inflammation, or circadian misalignment, melatonin’s effects are weaker and often below the threshold of perceived benefit.
The dose confusion makes this worse. Physiological melatonin production at night peaks around 30–60 picograms per milliliter in plasma. A 3 mg oral dose raises plasma melatonin to 100 times the physiological peak. A 10 mg dose reaches 300 times. Despite these levels, orexin suppression does not scale linearly with melatonin concentration. MT1 receptors on orexin neurons saturate — once enough receptors are bound, additional melatonin does not produce additional inhibition. Increasing the dose from 3 mg to 10 mg may extend the duration of elevated plasma melatonin, but it does not proportionally increase the suppressive force on orexin neurons.
This creates a practical question for anyone who finds melatonin ineffective. The melatonin-orexin balance can be unbalanced from either side:
- Melatonin-side problem: endogenous melatonin is delayed, suppressed by evening light, or reduced by pineal calcification. In this case, correctly timed low-dose melatonin (0.5–1 mg, 30–60 minutes before target sleep onset) can restore the balance.
- Orexin-side problem: orexin neurons are overactive due to chronic stress, HPA axis activation, pain, or other non-circadian inputs. In this case, melatonin addresses the wrong side of the equation. The orexin drive needs to be reduced independently.
A useful indicator: if falling asleep takes more than an hour despite consistent melatonin use at correct timing, and ongoing stress is present, the issue is more likely orexin-driven than melatonin-driven.
How Does Aging Degrade Both Sides of the Melatonin-Orexin Balance?
The push-pull between melatonin and orexin degrades from both directions with age. Kron et al. (2024), in a review published in Annual Review of Pharmacology and Toxicology, documented both trajectories.
On the melatonin side: pineal gland calcification reduces melatonin synthesis capacity across the lifespan. By age 60, nocturnal melatonin levels in many individuals are a fraction of what they were at age 20. The circadian melatonin rise that suppresses orexin neurons each evening becomes weaker — and in some older adults, barely detectable.
On the orexin side: while total orexin neuron count declines with age (postmortem studies show approximately 23% fewer orexin neurons in older vs. younger adults), the surviving neurons become hyperexcitable. Research in aged mice has identified impaired KCNQ2/3 potassium channels as the mechanism — these channels normally restrain orexin neuron firing, and their age-related decline lowers the threshold for orexin activation. For the full mechanistic evidence on age-related orexin hyperexcitability, see How Does Orexin Change With Age?.
The double degradation produces a compounding effect. Less melatonin means less MT1-mediated orexin suppression each evening. More excitable orexin neurons means a higher baseline of wake drive that needs to be suppressed. Each side makes the other worse: a smaller suppressive force acting on a larger target.
Comai and Gobbi (2024) added nuance to the melatonin side. Melatonin’s efficacy in promoting sleep depends on three factors: timing (administration relative to endogenous circadian phase), receptor subtype activation (MT1 for orexin suppression and REM regulation, MT2 for NREM promotion), and dose. In older adults, all three become less reliable — circadian phase advances and becomes less stable, receptor sensitivity may change, and the dose-response relationship tightens because endogenous production is already low.
Kron et al. (2024) reviewed dual orexin receptor antagonists (DORAs) — medications like suvorexant, lemborexant, and daridorexant that block orexin receptors (OX1R and OX2R). Muehlan et al. (2023) provide additional background on DORA pharmacology. These drugs reduce wake drive by preventing orexin from activating its downstream arousal targets. In older adults, DORAs normalized sleep architecture — including improvements in both sleep onset and wake-after-sleep-onset — without the sedation, cognitive impairment, fall risk, or dependency associated with benzodiazepines and Z-drugs.
The pharmacological logic here maps directly to the push-pull framework:
- Melatonin or ramelteon (an MT1/MT2 agonist) strengthens the push side — increasing the suppressive input that dampens orexin neuron firing.
- A DORA addresses the pull side — blocking the orexin receptors so that even if orexin neurons fire, the wake-promoting output is attenuated at its targets.
These are complementary mechanisms: one reduces the source (orexin neuron activity), the other blocks the output (orexin receptor activation). Published data on the combination comes from delirium-prevention procedures in hospitalized populations, not from primary insomnia randomized controlled trials. Any medication combination requires discussion with your doctor.
For individuals relying on melatonin alone after midlife, the implication is that supplemental melatonin addresses one side of a two-sided degradation. When both sides have deteriorated, a single-sided approach produces diminishing returns.
The melatonin-orexin balance is one component of circadian sleep regulation. Hormonal changes, cortisol timing, metabolic factors, and inflammatory causes might also be contributing to sleep fragmentation. Identifying which causes might be involved gives a more actionable picture.
Is Melatonin a Sleeping Pill?
Melatonin is a circadian timing molecule, not a sedative. It does not induce unconsciousness the way benzodiazepines or Z-drugs do. Its role is to communicate that night has arrived — and part of that communication involves suppressing orexin neurons via MT1 receptors, reducing the wake-promoting drive. Overdosing does not increase this suppression: MT1 receptors on orexin neurons saturate at concentrations well below what a 5–10 mg dose produces. Higher doses extend the duration of elevated plasma melatonin but do not proportionally increase receptor-mediated effects.
Can You Take Melatonin and a Dual Orexin Receptor Antagonist Together?
They target opposite sides of the same balance — complementary pharmacology. Melatonin (or the MT1/MT2 agonist ramelteon) reduces orexin neuron firing at the source. DORAs block the effect of orexin at its receptors on downstream arousal centers. Published data on the combination comes from delirium-prevention procedures in hospitalized older adults, not from primary insomnia randomized controlled trials. Any medication combination should be discussed with your doctor.
Does Anxiety Block Melatonin From Working?
Anxiety does not block melatonin at the receptor level. What anxiety does is activate the HPA axis, which raises cortisol and CRH. CRH directly activates orexin neurons in the lateral hypothalamus. If orexin is elevated by anxiety-driven stress activation, supplemental melatonin may provide insufficient MT1-mediated suppression to overcome the elevated orexin drive. The issue is not that anxiety interferes with melatonin binding — it amplifies the force that melatonin is working against.
Related Reading: circadian pillar + sibling orexin articles: What Is Orexin and Why Does It Keep You Awake? · How Does Orexin Change With Age? (additional sibling articles publishing soon)
References
1. Sharma, R., Sahota, P., & Thakkar, M. M. (2018). Melatonin promotes sleep in mice by inhibiting orexin neurons in the perifornical lateral hypothalamus. Journal of Pineal Research, 65(2), e12498. PMID: 29654707
2. Zisapel, N. (2018). New perspectives on the role of melatonin in human sleep, circadian rhythms and their regulation. British Journal of Pharmacology, 175(16), 3190–3199. PMID: 29318587
3. Gobbi, G., & Comai, S. (2019). Sleep well. Untangling the role of melatonin MT1 and MT2 receptors in sleep. Journal of Pineal Research, 66(3), e12544. PMID: 30586215
4. Muehlan, C., Roch, C., Vaillant, C., & Dingemanse, J. (2023). The orexin story and orexin receptor antagonists for the treatment of insomnia. Journal of Sleep Research, 32(6), e13902. PMID: 37086045
5. Palagini, L., Manni, R., Aguglia, E., Amore, M., Brugnoli, R., Bioulac, S., … & Bhatt, N. B. (2023). Current models of insomnia disorder: a theoretical review on the potential role of the orexinergic pathway with implications for insomnia treatment. Journal of Sleep Research, 32(4), e13825. PMID: 36786121
6. Biso, L., Bragazzi, N. L., Bonaso, M., Bersanetti, E., Garbarino, S., & Scarselli, M. (2025). Exogenous melatonin as a sleep-promoting agent beyond its chronobiotic properties: a scoping review of its effects on key sleep-wake neurotransmitters. Current Neuropharmacology, 24(3), 364–387. PMC: PMC13084770
7. Comai, S., & Gobbi, G. (2024). Melatonin, melatonin receptors and sleep: moving beyond traditional views. Journal of Pineal Research, 76(7), e13011. PMID: 39400423
8. Kron, J. O.-Z. J., Keenan, R. J., Hoyer, D., & Jacobson, L. H. (2024). Orexin receptor antagonism: normalizing sleep architecture in old age and disease. Annual Review of Pharmacology and Toxicology, 64, 359–386. PMID: 37708433
9. Yang, S., Chen, R., & Liang, F. (2026). Valeriana species and insomnia: multi-organ mechanisms and translational perspectives. Pharmaceutical Biology, 64(1), 615–638. PMC: PMC13094296
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 9 references cited
