Overview: Circadian disruption occurs when the body’s internal ~24-hour clock — controlled by the suprachiasmatic nucleus in the hypothalamus — loses alignment with the external light-dark cycle or with the body’s own organs and tissues. Five mechanisms link circadian disruption to fragmented sleep, 3am wakeups, and light shallow sleep:
- Orexin timing misalignment — the suprachiasmatic nucleus’s wake-promoting relay extends into the night or withdraws too early, fragmenting sleep-wake transitions and reducing sleep depth
- Peripheral clock decoupling — organs running on conflicting schedules produce metabolic and hormonal outputs that interrupt sleep at phase boundaries
- Temperature gate misalignment — the circadian drop in core body temperature that initiates sleep onset moves or flattens, delaying or preventing sleep entry
- Cortisol phase advance — the circadian cortisol rise begins hours earlier than intended wake time, producing characteristic early-morning waking
- Melatonin misalignment — the timing cue that tells the body night has arrived shifts or weakens, moving the sleep window away from intended bedtime
Aging, inflammation, hypoxia, insulin resistance, artificial light exposure, irregular schedules, caffeine, exercise timing, gut microbiome changes, and certain medications can each disrupt these pathways independently or in combination.
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 40+ references cited
What Is Circadian Rhythm?
Your body runs on a clock. The suprachiasmatic nucleus — a pair of small nuclei in the anterior hypothalamus — receives light input through the retinohypothalamic tract and uses that input to synchronize the body’s internal timing to the external light-dark cycle.
The molecular basis of this clock is a transcription-translation feedback loop. The proteins BMAL1 and CLOCK form a heterodimer that activates transcription of the PER and CRY genes. As PER and CRY proteins accumulate, they feed back to inhibit BMAL1/CLOCK activity, suppressing their own transcription. This loop takes approximately 24 hours to complete — and it runs in every cell in the body (Annals of Medicine, 2025).
The suprachiasmatic nucleus is the master pacemaker, but it is not the only clock. Every organ — the liver, gut, kidneys, adrenal glands, skeletal muscle — has its own peripheral clock running the same molecular loop. The suprachiasmatic nucleus synchronizes these peripheral clocks through neural projections, hormones (including cortisol and melatonin), and autonomic nervous output (Albrecht, Neuron, 2012).
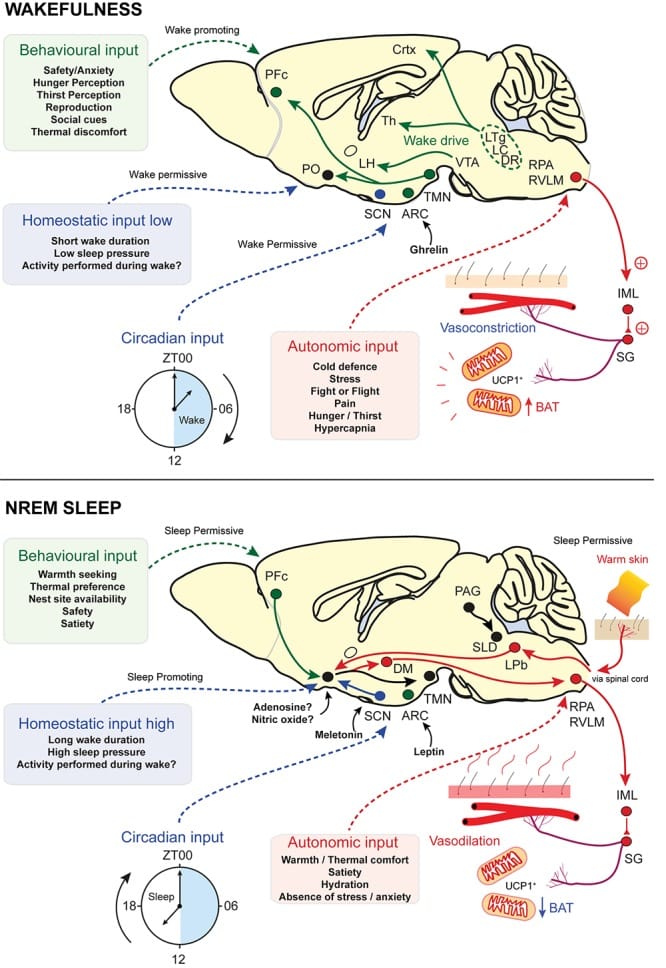
When circadian timing is intact, these distributed clocks coordinate: body temperature drops in the evening, melatonin rises, cortisol falls, and the brain transitions from wakefulness into consolidated sleep. When circadian timing breaks down, each of these coordinated outputs can go wrong independently.
The health consequences of circadian disruption extend beyond sleep: cardiovascular risk, metabolic regulation, cognitive function, and immune timing are all circadian-controlled. But the circadian clock doesn’t only regulate these broad health functions. It directly controls when and how you sleep — through at least five distinct mechanisms.
How Does Circadian Disruption Fragment Sleep and Contribute to 3AM Wakeups and Light Shallow Sleep?
What Is Circadian Disruption and How Does It Affect Sleep Architecture and Disrupt Sleep?
The suprachiasmatic nucleus does not directly generate sleep or wakefulness. It relays timing information through the dorsomedial hypothalamus to the orexin (hypocretin) neurons in the lateral hypothalamus — and these orexin neurons are what stabilize the boundary between sleep and wake states.
Orexin is a nonredundant integrator. A 2022 review in Journal of Internal Medicine (Jacobson, Hoyer, de Lecea) described the orexin neuropeptide as a hub that receives circadian, metabolic, and emotional inputs and converts them into a single consolidated wake-promoting output. When orexin-producing neurons are lost in humans, the result is narcolepsy type 1 — characterized by sleep intruding into wakefulness — establishing that orexin is essential for maintaining the stability of waking states. Orexin activity is temporally gated by the circadian clock: it amplifies wakefulness during the active phase and withdraws during the sleep phase.
What happens when the orexin relay weakens? A 2025 study in Neurobiology of Disease (Yuge et al.) tested this in a Rett syndrome mouse model with attenuated orexin receptor expression. These animals slept in short, fragmented bouts with frequent transitions between wakefulness and NREM sleep during their active period — the circadian clock itself was intact, but orexin’s stabilizing output was insufficient to maintain consolidated wake or sleep states. The deficit was selective: the clock was running, but the output couldn’t hold.
Aging may produce a milder version of this same pattern. A 2026 review in Ageing Research Reviews (Alhajaji et al.) described how normal aging is associated with reduced slow-wave and REM sleep, decreased sleep efficiency, increased fragmentation, and dampened circadian amplitude — a phenotype that resembles partial orexin weakening. Dual orexin receptor antagonists show promise for insomnia in older adults, suggesting that the orexin-circadian interface is undercharacterized but relevant in age-related sleep disruption.
What Are Peripheral Clocks, What Is Peripheral Clock Decoupling, and How Does It Contribute to Poor Sleep and Light Shallow Sleep?
The suprachiasmatic nucleus synchronizes peripheral organ clocks through electrical signaling, endocrine outputs, and metabolic cues (Albrecht, Neuron, 2012). But peripheral clocks also respond to local inputs — particularly feeding and fasting cycles — that can operate independently of light.
This creates a vulnerability. If feeding timing conflicts with the light-dark cycle — as it does with late-night eating or erratic meal schedules — peripheral clocks can decouple from the suprachiasmatic nucleus while the central clock remains intact.
A 2015 study in the FASEB Journal (Cuesta, Cermakian, Boivin) demonstrated that glucocorticoids can entrain molecular clock components in human peripheral cells — and that peripheral clocks in blood mononuclear cells take longer to reset than the central clock after schedule changes. Their work confirmed that central and peripheral clocks can run on different schedules simultaneously, with different re-entrainment rates.
A 2024 review in Advances in Experimental Medicine and Biology (Engin) mapped how this works at the molecular level: peripheral clocks in the liver respond to feeding/fasting state, nutrients, and sleep-wake cycles — distinct from the light-entrained suprachiasmatic nucleus. Night-eating specifically decouples peripheral clocks from central timing, while time-restricted eating restores alignment. In multi-oscillator models, this desynchronization is mediated by exosome-based intercellular communication between tissues.
Social jetlag — the gap between biological clock timing and social obligations — is common in industrialized populations and represents a chronic form of this misalignment.
How Does Core Body Temperature Change Throughout the Day and How Does That Affect Sleep Onset and Falling Asleep?
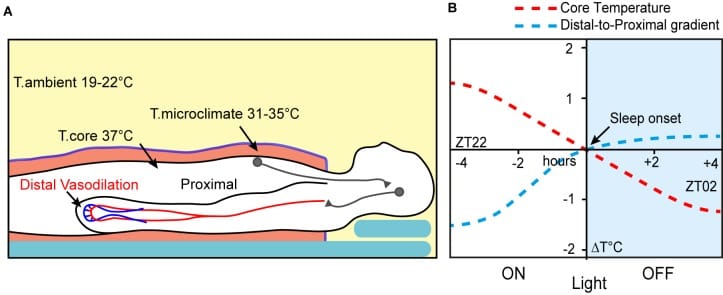
Sleep onset is gated by thermoregulation. As evening approaches, the suprachiasmatic nucleus reduces sympathetic tone to peripheral vasculature, allowing blood to flow to the hands and feet. Heat dissipates through the skin. Core body temperature drops — by approximately 1-1.5°C from its afternoon peak (Harding et al., Frontiers in Neuroscience, 2019). Warm-sensing neurons in the preoptic and anterior hypothalamus activate during sleep onset and NREM sleep, driving the autonomic changes that lower core temperature (Szymusiak, Handbook of Clinical Neurology, 2018).
A review in Neuropsychopharmacology (Krauchi and Wirz-Justice, 2001) found that the distal-proximal skin temperature gradient — the difference between hand/foot temperature and trunk temperature, reflecting heat loss from the core — was the single best predictor of how quickly someone falls asleep. It outperformed core body temperature itself, heart rate, melatonin onset, and subjective sleepiness.
When the circadian temperature rhythm is misaligned, this gate doesn’t open on time. A 2021 study in Sleep Medicine (Kimura et al.) demonstrated this in 28 school-age children with circadian rhythm sleep-wake disorder using fixed lights-out at 21:00, morning phototherapy, and light exercise. Sleep onset shifted from 23:53 to 21:17 (p < 0.001). Post-treatment mean core body temperature was lower (p = 0.011) and minimum sleep temperature was lower (p < 0.001) — as circadian phase realigned, the temperature rhythm followed, and the thermoregulatory gate opened earlier.
Hormonal changes can compound the temperature effect. A 2024 review in Menopause (Maki, Panay, Simon) described how hypothalamic KNDy neurons (kisspeptin/neurokinin B/dynorphin) integrate the gonadotropin-releasing hormone pathway with body temperature control and circadian sleep-wake regulation — linking hormone-driven thermoregulatory disruption to sleep onset failures.
How Does the Circadian Rhythm Affect Cortisol Timing and How Does Disrupted Cortisol Timing Disrupt Sleep and Contribute to 3AM Wakeups?
The hypothalamic-pituitary-adrenal axis follows a circadian pattern. Cortisol drops after sleep onset, reaches its lowest point in the first few hours of the night, then begins a gradual rise that peaks shortly after waking — the cortisol awakening response. Deep sleep actively suppresses cortisol output, and the morning rise helps transition the brain back to wakefulness.
A study in Hormone Research (Van Cauter et al., 1998) showed that in older adults, the 24-hour cortisol rhythm is dampened in amplitude and phase-advanced — the morning cortisol rise begins earlier. The cortisol nadir is higher. Sleep loss in young subjects produced the same endocrine disturbances as aging, suggesting that declining sleep quality may itself drive cortisol rhythm changes.
The functional consequence is measurable. A review in American Journal of Geriatric Psychiatry (Buckley and Schatzberg, 2005) linked elevated nocturnal cortisol and a blunted cortisol nadir to reduced slow-wave sleep and impaired declarative memory consolidation.
Because cortisol functions as a wake-promoting hormone, a phase-advanced cortisol rise can produce early-morning waking — the 2-3am wakeup where the mind is alert and active, not groggy or confused.
A 2023 review in Neuroscience and Biobehavioral Reviews (de Leeuw et al.) documented that the circadian clock regulates cortisol as part of a broad set of bodily functions, and aging, shift work, and jet lag each disrupt the circadian cortisol pattern.
This mechanism is distinct from the stress-driven cortisol elevation covered in the autonomic cause page. The autonomic page addresses chronic HPA axis overactivation — cortisol levels that are elevated because of sustained stress. This section addresses the clock advancing cortisol’s timing — the rise begins earlier, regardless of whether total cortisol is elevated.
How Does the Circadian Rhythm Affect Melatonin Release and How Does Disrupted Melatonin Timing Disrupt Sleep and Contribute to Light Shallow Sleep?
Melatonin acts through MT1 and MT2 receptors in the suprachiasmatic nucleus and functions as a circadian coordinator — telling the body when night has arrived — rather than as a sleep-inducing agent (Comai & Gobbi, Journal of Pineal Research, 2024).
The dim-light melatonin onset is the most accurate marker of circadian phase position. A review in Progress in Neuro-Psychopharmacology and Biological Psychiatry (Pandi-Perumal et al., 2007) established DLMO as the gold-standard tool for evaluating sleep onset problems, determining whether an individual is entrained to a 24-hour light/dark cycle, and identifying optimal timing for chronotherapy.
DLMO responds predictably to light exposure. Morning light advances circadian phase; evening light delays it. Exogenous melatonin has a phase response curve approximately 12 hours out of phase with the light curve — meaning melatonin administered in the afternoon advances the clock, while melatonin in the morning delays it (Lewy et al., Ciba Foundation Symposium, 1995).
When DLMO shifts, the consequences are measurable. A 2024 study in Sleep (Chakraborty et al.) found that myopic children had DLMO delayed by 1 hour and 8 minutes compared to non-myopic peers (9:07 PM vs. 7:59 PM, p = 0.002) and approximately 42% lower nocturnal melatonin output (p = 0.001). These children also had delayed sleep onset, reduced sleep quality, and more evening-type chronotype preference.
The existing TLV melatonin articles cover supplementation in detail. This section is about the clock’s melatonin timing cue — what happens when the internal signal moves, not what happens when you take melatonin as a supplement.
What Triggers or Worsens Circadian Sleep Disruption?
How Does the Gut Microbiome Influence Circadian Timing?
A 2025 review in Frontiers in Microbiology mapped this pathway in experimental models: butyrate produced by gut bacteria inhibits histone deacetylases (HDACs), leading to chromatin remodeling and enhanced expression of the clock gene PER2. Short-chain fatty acids can also reach the central nervous system by crossing the blood-brain barrier or by activating vagus nerve pathways, potentially modulating central circadian rhythms.
The relationship is bidirectional. Circadian disruption alters the gut microbiome, and an altered microbiome weakens circadian entrainment — creating a reinforcing loop. Time-restricted feeding has been shown to restore microbial rhythmicity in models of circadian disruption, but the improvement requires a functional intestinal circadian clock to translate into anti-inflammatory outcomes.
How Does Aging Change Circadian Function?
Van Cauter et al. (1998) documented the endocrine consequences: the cortisol rhythm flattens, the cortisol nadir rises, and the morning cortisol response phase-advances. A 2026 review in Ageing Research Reviews (Alhajaji et al.) described how these changes produce reduced slow-wave sleep, decreased sleep efficiency, and increased fragmentation — with insomnia prevalence reaching 20-40% in older populations.
A 2026 review in Frontiers in Neuroscience linked age-related melatonin decline to pineal calcification and reduced circadian amplitude, noting that melatonin’s antioxidant and mitochondrial-protective functions decline in parallel.
The degree of circadian weakening varies between individuals. Consistent light exposure, regular meal timing, and physical activity can help maintain circadian amplitude with age.
Can Medications Disrupt Circadian Timing?
Beta-adrenergic blockers reduce nocturnal melatonin secretion by interfering with the sympathetic pathway from the suprachiasmatic nucleus to the pineal gland — the same pathway that normally triggers melatonin production at night. For adults taking beta blockers for blood pressure or heart rate management, this can mean a delayed or blunted melatonin onset independent of any behavioral factor.
The timing of medication administration matters more broadly. Research in chronopharmacology has found that circadian rhythms can influence how effectively a drug works, depending on which circadian-regulated pathways it interacts with and when during the 24-hour cycle it is administered.
This is explanatory, not prescriptive. If a medication is serving a necessary function, that conversation belongs with your physician. The point is that medication effects on circadian timing are a recognized mechanism.
Can Inflammation Disrupt Peripheral Clocks Directly?
A 2024 study in the International Journal of Molecular Sciences showed that lipopolysaccharide-induced inflammation perturbed clock gene oscillations in the hypothalamus, hippocampus, and liver — and triggered neuroinflammation that altered the diurnal behavior of microglia. The clock disruption was a consequence of inflammation, not of any behavioral or lifestyle change.
A 2025 review in Clinical Science documented how inflammatory bowel disease dampens circadian rhythms in the colon, possibly by suppressing BMAL1 expression. This can create a reinforcing cycle: circadian disruption hinders the development of an adequate immune response, and inadequate immune regulation amplifies inflammation — which further suppresses clock gene expression.
The molecular interface is direct. A 2025 review in Frontiers in Immunology described how clock proteins interact directly with components of the NF-kB pathway — meaning inflammation and circadian regulation are mechanistically interlinked, not just correlated.
Does Low Oxygen Disrupt Peripheral Clocks?
A study in PNAS (Manella et al., 2020) showed that in vivo hypoxia phase-shifted peripheral clocks in a mouse model, but not uniformly — different tissues responded differently. Feeding restored the liver clock’s original phase, but had no effect on the lung and kidney clocks, which retained their hypoxia-induced phase shift. This means different peripheral clocks respond to different zeitgebers, and hypoxia can create intertissue desynchrony — organs literally running on different schedules.
For individuals with obstructive sleep apnea, this has direct relevance. Repeated oxygen desaturation events throughout the night create ongoing peripheral clock misalignment independent of any behavioral factor. A 2024 review in Circulation Research described the circadian clock-hypoxia interaction as a contributor to cardiovascular risk in sleep-disordered breathing.
Can Insulin Resistance Affect Circadian Timing?
A 2022 review in International Journal of Environmental Research and Public Health (Catalano et al.) described the feedback loop: peripheral clock desynchronization impairs insulin sensitivity, and impaired insulin signaling disrupts hepatic clock gene oscillations — creating a self-reinforcing cycle between metabolic and circadian disruption.
A 2024 review in the Journal of Pineal Research (Speksnijder et al.) examined how the suprachiasmatic nucleus controls glucose homeostasis through neural projections, glucocorticoids, melatonin, and the autonomic nervous system. When circadian desynchrony disrupts these coordinated outputs, glucose regulation loses its normal 24-hour pattern.
This is a metabolic-circadian feedback loop. Type 2 diabetes, metabolic syndrome, and chronic circadian misalignment can each initiate the cycle.
How Does Artificial Light at Night Affect the Circadian Clock?
A 2023 study in Communications Biology found that high melanopic light in the evening caused dose-dependent increases in sleep latency and reductions in melatonin concentration, while low melanopic light shortened time to fall asleep. A study in Chronobiology in Medicine (2024) reported that 2 hours of evening light exposure produced an average 1.1-hour circadian phase delay. A 2025 preprint (medRxiv, not yet peer-reviewed) found that melanopic equivalent daylight illuminance was a more sensitive predictor of sleep quality and structure than traditional photopic illuminance measures.
Does an Irregular Schedule Disrupt Circadian Timing?
A 2025 study in Sleep (Hasler et al.) measured circadian phase shifts between school nights and weekends in high-school students, demonstrating that even routine weekly schedule variation produces measurable DLMO shifts. A 2025 review in Circulation Research linked sleep irregularity to cardiometabolic risk, framing it as a component of multidimensional sleep health.
How Does Caffeine Affect the Circadian Clock?
A double-blind, placebo-controlled study in Science Translational Medicine (Burke et al., 2015) showed that a dose equivalent to a double espresso consumed 3 hours before bedtime delayed the circadian melatonin rhythm by approximately 40 minutes. In cell cultures, continuous caffeine exposure lengthened the period of the cellular circadian clock through adenosine receptor and cAMP pathways — suggesting caffeine affects circadian timing at the molecular level, not just through adenosine-mediated sleep pressure.
Can Exercise Timing Shift Circadian Rhythms?
Research on exercise timing and circadian rhythms has found that evening exercise can raise nocturnal body temperature and reduce REM sleep duration, while morning exercise tends to reinforce the central clock. A study on circadian phase response to exercise (Thomas et al., Journal of Physiology, 2020) found that morning exercise induced phase advance of approximately 0.6 hours.
Exercise-induced factors — heat, glucocorticoid release, and mechanical loading — can entrain peripheral clocks in tissues including skeletal muscle and connective tissue, making exercise timing a relevant non-photic zeitgeber for peripheral circadian alignment.
How Do You Know If Circadian Disruption Is Affecting Your Sleep?
What Does Circadian Sleep Disruption Feel Like?
- Can’t fall asleep at intended time despite being tired. The body is fatigued but the circadian clock has not opened the sleep window — melatonin onset has not occurred, core body temperature has not dropped, and the orexin wake-promoting drive is still active.
- Waking 2-3 hours before your alarm, fully alert. This is consistent with a phase-advanced cortisol rhythm — the circadian cortisol rise has arrived earlier than intended, triggering an alert wakeup before the desired wake time.
- Energy peak at the wrong time of day. Alert at midnight, sluggish at 8am — or the reverse. The circadian rhythm is running, but on a different schedule from the one your obligations require.
- Large weekend shift. Sleeping 1-2 or more hours later on free days compared to workdays suggests the biological clock is misaligned with the social schedule — the definition of social jetlag.
- Feeling jet-lagged without having traveled. The subjective experience of internal desynchronization — when peripheral clocks, temperature rhythm, cortisol rhythm, and melatonin timing are not aligned with each other or with your schedule.
What Can Be Measured?
- Melatonin rhythm via saliva sampling. Saliva samples collected at multiple timepoints under dim light conditions can identify when melatonin begins to rise — the DLMO — and how it compares to your intended sleep time. A gap between DLMO and bedtime indicates phase misalignment.
- Cortisol rhythm via saliva sampling. Salivary cortisol collected at multiple timepoints — at wake, 30 minutes post-wake, afternoon, and bedtime — reveals the shape of the 24-hour cortisol curve: whether it is flattened, whether the cortisol awakening response is blunted, and whether bedtime cortisol remains elevated or has phase-advanced.
- Skin temperature rhythm. Wearables that track overnight temperature trends can capture the circadian temperature rhythm. The amplitude of this rhythm correlates with sleep quality (Tai et al., JCSM, 2023).
- Actigraphy. Rest-activity pattern regularity over days to weeks captures circadian stability at the behavioral level. Irregular patterns suggest circadian misalignment even when individual nights look normal.
Can Multiple Causes Contribute at Once?
Find out which causes are driving your 3am wakeups with the 3AM Decoder →
Frequently Asked Questions
Is Circadian Disruption the Same as Insomnia?
The distinction matters because the interventions differ. Insomnia interventions typically focus on behavioral habits and cognitive patterns. Circadian interventions focus on light timing, meal timing, and realigning the phase of the internal clock.
Does Aging Always Weaken Circadian Rhythms?
The relationship between aging and circadian weakening is not deterministic. Some individuals in their 70s maintain robust circadian rhythms, while others show measurable dampening in their 50s. The modifiable factors — light, meals, exercise — are all zeitgebers that the suprachiasmatic nucleus uses to calibrate its output.
Can a Disrupted Circadian Rhythm Be Reset?
The differential re-entrainment rate matters. After a change in schedule, the suprachiasmatic nucleus may realign within 2-3 days, but peripheral clocks in blood cells, liver, and gut can lag by days to weeks. During this transition period, the body is running on multiple schedules simultaneously — which is why recovery from jet lag or shift work often takes longer than expected.
Is Melatonin Supplementation the Answer to Circadian Sleep Problems?
The existing TLV melatonin articles cover dosing, timing, and supplementation strategies in detail. This page focuses on the circadian mechanisms that determine whether the body’s own melatonin timing cue is aligned with the intended sleep schedule.
Related Reading
- How Long Does It Take to Reset Your Circadian Rhythm?
- Why Does Your Wake Time Advance by Decades — and Can You Reverse Circadian Phase Advance After 50?
- Can Chronic Inflammation Disrupt Your Circadian Clock Genes?
- What Is Orexin and Why Does It Keep You Awake at Night?
- What Happens to Your Circadian Clock Cells After 50?
- Autonomic Sleep Disruption
- Hormonal Sleep Disruption in Men
- Hormonal Sleep Disruption in Women
- Inflammatory Sleep Disruption
- Metabolic Sleep Disruption
References
Orexin and Sleep Architecture
1. Jacobson, L. H., Hoyer, D., & de Lecea, L. (2022). Hypocretins (orexins): The ultimate translational neuropeptides. Journal of Internal Medicine, 291(5), 533-556. https://pubmed.ncbi.nlm.nih.gov/35043499/
2. Yuge, K., Takahashi, T., Kawahara, Y., Sakai, Y., Sato, T., Kakuma, T., Nishi, A., Matsuishi, T., & Yamashita, Y. (2025). Attenuated orexinergic signaling underlies sleep-wake problems in a Mecp2-null mouse model of Rett syndrome. Neurobiology of Disease, 214, 107035. https://pubmed.ncbi.nlm.nih.gov/40684866/
3. Alhajaji, R., Jahrami, H., Pandi-Perumal, S. R., & BaHammam, A. S. (2026). Sleep health in the older adults: Architecture, circadian changes, and common sleep disorders. Ageing Research Reviews, 118, 103101. https://pubmed.ncbi.nlm.nih.gov/41825783/
4. Tabuchi, S., Tsunematsu, T., Kilduff, T. S., Sugio, S., Xu, M., Tanaka, K. F., Takahashi, S., Tominaga, M., & Yamanaka, A. (2013). Influence of inhibitory serotonergic inputs to orexin/hypocretin neurons on the diurnal rhythm of sleep and wakefulness. Sleep, 36(9), 1391-1404. https://pubmed.ncbi.nlm.nih.gov/23997373/
5. Riedel, C. S., Milan, J. B., Jorgensen, N. R., Jennum, P., & Juhler, M. (2026). Overnight dynamics of ventricular cerebrospinal fluid amyloid-beta, lactate and hypocretin in patients with hydrocephalus: A pilot study. Journal of Sleep Research, e70292. https://pubmed.ncbi.nlm.nih.gov/41607364/
Peripheral Clocks and Desynchronization
6. Albrecht, U. (2012). Timing to perfection: The biology of central and peripheral circadian clocks. Neuron, 74(2), 246-260. https://pubmed.ncbi.nlm.nih.gov/22542179/
7. Cuesta, M., Cermakian, N., & Boivin, D. B. (2015). Glucocorticoids entrain molecular clock components in human peripheral cells. FASEB Journal, 29(4), 1360-1370. https://pubmed.ncbi.nlm.nih.gov/25500935/
8. Engin, A. (2024). Misalignment of circadian rhythms in diet-induced obesity. Advances in Experimental Medicine and Biology, 1460, 27-71. https://pubmed.ncbi.nlm.nih.gov/39287848/
9. Boivin, D. B., Boudreau, P., & Kosmadopoulos, A. (2022). Disturbance of the circadian system in shift work and its health impact. Journal of Biological Rhythms, 37(1), 3-28. https://pubmed.ncbi.nlm.nih.gov/34969316/
10. Song, Q. X., Suadicani, S. O., Negoro, H., Jiang, H. H., Jabr, R., Fry, C., Xue, W., & Damaser, M. S. (2025). Disruption of circadian rhythm as a potential pathogenesis of nocturia. Nature Reviews Urology, 22(5), 276-293. https://pubmed.ncbi.nlm.nih.gov/39543359/
Core Body Temperature and Sleep Onset
11. Szymusiak, R. (2018). Body temperature and sleep. Handbook of Clinical Neurology, 156, 341-351. https://pubmed.ncbi.nlm.nih.gov/30454599/
12. Krauchi, K., & Wirz-Justice, A. (2001). Circadian clues to sleep onset mechanisms. Neuropsychopharmacology, 25(S5), S92-S96. https://pubmed.ncbi.nlm.nih.gov/11682282/
13. Kimura, S., Takaoka, Y., Toyoura, M., Kohira, S., & Ohta, M. (2021). Core body temperature changes in school-age children with circadian rhythm sleep-wake disorder. Sleep Medicine, 87, 97-104. https://pubmed.ncbi.nlm.nih.gov/34547649/
14. Maki, P. M., Panay, N., & Simon, J. A. (2024). Sleep disturbance associated with the menopause. Menopause, 31(8), 724-733. https://pubmed.ncbi.nlm.nih.gov/38916279/
15. Tai, Y., Obayashi, K., Yamagami, Y., & Saeki, K. (2023). Association between circadian skin temperature rhythms and actigraphic sleep measures in real-life settings. Journal of Clinical Sleep Medicine, 19(7), 1281-1292. https://pubmed.ncbi.nlm.nih.gov/37394793/
16. Harding, E. C., Franks, N. P., & Wisden, W. (2019). The temperature dependence of sleep. Frontiers in Neuroscience, 13, 336. https://pubmed.ncbi.nlm.nih.gov/31105512/
Cortisol Circadian Timing
17. Van Cauter, E., Plat, L., Leproult, R., & Copinschi, G. (1998). Alterations of circadian rhythmicity and sleep in aging: Endocrine consequences. Hormone Research, 49(3-4), 147-152. https://pubmed.ncbi.nlm.nih.gov/9550116/
18. Buckley, T. M., & Schatzberg, A. F. (2005). Aging and the role of the HPA axis and rhythm in sleep and memory-consolidation. American Journal of Geriatric Psychiatry, 13(5), 344-352. https://pubmed.ncbi.nlm.nih.gov/15879582/
19. de Leeuw, M., Verhoeve, S. I., van der Wee, N. J. A., van Hemert, A. M., Vreugdenhil, E., & Coomans, C. P. (2023). The role of the circadian system in the etiology of depression. Neuroscience and Biobehavioral Reviews, 153, 105383. https://pubmed.ncbi.nlm.nih.gov/37678570/
20. Liu, P. Y., & Reddy, R. T. (2022). Sleep, testosterone and cortisol balance, and ageing men. Reviews in Endocrine and Metabolic Disorders, 23(6), 1323-1339. https://pubmed.ncbi.nlm.nih.gov/36152143/
Melatonin and Dim-Light Melatonin Onset
21. Pandi-Perumal, S. R., Smits, M., Spence, W., Srinivasan, V., Cardinali, D. P., Lowe, A. D., & Kayumov, L. (2007). Dim light melatonin onset (DLMO): A tool for the analysis of circadian phase in human sleep and chronobiological disorders. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 31(1), 1-11. https://pubmed.ncbi.nlm.nih.gov/16884842/
22. Lewy, A. J., Sack, R. L., Blood, M. L., Bauer, V. K., Cutler, N. L., & Thomas, K. H. (1995). Melatonin marks circadian phase position and resets the endogenous circadian pacemaker in humans. Ciba Foundation Symposium, 183, 303-321. https://pubmed.ncbi.nlm.nih.gov/7656692/
23. Chakraborty, R., Seby, C., Scott, H., Tang, V., Kemps, E., Anstice, N., Juers, E., Lovato, N., Taranath, D. A., Mills, R. A., & Lack, L. C. (2024). Delayed melatonin circadian timing, lower melatonin output, and sleep disruptions in myopic, or short-sighted, children. Sleep, 47(1), zsad265. https://pubmed.ncbi.nlm.nih.gov/37823859/
24. Lim, D., Choi, S. J., Song, Y. M., Park, H. R., Joo, E. Y., & Kim, J. K. (2025). Enhanced circadian phase tracking: A 5-h DLMO sampling protocol using wearable data. Journal of Biological Rhythms, 40(3), 249-261. https://pubmed.ncbi.nlm.nih.gov/40017128/
25. Murray, J. M., Stone, J. E., Abbott, S. M., Bjorvatn, B., Burgess, H. J., Cajochen, C., … & Rajaratnam, S. M. W. (2024). A protocol to determine circadian phase by at-home salivary dim light melatonin onset assessment. Journal of Pineal Research, 76(5), e12994. https://pubmed.ncbi.nlm.nih.gov/39158010/
26. Comai, S., & Gobbi, G. (2024). Melatonin, melatonin receptors and sleep: Moving beyond traditional views. Journal of Pineal Research, 76(7), e13011. PMID 39400423. https://doi.org/10.1111/jpi.13011
Triggers and Disruptors
27. Burke, T. M., Markwald, R. R., McHill, A. W., Chinoy, E. D., Snider, J. A., Bessman, S. C., Jung, C. M., O’Neill, J. S., & Wright, K. P., Jr. (2015). Effects of caffeine on the human circadian clock in vivo and in vitro. Science Translational Medicine, 7(305), 305ra146. https://pubmed.ncbi.nlm.nih.gov/26378246/
28. Zheng, B., Wang, L., Sun, S., Yuan, X., & Liang, Q. (2025). The molecular interplay between the gut microbiome and circadian rhythms: An integrated review. Frontiers in Microbiology, 16, 1712516. https://pubmed.ncbi.nlm.nih.gov/41425940/
29. Chauhan, S., et al. (2024). Systemic inflammation disrupts circadian rhythms and diurnal neuroimmune dynamics. International Journal of Molecular Sciences, 25(13), 7458. https://www.mdpi.com/1422-0067/25/13/7458
30. Collins, S. L., et al. (2025). The circadian clock, metabolism, and inflammation — the holy trinity of inflammatory bowel diseases. Clinical Science, 139(13), 777-798. https://portlandpress.com/clinsci/article/139/13/777/236298/
31. Frontiers in Immunology. (2025). Circadian clocks and adaptive immune function: From mechanisms to therapeutic applications. Frontiers in Immunology, 16, 1697854. https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1697854/full
32. Manella, G., Aviram, R., Bolshette, N., Muvkadi, S., Golik, M., Smith, D. F., & Asher, G. (2020). Hypoxia induces a time- and tissue-specific response that elicits intertissue circadian clock misalignment. PNAS, 117(1), 779-786. PMID 31848250. https://doi.org/10.1073/pnas.1914112117
33. Circulation Research. (2024). Circadian clock and hypoxia. Circulation Research, 135(8), 913-928. https://www.ahajournals.org/doi/10.1161/CIRCRESAHA.124.323518
34. Catalano, F., De Vito, F., Cassano, V., Fiorentino, T. V., Sciacqua, A., & Hribal, M. L. (2022). Circadian clock desynchronization and insulin resistance. International Journal of Environmental Research and Public Health, 20(1), 29. https://pubmed.ncbi.nlm.nih.gov/36612350/
35. Speksnijder, E. M., Bisschop, P. H., Siegelaar, S. E., Stenvers, D. J., & Kalsbeek, A. (2024). Circadian desynchrony and glucose metabolism. Journal of Pineal Research, 76(4), e12956. https://pubmed.ncbi.nlm.nih.gov/38695262/
Additional Sources
36. Annals of Medicine. (2025). Circadian clock genes and insomnia: Molecular mechanisms and therapeutic implications. Annals of Medicine, 57(1), 2576643. https://www.tandfonline.com/doi/full/10.1080/07853890.2025.2576643
37. Communications Biology. (2023). Melanopic irradiance defines the impact of evening display light on sleep latency, melatonin and alertness. Communications Biology, 6, 163. https://www.nature.com/articles/s42003-023-04598-4
38. Chronobiology in Medicine. (2024). Impacts of blue light exposure from electronic devices on circadian rhythm and sleep disruption in adolescent and young adult students. Chronobiology in Medicine, 6(4), 167. https://www.chronobiologyinmedicine.org/journal/view.php?number=167
39. Circulation Research. (2025). Sleep irregularity, circadian disruption, and cardiometabolic disease risk. Circulation Research, 137(4), 525613. https://www.ahajournals.org/doi/10.1161/CIRCRESAHA.125.325613
40. Hasler, B. P., Oryshkewych, N., Wallace, M. L., Clark, D. B., Siegle, G. J., & Buysse, D. J. (2025). Circadian phase in high-school students: Weekday-weekend shifts and relationships to other sleep/circadian characteristics. Sleep, 48(4), zsaf031. https://pubmed.ncbi.nlm.nih.gov/39901722/
41. Frontiers in Neuroscience. (2026). Rhythms of life: Melatonin, nutrition, sleep, and antioxidant strategies for healthy aging. Frontiers in Neuroscience, 20, 1736978. https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2026.1736978/full
