Millions of adults over 40 take beta blockers for blood pressure or heart rate management. Insomnia is listed as a side effect on the packaging, but the mechanism behind it is rarely explained — and it is not sedation or stimulation. It is a pharmacological disruption of the circadian melatonin production pathway. Understanding this mechanism matters for long-term brain health, because melatonin does more than time sleep onset: it is a neuroprotective antioxidant, and chronic suppression has measurable consequences.
This article covers the pineal mechanism, which beta blockers suppress melatonin the most, the evidence for melatonin supplementation as a countermeasure, and what prolonged melatonin suppression may do to the brain. It does not cover all medications that affect sleep — for the broader picture of how circadian disruption drives 3 AM wakeups, see the Circadian Rhythm Disruption overview on circadian sleep disruption.
?-
How Do Beta Blockers Block Melatonin Production in the Pineal Gland?
Melatonin production follows a neurochemical chain each night. The suprachiasmatic nucleus (SCN) — the master circadian clock in the hypothalamus — sends a timing instruction down a sympathetic nerve pathway to the superior cervical ganglion. From there, postganglionic neurons release norepinephrine onto the surface of pinealocytes in the pineal gland. Norepinephrine binds to beta-1 adrenergic receptors on these cells, which activates cyclic AMP (cAMP), then protein kinase A, then AANAT — the rate-limiting enzyme that converts serotonin into N-acetylserotonin, the immediate precursor to melatonin. Beta blockers sit on the beta-1 receptor and prevent norepinephrine from binding. The entire downstream cascade — cAMP, protein kinase A, AANAT activation, melatonin synthesis — stops.
Cardinali and Vacas (1987) described the molecular pathway from SCN timing instruction to melatonin output in a comprehensive review of pineal regulation (Cardinali & Vacas, 1987).
Rommel and Demisch (1994) confirmed this in humans. In a cohort of 42 hypertensive adults, 10 weeks of propranolol or ridazolol produced a 50% reduction in urinary 6-sulfatoxymelatonin — the primary metabolite used to measure melatonin output. This was a direct, measurable halving of melatonin production from beta-1 receptor blockade (Rommel & Demisch, 1994).
A 2021 case report demonstrated the pathway is pharmacologically controllable in both directions. Gehrman and Anafi worked with a sighted adult with non-24-hour sleep-wake disorder — a condition where the circadian clock free-runs instead of locking to the 24-hour day. Standard melatonin supplementation alone did not stabilize the rhythm, because the individual’s endogenous melatonin was being produced at the wrong circadian phase and competing with the exogenous dose. The physicians added metoprolol (a beta blocker) to suppress the mistimed endogenous melatonin, then administered exogenous melatonin at the desired phase. Sleep onset advanced and stabilized. In this individual, the pineal beta-1 pathway proved pharmacologically controllable — it was suppressed and then restored to re-time circadian output (Gehrman & Anafi, 2021).
?-
Which Beta Blockers Suppress Melatonin the Most?
Not all beta blockers affect sleep equally. The variable that determines the degree of sleep disruption is lipophilicity — how readily the drug dissolves in fat and crosses the blood-brain barrier.
Betts and Alford (1985) tested this directly with polysomnography. In a placebo-controlled crossover trial, propranolol, metoprolol, and pindolol (all lipophilic) increased nocturnal awakenings and vivid dream recall compared to placebo. Atenolol (hydrophilic) did not. The distinction in subjective reports was consistent: agents that cross into the central nervous system increased awakenings and dream recall; atenolol did not, though it did reduce REM sleep frequency (Betts & Alford, 1985).
Eddin et al. (2025) confirmed this pattern in a systematic review and meta-analysis including 44 randomized controlled trials. Lipophilic beta blockers showed higher neuropsychiatric risk — including insomnia, vivid dreams, and dizziness — than hydrophilic agents. Propranolol carried the highest risk: a pooled risk ratio of 3.13 for dizziness versus placebo, compared to 1.72 for beta blockers as a class (Eddin et al., 2025).
Here is how common beta blockers rank by lipophilicity:
| Beta Blocker | Lipophilicity | Blood-Brain Barrier Penetration | Sleep Disruption Risk |
|?-|?-|?-|?-|
| Propranolol | High | Crosses readily | Higher |
| Metoprolol | Moderate | Crosses | Moderate-to-higher |
| Carvedilol | Moderate | Crosses | Moderate |
| Bisoprolol | Low-to-moderate | Limited crossing | Lower |
| Atenolol | Low | Minimal crossing | Lower |
| Nadolol | Low | Minimal crossing | Lower |
This is not a recommendation to change medications — it is information to discuss with a physician. If sleep disruption coincides with starting or switching a beta blocker, the lipophilicity of the drug is a relevant variable to examine.
?-
Can Melatonin Supplementation Fix Beta-Blocker Insomnia?
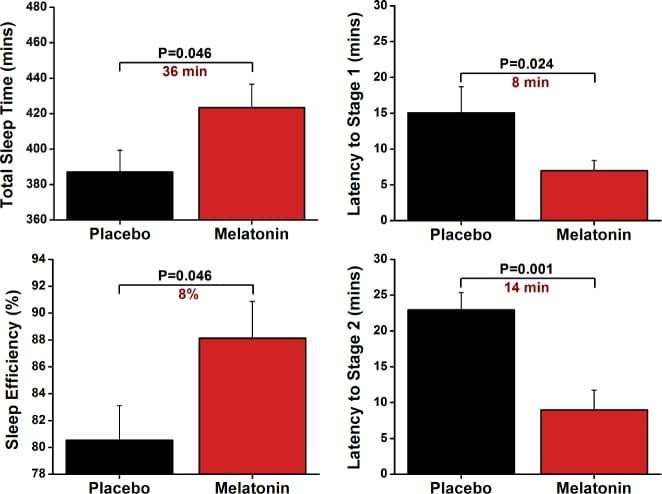
Scheer et al. (2012) conducted the definitive trial on this question. In a randomized, double-blind, placebo-controlled, parallel-group design, 16 hypertensive adults taking atenolol or metoprolol were randomized to receive either 2.5 mg of sustained-release melatonin or placebo nightly for three weeks. The results, verified by polysomnography:
- Total sleep time increased by 36 minutes
- Sleep efficiency improved by 7.6%
- Stage 2 sleep increased by 41 minutes
- Sleep onset latency to Stage 2 decreased by 14 minutes
These are large effect sizes for a low-dose, low-risk approach. For context, a 36-minute increase in total sleep time exceeds the effect size of many prescription sleep medications (Scheer et al., 2012).
The pharmacological rationale is straightforward: the drug blocks the production pathway, and exogenous melatonin replaces what the gland cannot produce while the blockade is active.
Exogenous melatonin during beta-blocker use is replacement, not supplementation in the conventional sense. The pineal gland’s output is pharmacologically suppressed. Exogenous melatonin restores what would otherwise be present.
On dosing: the Scheer RCT used 2.5 mg sustained-release, taken one hour before desired sleep onset. The sustained-release formulation matters because it more closely mimics the gland’s natural secretion profile — a gradual rise rather than a spike. Doses in the range of 0.5-3 mg are consistent with the replacement model; higher doses are not necessary when the goal is restoring a physiological level rather than inducing sedation.
?-
Can Prolonged Beta-Blocker Use Damage Your Brain?
Tikhomirova et al. (2022) studied 224 individuals — 114 on beta blockers and 110 controls — using both polysomnography and brain MRI. The researchers identified a melatonin threshold: individuals whose urinary melatonin metabolite fell at or below 16.8 micrograms per day showed both worse sleep parameters and more white-matter gliosis on imaging. White-matter gliosis is a marker of vascular brain damage — small-vessel injury visible on MRI that is associated with cognitive decline over time (Tikhomirova et al., 2022).
Melatonin’s role extends beyond sleep timing. It functions as an antioxidant and free radical scavenger, protects mitochondrial membranes, and reduces neuroinflammation. Chronic suppression of melatonin production may reduce these protections during the hours when the brain’s glymphatic clearance — the process that removes metabolic waste, including amyloid-beta — peaks. The connection between how melatonin suppresses orexin to enable sleep onset and these downstream neuroprotective effects underscores why the melatonin pathway matters beyond sleep onset alone.
This is an observational finding, not causal proof. The study cannot determine whether low melatonin caused the white-matter changes or whether both reflect a shared underlying vascular vulnerability. But the association between chronic melatonin suppression and measurable brain changes in individuals on long-term beta blockers is a reason to bring this up with a physician — especially for adults over 40 who are already managing cardiovascular risk factors and want to protect cognitive function as they age.
?-
Do Antidepressants Affect Your Body Clock?
SSRIs — selective serotonin reuptake inhibitors — increase serotonin concentration in the synaptic cleft. In the dorsal raphe nucleus, elevated serotonin suppresses REM-on neurons, which reduces both REM duration and REM density. These are well-characterized pharmacological effects of the drug class. For some individuals, this manifests as lighter sleep, more awakenings, or a sense that sleep is less restorative even when total hours are adequate.
Tricyclic antidepressants affect sleep through a different mechanism. Their anticholinergic properties reduce acetylcholine-driven REM initiation, which can suppress REM sleep and alter the timing of sleep-stage cycling. The sedating effects of some tricyclics (like amitriptyline) mask the REM suppression — the individual falls asleep faster but may spend less time in the sleep stages that support memory consolidation and emotional processing.
The broader field studying these interactions — chronopharmacology — examines how drug timing, drug class, and circadian phase interact to produce different outcomes. Eddin et al. (2025) noted that the neuropsychiatric effects of cardiovascular medications, including sleep disruption, vary based on whether the agent crosses the blood-brain barrier — the same lipophilicity principle that governs beta-blocker sleep effects.
For the full picture of how circadian disruption from multiple sources compounds to produce 3 AM wakeups, see the overview on circadian sleep disruption.
?-
Do Nonsteroidal Anti-Inflammatory Drugs or Steroids Interfere With Circadian Timing?
Corticosteroids like prednisone and dexamethasone bind to the same glucocorticoid receptors as endogenous cortisol. When taken in the evening or at high doses, they mimic a cortisol surge at a time when the body’s own cortisol should be at its lowest — during the hours leading into sleep. This can produce the “wired at 3 AM” pattern that many people on corticosteroids report: the exogenous glucocorticoid overrides the circadian cortisol trough, maintaining sympathetic tone and arousal when the brain should be in its deepest recovery phases. For more on how chronic stress hijacks arousal pathways, the cortisol-orexin interaction is covered in detail.
NSAIDs affect circadian timing through a different pathway. Prostaglandins — which NSAIDs inhibit — are involved in thermoregulation and sleep-wake modulation. Prostaglandin D2 is one of the strongest endogenous sleep-promoting substances. Suppressing its production with evening NSAID use can delay sleep onset or reduce sleep depth, though the magnitude varies by individual and by which NSAID is used.
Both drug classes illustrate a broader principle: medications that affect hormonal or inflammatory pathways can alter circadian timing as a downstream consequence, even when sleep disruption is not their intended or expected effect.
?-
Do Blood Pressure Medications Work Better at Night?
The concept behind chronotherapy is that drug efficacy and side-effect profiles vary depending on when in the 24-hour cycle the medication is taken. Blood pressure follows a circadian rhythm — it typically peaks in the morning and dips at night. Medications that target this rhythm may produce different results depending on whether they are taken at the peak or the trough of the cycle.
For beta blockers, evening dosing has been explored as a way to reduce melatonin suppression. The rationale: if the drug’s peak plasma concentration coincides with hours when melatonin production has already completed its primary window, the degree of suppression may be reduced. This is a hypothesis being investigated, not an established recommendation.
Dosing timing is one of the variables to raise with a physician when sleep disruption accompanies beta-blocker use — alongside lipophilicity, dose, and whether melatonin supplementation is appropriate.
?-
Sleep disruption from medication is rarely a single-cause problem. Beta-blocker-induced melatonin suppression might compound with hormonal changes, metabolic changes, or autonomic imbalance — especially in adults over 40 managing multiple health conditions. Identifying which causes might be contributing is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
?-
Frequently Asked Questions
Do all beta blockers suppress melatonin equally?
Propranolol, which has the highest lipophilicity among commonly prescribed beta blockers, shows the greatest neuropsychiatric risk in meta-analyses. Atenolol, which remains largely peripheral, shows fewer subjective sleep-disruption effects in polysomnography studies. If sleep disruption is a concern, the lipophilicity of the specific beta blocker is a variable worth discussing with a physician.
How much melatonin should someone on a beta blocker take?
The sustained-release formulation is relevant: it more closely mimics the pineal gland’s natural secretion profile, which is a gradual rise rather than a spike. The goal is replacement of suppressed endogenous melatonin, not pharmacological sedation. This distinction matters for dose selection and for explaining why the effective dose is lower than many over-the-counter melatonin products.
Is beta-blocker melatonin suppression reversible?
The reversibility of the melatonin suppression itself is well-supported by the pharmacological mechanism: the drug occupies the receptor, and when the drug is absent, the receptor is available again. The Gehrman and Anafi (2021) case report demonstrated that the pathway can be pharmacologically controlled in both directions — suppressed and then restored — within a single treatment protocol.
?-
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Does Your Circadian Clock Control When Insulin Works? Prediabetes, the Dawn Phenomenon, and Sleep — how circadian insulin timing, glucose rhythm, and dawn physiology affect sleep continuity
- Can Fixing Your Gut Fix Your Sleep? How Gut Bacteria Entrain Your Body Clock — how gut bacteria, the vagus nerve, and microbial rhythms interact with sleep timing
- Can Chronic Inflammation Disrupt Your Circadian Clock Genes? — how cytokines and NF-kB signaling suppress central and peripheral clock genes
- Can Your Body Temperature Keep You From Falling Asleep? — how thermoregulation, vasodilation, and temperature rhythm affect sleep onset and depth
- Why Are You Still Exhausted After Months on CPAP? — how intermittent hypoxia can suppress clock genes even after breathing normalizes
References
1. Cardinali, D. P., & Vacas, M. I. (1987). Cellular and molecular mechanisms controlling melatonin release by mammalian pineal glands. Cellular and Molecular Neurobiology, 7(4), 323–337. https://pubmed.ncbi.nlm.nih.gov/2897878/
2. Rommel, T., & Demisch, L. (1994). Influence of chronic beta-adrenoreceptor blocker treatment on melatonin secretion and sleep quality in patients with essential hypertension. Journal of Neural Transmission — General Section, 95(1), 39–48. https://pubmed.ncbi.nlm.nih.gov/7857585/
3. Betts, T. A., & Alford, C. (1985). Beta-blockers and sleep: A controlled trial. European Journal of Clinical Pharmacology, 28(Suppl), 65–68. https://pubmed.ncbi.nlm.nih.gov/2865152/
4. Scheer, F. A. J. L., Morris, C. J., Garcia, J. I., Smales, C., Kelly, E. E., Marks, J., Malhotra, A., & Shea, S. A. (2012). Repeated melatonin supplementation improves sleep in hypertensive patients treated with beta-blockers: A randomized controlled trial. SLEEP, 35(10), 1395–1402. https://pubmed.ncbi.nlm.nih.gov/23024438/
5. Gehrman, P. R., & Anafi, R. C. (2021). Treatment of a patient with a circadian sleep-wake disorder using a combination of melatonin and metoprolol. Journal of Clinical Sleep Medicine, 17(10), 2121–2124. https://pubmed.ncbi.nlm.nih.gov/34032203/
6. Tikhomirova, O. V., Zybina, N. N., & Kozhevnikova, V. V. (2021). The effect of long-term beta-blockers on melatonin secretion, sleep quality, and vascular brain damage. Zhurnal Nevrologii i Psikhiatrii Imeni S.S. Korsakova, 121(8), 7–12. https://pubmed.ncbi.nlm.nih.gov/34481429/
7. Eddin, L. E., Preyra, R., Ahmadi, F., Jafari, A., Omrani, M. A., & Muanda, F. T. (2025). Beta-blockers and risk of neuropsychiatric disorders: A systematic review and meta-analysis. British Journal of Clinical Pharmacology, 91(2), 325–337. https://pubmed.ncbi.nlm.nih.gov/39658346/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 7 references cited
