If you wake between 3 AM and 5am with blood sugar-driven alertness, the cause may not be what you ate — it may be when your circadian clock signaled your liver to produce glucose. Insulin sensitivity peaks in the morning and falls through the evening. The clock genes that time this rhythm also time your cortisol curve and sleep-wake cycle. When clock precision degrades through late eating, erratic light exposure, or metabolic stress, insulin timing deteriorates alongside everything else the clock coordinates.
This article covers the circadian insulin sensitivity rhythm, the Dawn Phenomenon as a clock-driven event, how meal timing interacts with circadian insulin peaks, and whether clock gene disruption causes prediabetes or the reverse. For the reverse direction — insulin resistance as a cause of poor sleep — see Circadian Sleep Disruption: What Breaks Your Body Clock and Keeps You Awake.
?-
Does Insulin Sensitivity Follow a Circadian Rhythm?
Lucidi et al. (2023) used the hyperinsulinemic-isoglycemic clamp — a gold-standard method for measuring insulin sensitivity — across different times of day in the same individuals. In healthy participants, insulin sensitivity was highest in the morning and lowest in the evening. In participants with early type 2 diabetes, the diurnal pattern was reversed: insulin sensitivity was at its nadir in the morning and higher in the afternoon — opposite to the healthy pattern. This means that one of the earliest changes associated with progression toward type 2 diabetes is not just lower insulin sensitivity overall — it is a reversal of the circadian insulin curve (Lucidi et al., 2023).
The question is whether this rhythm is driven by the circadian clock itself or is a secondary consequence of sleep and behavior patterns. Wefers et al. (2018) addressed this with a rapid-shift protocol — an experiment that separates the effects of sleep from the effects of the internal clock by abruptly shifting the behavioral cycle by 12 hours while keeping participants in dim light to prevent central clock adjustment. Under these conditions, short-term circadian misalignment alone — without sleep deprivation — reduced skeletal muscle insulin sensitivity by impairing nonoxidative glucose disposal. Gene expression analysis showed upregulation of fatty acid metabolism genes in muscle tissue, indicating altered gene expression profiles (Wefers et al., 2018).
This distinction matters. The insulin sensitivity rhythm is not a byproduct of sleeping at night and eating during the day. The clock drives it at the transcriptional level, in skeletal muscle — the primary site of glucose disposal in the body. When the clock is misaligned, the muscle tissue changes which genes it expresses, and glucose handling deteriorates within days.
For adults over 40 — particularly those with fasting glucose in the prediabetic range (100-125 mg/dL) — this circadian rhythm means that glucose tolerance is not the same throughout the day. A meal at 8am and the same meal at 8pm produce different insulin responses, different blood sugar excursions, and different metabolic outcomes. That difference is clock-driven, and it is among the earliest changes observed in the progression toward type 2 diabetes.
?-
What Is the Dawn Phenomenon and Why Is It a Circadian Event?
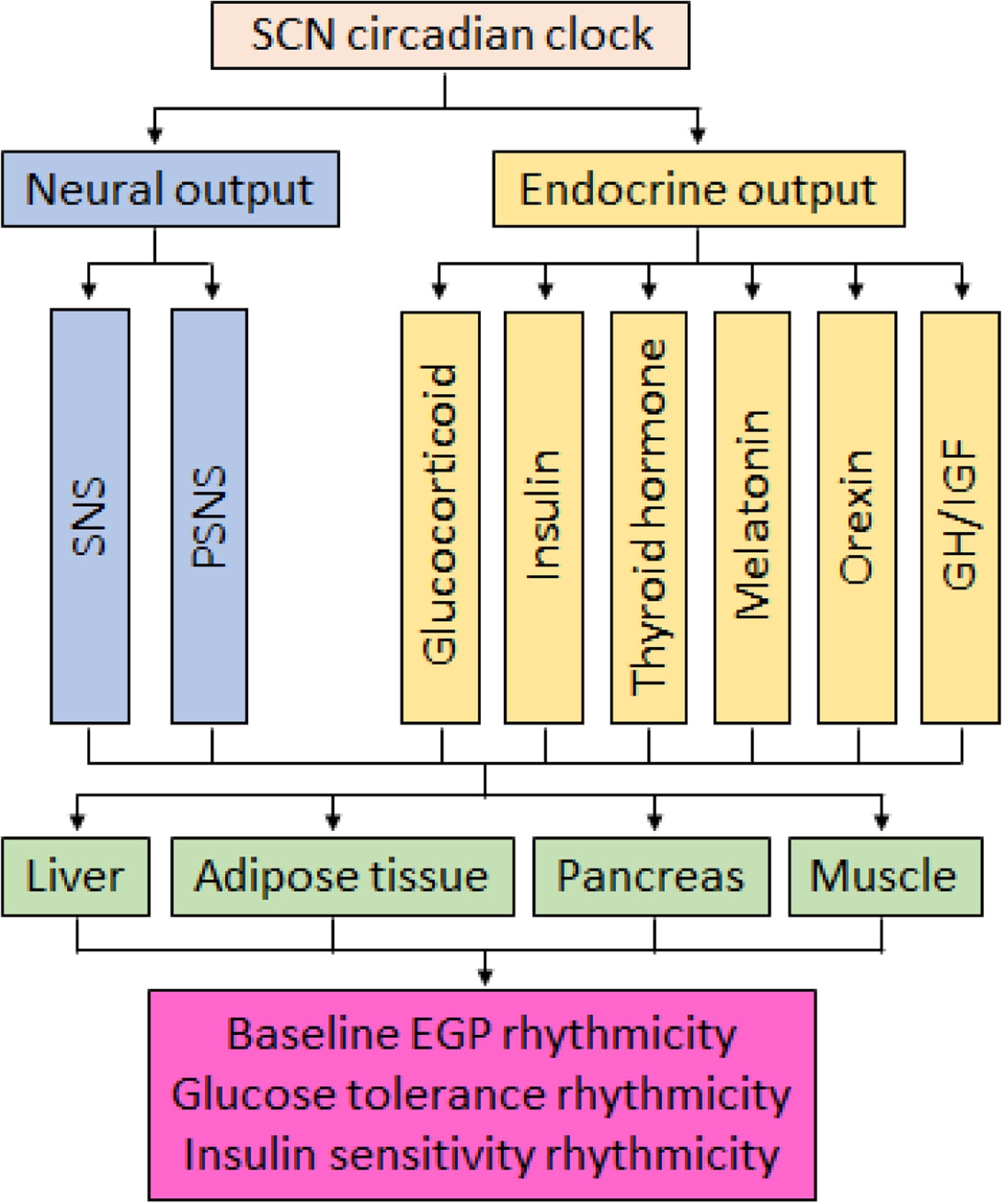
The Dawn Phenomenon is often described as a diabetes management problem — an inconvenient blood sugar spike that complicates morning readings. But Peng et al. (2022) reframed it as a circadian coordination event. The SCN — the master clock in the hypothalamus — orchestrates the Dawn Phenomenon through complementary pathways: autonomic nervous regulation of the liver, and hormonal cascades that converge on hepatic glucose output (Peng et al., 2022).
The sequence is specific. The SCN regulates a growth hormone (GH) rise that occurs shortly after sleep onset. GH promotes hepatic glucose mobilization. Several hours later, the SCN drives a cortisol rise via the hypothalamic-pituitary-adrenal (HPA) axis, peaking before waking. Cortisol sustains hepatic gluconeogenesis — the liver’s production of new glucose from non-carbohydrate substrates. Together, these two hormonal events prepare the body for waking by increasing circulating glucose before the first meal.
In a healthy circadian rhythm, this pre-waking glucose rise is matched by a corresponding rise in insulin sensitivity. The SCN coordinates both: glucose output goes up, and insulin’s ability to manage that glucose also goes up. Speksnijder et al. (2024) detailed the neural and endocrine pathways through which the SCN controls glucose homeostasis — autonomic projections to the liver and pancreas that synchronize insulin release with hepatic glucose production (Speksnijder et al., 2024).
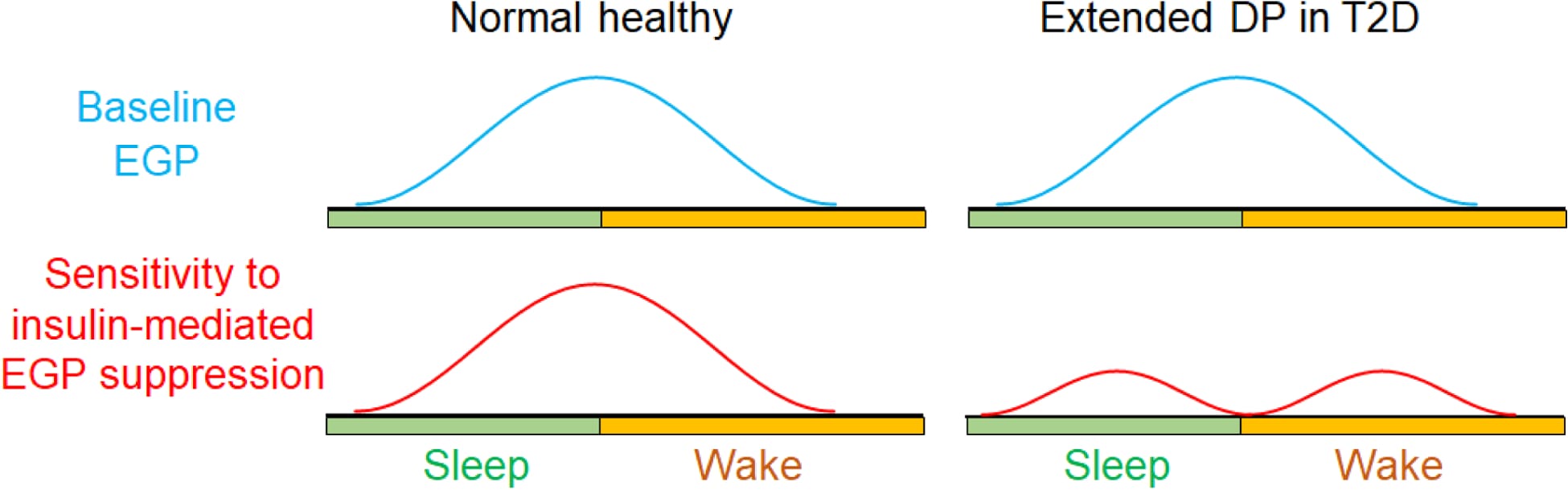
When the circadian clock loses coordination — when the insulin sensitivity rhythm flattens or reverses while the GH and cortisol pulses continue on schedule — the result is unopposed hepatic glucose output during the hours when the body is least equipped to handle it. This is the extended Dawn Phenomenon. The glucose rise is not caused by eating. It is caused by a loss of clock coordination between hormone-driven glucose production and clock-driven insulin sensitivity. This may be one reason many people with prediabetes wake between 3 AM and 5am: the blood sugar rise may be large enough to trigger a counter-regulatory adrenaline response that produces arousal.
?-
Can Time-Restricted Eating Reset Your Circadian Insulin Timing?
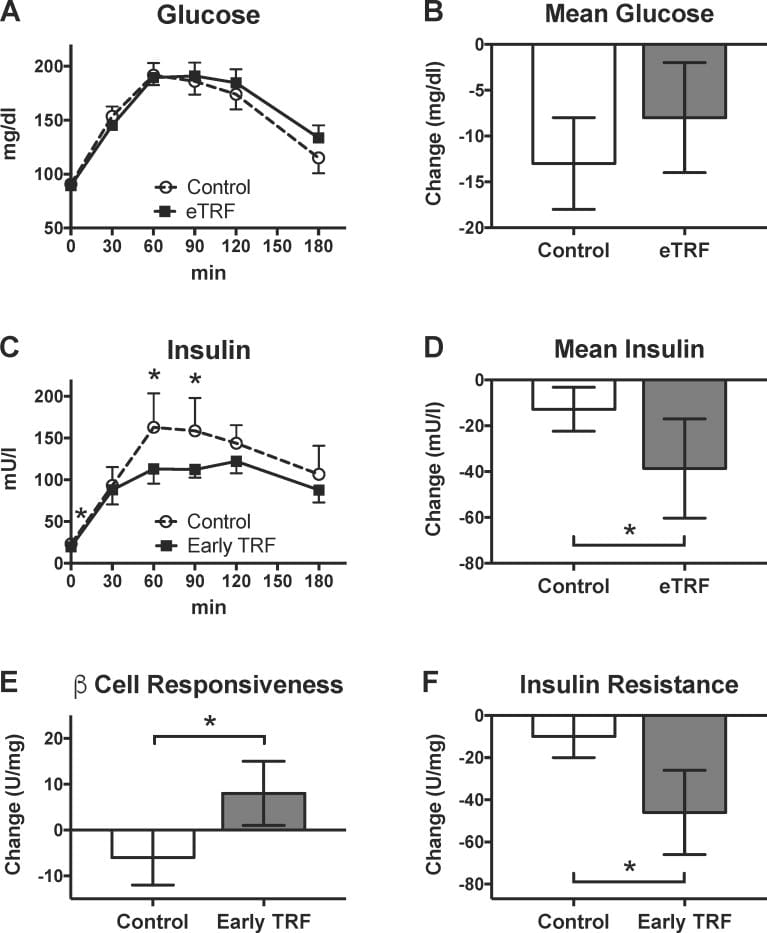
Sutton et al. (2018) conducted the first controlled human feeding trial of early time-restricted eating (eTRE) in men with prediabetes. Participants ate within a 6-hour window (last meal before 3pm) for five weeks, then within a 12-hour window for five weeks, in a crossover design with matched caloric intake. During the early eating window: insulin sensitivity improved, beta-cell responsiveness increased, blood pressure dropped, and oxidative stress markers decreased — all without weight loss (Sutton et al., 2018).
The mechanism is circadian alignment. By placing all food intake during the hours when insulin sensitivity is at its circadian peak (morning through early afternoon), the body processes glucose and lipids with the metabolic machinery that the clock has already primed for nutrient handling. Eating the same food later in the day — when the clock has transitioned the pancreas and liver toward lower insulin sensitivity — requires the body to process nutrients during a phase when glucose disposal capacity is reduced.
Vahlhaus et al. (2025) extended this finding. In a study of 46 twin pairs (92 participants), they found that what matters is not the absolute clock time of eating but eating timing relative to the individual’s own internal clock. Participants who ate later relative to their personal circadian phase had lower insulin sensitivity and higher HOMA-IR, independent of total caloric intake and sleep duration. The association was heritable — eating-timing phenotypes ran in families — suggesting that the link between circadian meal timing and insulin sensitivity has a genetic component (Vahlhaus et al., 2025).
This has a practical consequence. The standard advice to “eat earlier” is incomplete. A person whose circadian clock runs late (a later chronotype) may not benefit from eating at 7am if their internal clock considers that the biological equivalent of 4am. The insulin sensitivity advantage comes from eating during your biological morning — which depends on your individual circadian phase, not on what the clock on the wall says.
?-
Does Clock Gene Disruption Cause Prediabetes — or Does Prediabetes Disrupt the Clock?
Rakshit and Matveyenko (2021) demonstrated the clock-to-diabetes direction with precision. They overexpressed BMAL1 in pancreatic beta cells and observed augmented glucose-stimulated insulin secretion and protection against obesity-induced glucose intolerance. When they used nobiletin — a citrus flavonoid that enhances BMAL1 activity — the same protective effect appeared, and it was BMAL1-dependent: without functional BMAL1, nobiletin had no effect. This positions the circadian clock as a regulator of insulin secretion capacity, not a secondary factor in beta-cell health (Rakshit & Matveyenko, 2021).
Tran et al. (2024) reviewed the diabetes-to-clock direction, detailing how clock disruption is associated with reduced insulin secretion, lower insulin sensitivity, and impaired hepatic glucose homeostasis through the BMAL1/CLOCK transcriptional control of beta-cell genes and CRY-mediated suppression of hepatic gluconeogenesis. A meta-analysis of observational studies cited by Tran et al. found that shift work is associated with approximately 10% higher risk of type 2 diabetes (RR = 1.10) (Tran et al., 2024).
Catalano et al. (2022) added the peripheral clock dimension. When peripheral clocks in the liver, muscle, and adipose tissue desynchronize from the central SCN clock — as happens with irregular sleep schedules, jet lag, or shift work — insulin sensitivity declines in those tissues independently. The liver clock times gluconeogenesis and glycogen storage. The muscle clock times glucose uptake and fatty acid oxidation. The adipose tissue clock times lipid storage and adipokine secretion. When these organ-specific clocks lose synchrony with each other, glucose handling deteriorates across multiple organs at once (Catalano et al., 2022).
The bidirectional nature of this relationship means that prediabetes and circadian disruption can reinforce each other. As the clock degrades, insulin timing worsens — and as insulin timing worsens, metabolic stress feeds back onto the clock. For adults whose blood sugar is trending upward alongside worsening sleep, the two changes may share a common circadian mechanism rather than being independent problems.
?-
What Is Circadian Syndrome?
Metabolic Syndrome is defined by a cluster of five features: abdominal obesity, high triglycerides, low HDL cholesterol, elevated blood pressure, and elevated fasting glucose. Circadian Syndrome adds three more: short or disrupted sleep, reduced physical activity, and depressed mood. The rationale is that these eight features tend to co-occur because they are all regulated by, or responsive to, the circadian clock.
Epidemiological data support these connections. Shift work — a disruption of circadian timing — is associated with elevated rates of type 2 diabetes, cardiovascular disease, mood disorders, and obesity. The Circadian Syndrome framework proposes that addressing the circadian disruption — rather than managing each component separately — may be a more effective approach because the components share upstream circadian machinery.
?-
Circadian insulin timing disruption may compound with hormonal, inflammatory, autonomic, or other metabolic causes of sleep fragmentation. In adults over 40 managing blood sugar, multiple causes often overlap. Identifying which causes might be contributing is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
?-
Frequently Asked Questions
Does Eating Late Raise Blood Sugar?
The Lucidi et al. (2023) isoglycemic clamp data quantified this: insulin sensitivity in healthy individuals was lower in the evening than in the morning. A meal consumed during the low-sensitivity phase requires more insulin to achieve the same glucose disposal — and if the pancreas cannot produce enough additional insulin (as in prediabetes), blood sugar stays elevated longer (Lucidi et al., 2023).
Vahlhaus et al. (2025) showed this is not just about external clock time. The insulin sensitivity penalty from late eating is relative to the individual’s internal circadian phase. A person with a naturally late chronotype who eats at 9pm may experience a smaller effect than an early chronotype eating at the same time — because their biological evening starts later. The practical takeaway: late eating is associated with higher blood sugar, but “late” is defined by your circadian clock, not by the hour on your watch (Vahlhaus et al., 2025).
Can Light at Night Raise Blood Sugar?
The suprachiasmatic nucleus receives light information from melanopsin-containing retinal ganglion cells. This light input synchronizes the master clock to the 24-hour day. When light exposure extends into the biological night — from screens, overhead lighting, or environmental light pollution — it suppresses melatonin and can delay the suprachiasmatic nucleus’s phase. This may shift the hormonal cascade it coordinates, including the cortisol rise timing, the insulin sensitivity rhythm, and hepatic glucose output scheduling (Peng et al., 2022).
Circadian misalignment — as demonstrated by Wefers et al. (2018) in a rapid-shift protocol — can produce skeletal muscle insulin resistance through altered gene expression, independent of sleep loss (Wefers et al., 2018). For someone managing prediabetes, evening light hygiene is a glucose regulation consideration, not only a sleep consideration.
Can Shift Work Cause Prediabetes?
Tran et al. (2024) reviewed the epidemiological and mechanistic data on shift work and type 2 diabetes, citing a meta-analysis (Gao et al., 2020) that found shift work associated with approximately 10% elevated risk (RR = 1.10) (Tran et al., 2024). Catalano et al. (2022) detailed the organ-level mechanism: when peripheral clocks in the liver, muscle, and adipose tissue desynchronize from the central clock — as they do during shift work — insulin sensitivity declines in each tissue independently, and glucose handling deteriorates across multiple organs simultaneously (Catalano et al., 2022).
?-
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Can Fixing Your Gut Fix Your Sleep? How Gut Bacteria Entrain Your Body Clock — how gut bacteria, the vagus nerve, and microbial rhythms interact with sleep timing
- Can Chronic Inflammation Disrupt Your Circadian Clock Genes? — how cytokines and NF-kB signaling suppress central and peripheral clock genes
- Can Your Body Temperature Keep You From Falling Asleep? — how thermoregulation, vasodilation, and temperature rhythm affect sleep onset and depth
- Why Are You Still Exhausted After Months on CPAP? — how intermittent hypoxia can suppress clock genes even after breathing normalizes
- Do Beta Blockers Suppress Melatonin? What Blood Pressure Medication Does to Your Body Clock — how beta blockers, melatonin suppression, and medication timing affect circadian sleep
References
- Catalano, F., De Vito, F., Cassano, V., Fiorentino, T. V., Sciacqua, A., & Hribal, M. L. (2022). Circadian clock desynchronization and insulin resistance. International Journal of Environmental Research and Public Health, 20(1), 29. https://pubmed.ncbi.nlm.nih.gov/36612350/
- Lucidi, P., Perriello, G., Porcellati, F., Pampanelli, S., De Fano, M., Tura, A., Bolli, G. B., & Fanelli, C. G. (2023). Diurnal cycling of insulin sensitivity in type 2 diabetes: Evidence for deviation from physiology at an early stage. Diabetes, 72(10), 1364-1373. https://pubmed.ncbi.nlm.nih.gov/37440717/
- Peng, F., Li, X., Xiao, F., Zhao, R., & Sun, Z. (2022). Circadian clock, diurnal glucose metabolic rhythm, and dawn phenomenon. Trends in Neurosciences, 45(6), 471-482. https://pubmed.ncbi.nlm.nih.gov/35466006/
- Rakshit, K., & Matveyenko, A. V. (2021). Induction of core circadian clock transcription factor Bmal1 enhances β-cell function and protects against obesity-induced glucose intolerance. Diabetes, 70(1), 143-154. https://pubmed.ncbi.nlm.nih.gov/33087455/
- Speksnijder, E. M., Bisschop, P. H., Siegelaar, S. E., Stenvers, D. J., & Kalsbeek, A. (2024). Circadian desynchrony and glucose metabolism. Journal of Pineal Research, 76(4), e12956. https://pubmed.ncbi.nlm.nih.gov/38695262/
- Sutton, E. F., Beyl, R., Early, K. S., Cefalu, W. T., Ravussin, E., & Peterson, C. M. (2018). Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with prediabetes. Cell Metabolism, 27(6), 1212-1221.e3. https://pubmed.ncbi.nlm.nih.gov/29754952/
- Tran, H. T., Kondo, T., Ashry, A., Fu, Y., Okawa, H., Sawangmake, C., & Egusa, H. (2024). Effect of circadian clock disruption on type 2 diabetes. Frontiers in Physiology, 15, 1435848. https://pubmed.ncbi.nlm.nih.gov/39165284/
- Vahlhaus, J., Peters, B., Hornemann, S., Ost, A. C., Kruse, M., Busjahn, A., Pfeiffer, A. F. H., & Pivovarova-Ramich, O. (2025). Later eating timing in relation to an individual internal clock is associated with lower insulin sensitivity and affected by genetic factors. EBioMedicine, 116, 105737. https://pubmed.ncbi.nlm.nih.gov/40305967/
- Wefers, J., van Moorsel, D., Hansen, J., Connell, N. J., Havekes, B., Hoeks, J., van Marken Lichtenbelt, W. D., Duez, H., Phielix, E., Kalsbeek, A., Boekschoten, M. V., Hooiveld, G. J., Hesselink, M. K. C., Kersten, S., Staels, B., Scheer, F. A. J. L., & Schrauwen, P. (2018). Circadian misalignment induces fatty acid metabolism gene profiles and compromises insulin sensitivity in human skeletal muscle. Proceedings of the National Academy of Sciences, 115(30), 7789-7794. https://pubmed.ncbi.nlm.nih.gov/29987027/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 9 references cited
