Lying in bed exhausted, heart rate elevated, mind cycling through the same thoughts — this experience has a name: hyperarousal. And it has a molecular driver.
Hyperarousal is not a vague psychological state. It is a measurable condition in which wake-promoting brain regions remain active during the nighttime hours, when they should be suppressed. The primary molecule sustaining that activity is orexin — a neuropeptide produced by neurons in the lateral hypothalamus that stabilizes wakefulness. Under chronic stress, the corticotropin-releasing hormone (CRH) to orexin circuit keeps those neurons firing when sleep pressure says they should be off.
This article covers the CRH-orexin-cortisol circuit, the neuroimaging evidence for hyperarousal, and what happens when dual orexin receptor antagonists (DORAs) interrupt the loop. Stress-driven orexin activation interacts with circadian timing — for the broader picture of how circadian mechanisms fragment sleep, see the parent pillar on Circadian Rhythm Disruption and Sleep.
How Do Orexin Neurons Connect to the Stress Response?
Winsky-Sommerer et al. (2004) identified the anatomical and functional connection between the stress response and orexin. Using immunohistochemistry in mice, the researchers found CRH-containing axon terminals forming synapses directly on hypocretin (orexin)-producing neurons in the lateral hypothalamus. The contact was verified at the electron microscopy level — CRH boutons made asymmetric synapses (a marker of excitatory input) on orexin cell bodies and dendrites.
The functional consequence: when CRH was applied to orexin neurons in brain slices, it depolarized them in a dose-dependent manner. At 100 nM, CRH increased both membrane potential and firing rate. This depolarization was blocked by astressin, a CRH-R1 antagonist — establishing that the effect runs through CRH receptor 1. In live animals, restraint stress and footshock activated orexin neurons (measured by c-Fos expression), and this activation was reduced in CRH-R1 knockout mice.
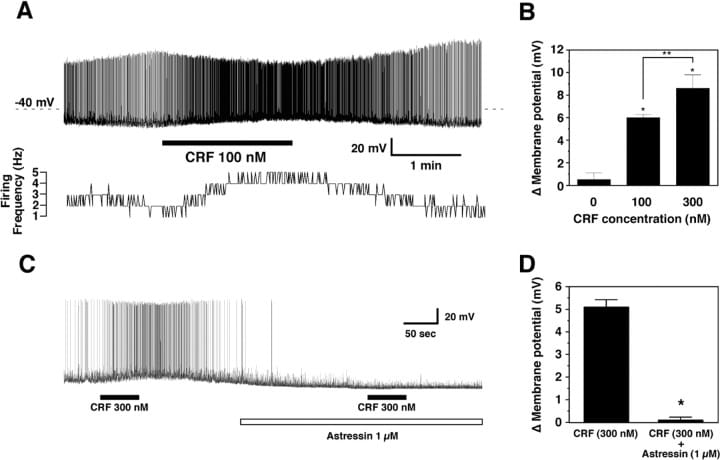
The circuit is bidirectional. Orexin neurons project to the paraventricular nucleus of the hypothalamus (PVN) — the structure that produces CRH and initiates the hypothalamic-pituitary-adrenal (HPA) axis. When orexin activates the PVN, CRH release increases, which stimulates ACTH from the pituitary, which drives cortisol from the adrenal glands. That cortisol and CRH, in turn, can further activate orexin neurons.
Yun et al. (2017) demonstrated that blocking orexin receptor 2 (OX2R) can interrupt this loop. In mice subjected to cage-exchange stress (a social stressor), OX2R knockout animals showed abolished stress-induced ACTH release compared to wild-type controls. The selective OX2R antagonist JNJ-42847922 reproduced this effect pharmacologically — pre-dosing with the antagonist before the stressor blocked the ACTH surge. A dual OX1R/OX2R antagonist (SB-649868) produced the same result.
The implication: orexin actively drives the hormonal stress response through OX2R. The loop runs: stress triggers CRH release in the hypothalamus, CRH depolarizes orexin neurons via CRH-R1, orexin neurons activate the PVN through OX2R, the PVN releases more CRH and drives cortisol production. Under acute stress, this loop resolves when the stressor ends. Under chronic stress, it can become self-sustaining.
What Does Hyperarousal Look Like in the Brain?
Kay and Buysse (2017) reviewed PET (positron emission tomography) and fMRI (functional magnetic resonance imaging) evidence comparing cerebral glucose metabolism in 44 people with primary insomnia and 40 good sleepers. The findings showed a pattern consistent with hyperarousal across both states: during waking, people with insomnia showed altered regional glucose metabolism. During NREM sleep, the same pattern persisted — brain regions involved in conscious awareness that should deactivate during sleep did not fully deactivate.
The researchers proposed a model in which insomnia represents a state of elevated wake drive and elevated conscious awareness that persists into sleep. In healthy sleep, wake-promoting regions reduce their activity, allowing consolidated NREM and REM to proceed. In insomnia, those regions remain partially active — the brain is in a state between full wakefulness and full sleep, without committing to either.
This is what “wired but tired” looks like at the neural level: the brain cannot fully transition into sleep because wake-promoting circuits — including those driven by orexin — remain active. Sleep pressure (adenosine accumulation) is high, producing the sensation of exhaustion. But cortical arousal persists, producing the racing mind and inability to disengage.
Vgontzas et al. (2001) provided the hormonal evidence. The study measured cortisol and ACTH (adrenocorticotropic hormone) levels every 30 minutes across 24 hours in people with insomnia compared to controls. People with insomnia had elevated cortisol and ACTH across the full 24-hour period, with the largest elevations occurring in the evening and early night — the hours when cortisol should be at its circadian nadir (lowest point). People with sleep efficiency below 70% — meaning they spent more than 30% of time in bed awake — had the highest cortisol levels.
This timing matters. Evening and early-night cortisol elevation directly opposes the circadian suppression of orexin. The circadian clock normally reduces orexin neuron firing at night (through the SCN-DMH relay described in the parent article on orexin and circadian wake control). But elevated evening CRH and cortisol provide excitatory input to orexin neurons that competes with the circadian suppression. If the stress-driven excitation is strong enough, it overrides the circadian gate — and orexin neurons fire into the night.
Dressle et al. (2022) conducted a meta-analysis of 20 studies including 449 people with insomnia and 462 controls. The pooled effect for cortisol elevation in insomnia was a standardized mean difference (SMD) of 0.50 — a moderate effect. The analysis also found a dose-response relationship: greater insomnia severity was associated with higher cortisol levels. People whose insomnia included short sleep duration (objectively measured, not just perceived) showed the strongest cortisol elevations, consistent with the hyperarousal phenotype — these individuals had insomnia driven by physiological activation rather than misperception of sleep quality.
The convergence of these findings — elevated brain glucose metabolism, 24-hour cortisol and ACTH elevation peaking in the evening, and a dose-response relationship between insomnia severity and cortisol — maps onto the CRH-orexin loop described above. Chronic stress elevates CRH, CRH activates orexin, orexin drives further cortisol production, and cortisol levels remain elevated during the hours when sleep should be consolidating.
Can Breaking the Orexin-Cortisol Loop Reduce Hyperarousal?
Nakamura and Nagamine (2017) studied 40 people with psychiatric conditions (depression, anxiety, schizophrenia, or bipolar disorder) who had comorbid insomnia. Participants received suvorexant (a dual orexin receptor antagonist) for 8 weeks. By Week 8, cortisol levels had decreased, and scores on standardized measures of sleep quality (Pittsburgh Sleep Quality Index), anxiety (Generalized Anxiety Disorder-7), and depression (Patient Health Questionnaire-9) all improved.
The cortisol reduction is notable because suvorexant blocks orexin receptors — it does not target cortisol or the HPA axis directly. If blocking orexin receptors reduces cortisol, this supports the loop model: orexin drives cortisol production through the PVN, and interrupting orexin receptor activation reduces the downstream hormonal output. The sleep improvement, anxiety reduction, and cortisol decrease all moved together, consistent with a single underlying circuit being modulated.
A caveat: the Nakamura study was observational. There was no placebo control group. Cortisol can decrease with improved sleep from any cause, and psychiatric medications were continued during the study. The cortisol reduction cannot be attributed solely to orexin receptor blockade. Controlled studies isolating suvorexant’s effect on cortisol from its effect on sleep are needed to establish the mechanism.
Prajapati et al. (2026) reviewed the evidence connecting orexin to post-traumatic stress disorder (PTSD). Traumatic stress can chronically activate orexin neurons through sustained CRH elevation — the same CRH-orexin circuit described above, but driven by trauma-related HPA axis activation rather than general life stress. The review noted that CSF orexin-A levels negatively correlate with PTSD severity: people with more severe PTSD tend to have lower CSF orexin-A. This may reflect orexin neuron exhaustion or downregulation after prolonged overactivation — similar to the receptor desensitization seen in other neuropeptide circuits under chronic stimulation.
The PTSD evidence adds a dimension to the stress-orexin relationship. Acute and moderate chronic stress appear to elevate orexin activity, producing the hyperarousal and sleep disruption described above. But severe, prolonged traumatic stress may push past elevation into depletion — a trajectory that could explain why some people with chronic stress-related insomnia initially experience hyperarousal (wired but tired) and later transition to hypersomnia or pervasive fatigue. This trajectory remains a hypothesis and requires longitudinal human studies to validate.
For the broader question of how orexin receptor blockade compares to traditional sedative-hypnotics, see DORAs vs. Ambien: How Orexin-Blocking Sleep Medications Work Differently in Your Brain.
Stress-driven orexin overactivation is one of several causes of sleep fragmentation and difficulty staying asleep. Cortisol timing, melatonin decline, metabolic, and inflammatory factors might also be contributing. Identifying which causes might be active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Is “Wired but Tired” the Same as Anxiety-Driven Insomnia?
Does the Cortisol-Orexin Loop Reset on Its Own?
Can Exercise Reduce Orexin-Driven Hyperarousal?
Related Reading:
- Circadian Rhythm Disruption and Sleep — The full cause reference page covering all five circadian mechanisms
- What Is Orexin and Why Does It Keep You Awake When Your Body Clock Says Sleep?
- How Does Orexin Change With Age? Why Sleep Fragments More After 50
- How Melatonin Suppresses Orexin: The Push-Pull That Decides Whether You Sleep or Stay Awake
- Can You Lower Orexin Naturally? What Controls Your Brain’s Wake Drive
- DORAs vs. Ambien: How Orexin-Blocking Sleep Medications Work Differently in Your Brain
References
Dressle, R. J., Feige, B., Spiegelhalder, K., Schmucker, C., Benz, F., Mey, N. C., & Riemann, D. (2022). HPA axis activity in patients with chronic insomnia: A systematic review and meta-analysis of case-control studies. Sleep Medicine Reviews, 62, 101588. https://pubmed.ncbi.nlm.nih.gov/35091194/
Kay, D. B., & Buysse, D. J. (2017). Hyperarousal and beyond: New insights to the pathophysiology of insomnia disorder through functional neuroimaging studies. Brain Sciences, 7(3), 23. https://pubmed.ncbi.nlm.nih.gov/28241468/
Nakamura, M., & Nagamine, T. (2017). Neuroendocrine, autonomic, and metabolic responses to an orexin antagonist, suvorexant, in psychiatric patients with insomnia. Innovations in Clinical Neuroscience, 14(3-4), 30-37. https://pubmed.ncbi.nlm.nih.gov/28584695/
Prajapati, S. K., Majumdar, S., & Ahmed, S. (2026). Orexin signaling at the interface of sleep disturbance and PTSD. Neuropsychopharmacology. https://pubmed.ncbi.nlm.nih.gov/41957427/
Vgontzas, A. N., Bixler, E. O., Lin, H. M., Prolo, P., Mastorakos, G., Vela-Bueno, A., … & Chrousos, G. P. (2001). Chronic insomnia is associated with nyctohemeral activation of the hypothalamic-pituitary-adrenal axis: clinical implications. Journal of Clinical Endocrinology & Metabolism, 86(8), 3787-3794. https://pubmed.ncbi.nlm.nih.gov/11502812/
Winsky-Sommerer, R., Yamanaka, A., Diano, S., Borok, E., Roberts, A. J., Sakurai, T., … & de Lecea, L. (2004). Interaction between the corticotropin-releasing factor system and hypocretins (orexins): a novel circuit mediating stress response. Journal of Neuroscience, 24(50), 11439-11448. https://pubmed.ncbi.nlm.nih.gov/15601950/
Yun, S., Wennerholm, M., Shelton, J. E., Bonaventure, P., Letavic, M. A., Shireman, B. T., Lovenberg, T. W., & Dugovic, C. (2017). Selective inhibition of orexin-2 receptors prevents stress-induced ACTH release in mice. Frontiers in Behavioral Neuroscience, 11, 83. https://pubmed.ncbi.nlm.nih.gov/28533747/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
