Adults over 40 may have sleep vulnerabilities that make semaglutide feel different than it does in younger adults. Age-related declines in deep sleep, muscle mass, and sex hormones create a changed baseline that GLP-1 drugs can add to. Semaglutide-driven muscle loss may increase sarcopenia risk in older adults who are already losing muscle, especially when lean mass is low. Hormonal changes from menopause and age-related testosterone decline can add sleep-fragmentation pathways that overlap with appetite, weight-loss, and metabolic changes on GLP-1 therapy. The same dose may be experienced differently at 45 than at 30.
Middle-aged and older adults are well represented in semaglutide studies, but age-specific sleep outcomes are still thin. Adults over 40 already experience declining deep sleep, changing body composition, and hormonal changes that independently fragment sleep.
This article covers four ways aging physiology intersects with semaglutide to produce distinct sleep outcomes: body-weight and body-composition context, accelerated muscle loss, hormonal overlay from menopause and age-related testosterone changes, and a slower adaptation timeline. GLP-1 drugs affect sleep through several distinct pathways. This article covers why those pathways may feel different in adults over 40. For the full overview, see the parent article on GLP-1 drugs and sleep.
Why Does Ozempic Affect Sleep Differently After 40
After 40, baseline sleep architecture is often changing. Deep sleep (N3) declines across adulthood, and total sleep efficiency drops progressively. Semaglutide’s sleep-related side effects can land on this changed baseline. Body weight also modifies drug exposure: lower body weight may increase relative exposure, while age-related body-composition changes can affect tolerability even when formal age-based dose adjustment is not required.
Deep sleep declines across adulthood, with sex and individual differences. Sleep efficiency drops. Awakenings increase. This is the changed baseline on which GLP-1 drugs act.
A 2024 review of semaglutide pharmacokinetics found that body weight is a covariate that may affect drug exposure, with lower body weight associated with higher relative exposure (Yang & Yang, 2024). In older adults with sarcopenic obesity – high fat mass, low muscle mass – the same milligram dose may be tolerated differently than in younger adults with higher lean mass.
That review also found no formal dose adjustment is required based on age (Yang & Yang, 2024). But the absence of a required adjustment does not mean the absence of a difference. The pharmacokinetics are similar; the body the drug is acting on is not.
Older adults may have less metabolic reserve to absorb GLP-1-driven caloric restriction. That matters because age-related changes in muscle, mitochondrial function, and NAD+ metabolism can reduce the reserve available for appetite suppression and weight-loss stress.
A 2025 study in Cell Metabolism adds animal evidence for age-specific GLP-1 biology: GLP-1 receptor agonist administration produced anti-aging molecular effects in aged mice, but the same administration in young adult mice did not reproduce the anti-aging pattern (Huang et al., 2025). The drug engages aging-specific biological pathways that are not activated in younger physiology in that mouse model.
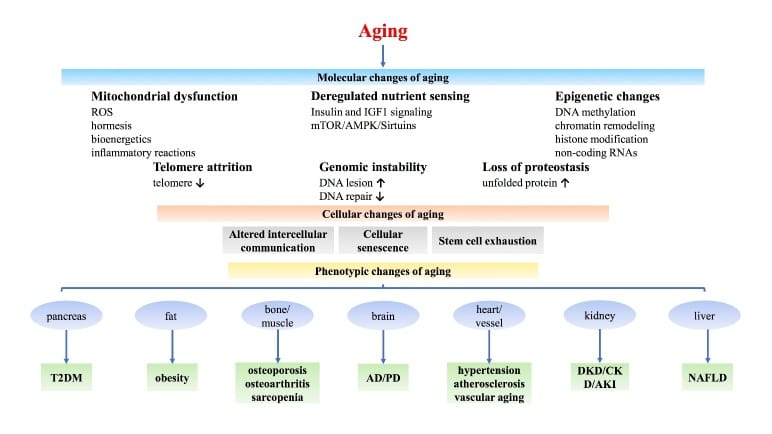
GLP-1 receptor agonists counteract oxidative stress, cellular senescence, and chronic inflammation – three canonical drivers of aging (Peng et al., 2022). Because those pathways change with age, adaptation to GLP-1 side effects may be slower in some older adults, but that remains a working hypothesis rather than a settled trial finding.
Does Ozempic Accelerate Age-Related Muscle Loss That Affects Sleep
A 24-month study of older adults with type 2 diabetes found that semaglutide was associated with disproportionate muscle mass loss and functional decline – grip strength showed sex-specific changes and gait speed diminished (Ren et al., 2025). In adults over 40 who are already moving toward age-related muscle loss, semaglutide-driven weight loss can accelerate the trajectory toward sarcopenia. Reduced muscle mass can lower metabolic reserve and glucose-buffering capacity, two sleep-relevant pathways when appetite is suppressed.
The 24-month retrospective cohort by Ren et al. (2025) enrolled 220 older adults with type 2 diabetes receiving semaglutide and 212 controls. Semaglutide was associated with skeletal muscle mass reduction disproportionate to overall weight loss. Grip strength declined continuously in women, initially improved and later declined in men, while gait speed diminished in both sexes.
Three independent predictors of muscle loss emerged: higher semaglutide dosage, lower baseline muscle mass, and lower baseline gait speed (Ren et al., 2025). The people with the least muscle to spare lose the largest proportion.
A case report illustrates the individual-level impact. A 74-year-old male lost 8 kg on semaglutide, with parallel muscle bulk reduction and progressive fatigue over two years. After other causes were excluded, semaglutide-associated sarcopenia was identified. His condition improved with dose reduction and resistance exercise (Mohamad, 2025).
Muscle loss can matter for sleep because skeletal muscle is metabolically active tissue. Lower muscle mass can reduce resting energy expenditure and leave less reserve during periods of appetite suppression.
Muscle mass also affects glucose buffering. Skeletal muscle is the primary site for glucose disposal. Less muscle means less capacity to absorb blood glucose, which can make overnight metabolic regulation more vulnerable when food intake is low.
Adults over 40 already face age-related declines in testosterone, growth hormone, and IGF-1 – all of which can affect the body’s capacity to rebuild muscle. Semaglutide-driven caloric restriction without concurrent resistance exercise creates conditions where muscle breakdown may exceed the body’s ability to rebuild.
The first prospective trial designed to measure these effects in older adults is underway. Cortes et al. (2024) designed a randomized controlled trial for adults 65 and older, measuring lean mass via DXA alongside aging biomarkers including IL-6, TNF-alpha, and CRP – explicitly acknowledging that the benefits and risks of semaglutide in older adults are “understudied.”
Does Menopause or Andropause Make Ozempic Sleep Side Effects Worse
Menopause and age-related testosterone decline independently can fragment sleep through declining estrogen, progesterone, and testosterone – hormones involved in sleep architecture, temperature control, and arousal thresholds. Semaglutide can add appetite, weight-loss, gastrointestinal, and metabolic pressures on top of this hormonal context. But the relationship is not purely additive: postmenopausal women on hormone therapy lost more weight on semaglutide than those without hormone therapy, suggesting hormonal status may modify response to semaglutide – creating a complex interaction where hormonal status modifies drug efficacy and may also modify side effects.
Menopause sleep disruption is well-characterized. Declining estrogen is associated with insomnia symptoms and sleep-maintenance problems. Progesterone metabolites can support sleep through GABA-A receptor modulation. Vasomotor episodes – hot flashes and night sweats – directly fragment sleep continuity. This is the baseline onto which semaglutide effects are added for women over 40.
A retrospective cohort study compared semaglutide weight loss between postmenopausal women using hormone therapy (n=16) and those not using it (n=90). Women on hormone therapy achieved 16% total body weight loss versus 12% at 12 months (p=0.04), an association that held after multivariate adjustment (Hurtado et al., 2024). The finding suggests a possible pharmacodynamic interaction between hormone status and semaglutide response.
This creates a paradox for menopausal women. More weight loss is potentially better for sleep through reduced sleep apnea and fewer night sweats. But more aggressive caloric restriction is potentially worse through greater fatigue and metabolic disruption. The net sleep outcome depends on which effect dominates.
For men, the interaction differs. A 2025 meta-analysis found GLP-1 receptor agonists were associated with increased bioavailable testosterone in men with obesity-related hypogonadism (Orra et al., 2025). Low testosterone is associated with lower slow-wave sleep and increased nocturnal wakefulness, although body weight explains part of that relationship. GLP-1-driven testosterone recovery could matter for sleep in some men, but direct sleep outcomes were not tested in that testosterone meta-analysis.
The sarcopenia data is sex-differentiated. In the Ren et al. (2025) cohort, women experienced continuous grip strength decline on semaglutide, while men showed initial improvement followed by decline – suggesting sex-specific functional patterns during semaglutide therapy in older adults with type 2 diabetes.
Does Age Affect How Long Ozempic Sleep Side Effects Last
Older adults may adapt more slowly to semaglutide’s sleep-related side effects because aging changes sleep architecture, muscle reserve, mitochondrial function, and metabolic flexibility. NAD+ levels decline with age in human tissue, reducing the cellular energy available for adaptive responses. The same dose-escalation schedule used in younger adults may warrant a more individualized stabilization window when side effects affect sleep.
Adaptation to any new drug requires cellular work. Neural plasticity declines with age. Metabolic compensation capacity is reduced in bodies with lower mitochondrial function.
GLP-1 receptor agonists counteract oxidative stress, cellular senescence, and chronic inflammation – three primary drivers of aging (Peng et al., 2022). Those pathways also change with age, which is one reason adaptation may vary between individuals.
The 2025 Cell Metabolism study adds animal evidence. GLP-1 receptor agonist administration engaged age-specific biological pathways in aged organisms that were not activated in younger physiology (Huang et al., 2025). The drug is doing different things in an older body in that mouse model.
Optimal dosages and long-term safety profiles for aging-specific applications remain undetermined (Chavda et al., 2024). The titration schedules were designed for mixed-age populations and may not account for age-related adaptation differences.
This matters for dose escalation. When sleep disruption appears during dose escalation, the relevant question is whether the next increase is happening before appetite, GI symptoms, energy, and sleep have stabilized. That decision belongs in the medication-management conversation.
There is a counterbalancing benefit. A 2025 meta-analysis found that GLP-1 receptor agonists reduce the apnea-hypopnea index by 16.6 events per hour in adults with obstructive sleep apnea (Kow et al., 2025). For older adults with sleep apnea, this improvement may eventually outweigh the acute sleep disruption. The timing of that tradeoff has not been directly tested in older adults with GLP-1-related sleep complaints.
Age can add vulnerability to several pathways through which GLP-1 drugs may affect sleep – from blood sugar instability to hormonal fragmentation. Metabolic sleep disruption in adults over 40 rarely involves a single mechanism. It overlaps with circadian changes, hormonal decline, and autonomic changes already underway before the first injection.
Find out which causes might be driving your 3am wakeups: https://sleep.thelongevityvault.com/decoderutm_source=website&utm_medium=article&utm_campaign=metabolic-cluster-ozempic-sleep-after-40
Frequently Asked Questions
Should Older Adults Start Ozempic at a Lower Dose to Protect Sleep
Semaglutide labeling does not recommend dose adjustments based on age, and pharmacokinetic data shows the drug behaves predictably across age groups (Yang & Yang, 2024). But the body it acts on is different after 40 – less muscle, less deep sleep, less hormonal resilience. The professional managing the medication may choose slower titration in older adults, extending the interval between dose increases to allow more time for adaptation at each level.
Pharmacokinetics describe how the body handles the drug – not how the drug affects the body. The body at 50 has less deep sleep to lose, less muscle to buffer blood glucose, and less hormonal resilience. Whether slower titration – eight weeks between dose increases instead of four – reduces sleep disruption in older adults has not been tested. It remains a judgment between an individual and their prescriber.
Does Ozempic Interact With Menopause Hormone Therapy for Sleep
No direct pharmacokinetic interaction between semaglutide and estrogen or progesterone has been identified. But the Hurtado et al. (2024) study showed postmenopausal women on hormone therapy lost more weight on semaglutide – suggesting a pharmacodynamic interaction that modifies how the body responds to the drug. For sleep, this creates a two-sided dynamic: more weight loss may improve sleep through sleep apnea reduction, but more aggressive caloric restriction may worsen metabolic sleep disruption.
The interaction is pharmacodynamic – it changes how the body responds – rather than pharmacokinetic (Hurtado et al., 2024). Hormone therapy may support sleep in some women: estrogen reduces vasomotor episodes, and progesterone has sedative effects through GABA receptor modulation. But the stronger weight-loss response may also change appetite, energy intake, and fatigue, and the downstream metabolic effects on sleep may also be amplified.
Is Ozempic Muscle Loss More Dangerous for Sleep After 50
After 50, baseline muscle loss becomes more relevant to function, glucose handling, and resilience during weight loss. Semaglutide-driven weight loss adds pharmacological muscle loss on top of this age-driven decline. The 24-month cohort study found that lower baseline muscle mass predicted greater muscle loss on semaglutide, meaning the people with the least muscle to spare lose the largest proportion (Ren et al., 2025).
Less muscle means less metabolic reserve during appetite suppression and weight loss. After 50, testosterone, growth hormone, and IGF-1 all decrease, reducing the body’s capacity to rebuild muscle. Resistance exercise at this age is one evidence-aligned strategy to preserve the muscle mass that supports sleep quality.
Does Age Affect How Well Sleep Supplements Work on Ozempic
Age changes the biochemistry of sleep supplementation. Magnesium absorption decreases with age. Melatonin production declines, and exogenous melatonin may produce higher levels in older adults at the same dose. Protein intake can also fall during semaglutide-related appetite suppression, which matters because older adults need enough amino acids to preserve lean tissue.
Magnesium is absorbed less efficiently with age. Melatonin supplementation studies in adults 55 and older show dose-dependent increases in circulating melatonin, with higher elevations reported in older adults than younger adults. Glycine supports overnight temperature regulation, but its effects depend on adequate protein status – which semaglutide-driven appetite suppression can compromise. None of these interactions have been studied in older adults taking GLP-1 drugs.
Should People Over 40 Monitor Sleep Quality More Closely on Ozempic
The evidence supports closer monitoring. Adults over 40 have a changed sleep baseline, slower adaptation to drug effects, and higher sarcopenia risk – all of which can manifest as gradual sleep deterioration that is easy to attribute to normal aging. Tracking sleep metrics before and during semaglutide can create a record that informs dose decisions with the professional managing the medication (Cortes et al., 2024).
A practical approach: one to two weeks of sleep tracking before starting semaglutide, measuring total sleep time, wake-after-sleep-onset, and subjective energy. Continue tracking through each dose escalation. Sleep deterioration on semaglutide in older adults may be gradual rather than acute, making a pre-drug comparison point important.
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
References
- Baker, F. C., Lampio, L., Saaresranta, T., & Polo-Kantola, P. (2018). Sleep and sleep disorders in the menopausal transition. Sleep Medicine Clinics, 13(3), 443-456. https://pubmed.ncbi.nlm.nih.gov/30098758/
- Barbagallo, M., Veronese, N., & Dominguez, L. J. (2021). Magnesium in aging, health and diseases. Nutrients, 13(2), 463. https://pubmed.ncbi.nlm.nih.gov/33573164/
- Barrett-Connor, E., Dam, T.-T., Stone, K., Harrison, S. L., Redline, S., Orwoll, E., & Osteoporotic Fractures in Men Study Group. (2008). The association of testosterone levels with overall sleep quality, sleep architecture, and sleep-disordered breathing. The Journal of Clinical Endocrinology & Metabolism, 93(7), 2602-2609. https://pubmed.ncbi.nlm.nih.gov/18413429/
- Chavda, V. P., Balar, P. C., Vaghela, D. A., & Dodiya, P. (2024). Unlocking longevity with GLP-1: A key to turn back the clock Maturitas, 186, 108028. https://pubmed.ncbi.nlm.nih.gov/38815535/
- Cortes, T. M., Vasquez, L., Serra, M. C., Robbins, R., Stepanenko, A., Brown, K., Barrus, H., Campos, A., Espinoza, S. E., & Musi, N. (2024). Effect of semaglutide on physical function, body composition, and biomarkers of aging in older adults with overweight and insulin resistance: Protocol for an open-labeled randomized controlled trial. JMIR Research Protocols, 13, e62667. https://pubmed.ncbi.nlm.nih.gov/39269759/
- Huang, J., Kwok, A. J., Li, J. C. Y., Chiu, C. L. H., Ip, B. Y., Tung, L. Y., Chan, R. C. H., Chan, D. C. W., Wang, Z., Zheng, X., Chow, H. T., Lo, M. P. S., Li, Z., Lin, N., Wang, M., Yan, L. Y. C., Wu, W. K. K., Chow, K. H., Lin, W. J., …, & Ko, H. (2025). Body-wide multi-omic counteraction of aging with GLP-1R agonism. Cell Metabolism, 37(12), 2362-2380.e8. https://pubmed.ncbi.nlm.nih.gov/41265449/
- Hurtado, M. D., Tama, E., Fansa, S., Ghusn, W., Anazco, D., Acosta, A., Faubion, S. S., & Shufelt, C. L. (2024). Weight loss response to semaglutide in postmenopausal women with and without hormone therapy use. Menopause, 31(4), 266-274. https://pubmed.ncbi.nlm.nih.gov/38446869/
- Kow, C. S., Ramachandram, D. S., Hasan, S. S., & Thiruchelvam, K. (2025). Efficacy and safety of GLP-1 receptor agonists in the management of obstructive sleep apnea in individuals without diabetes: A systematic review and meta-analysis of randomized, placebo-controlled trials. Sleep Medicine, 129, 40-44. https://pubmed.ncbi.nlm.nih.gov/39978242/
- Mander, B. A., Winer, J. R., & Walker, M. P. (2017). Sleep and human aging. Neuron, 94(1), 19-36. https://pubmed.ncbi.nlm.nih.gov/28384471/
- Massudi, H., Grant, R., Braidy, N., Guest, J., Farnsworth, B., & Guillemin, G. J. (2012). Age-associated changes in oxidative stress and NAD+ metabolism in human tissue. PLOS ONE, 7(7), e42357. https://pubmed.ncbi.nlm.nih.gov/22848760/
- Mohamad, A. A. (2025). A case report of semaglutide induced sarcopenia: causes of fatigue in older adults. Korean Journal of Family Medicine, 46(4), 288-291. https://pubmed.ncbi.nlm.nih.gov/40223309/
- Nolan, B. J., Liang, B., & Cheung, A. S. (2021). Efficacy of micronized progesterone for sleep: A systematic review and meta-analysis of randomized controlled trial data. The Journal of Clinical Endocrinology & Metabolism, 106(4), 942-951. https://pubmed.ncbi.nlm.nih.gov/33245776/
- Ohayon, M. M., Carskadon, M. A., Guilleminault, C., & Vitiello, M. V. (2004). Meta-analysis of quantitative sleep parameters from childhood to old age in healthy individuals: Developing normative sleep values across the human lifespan. Sleep, 27(7), 1255-1273. https://pubmed.ncbi.nlm.nih.gov/15586779/
- Orra, S. H., Martinez, J. V. N., Porto, B. C., Passerotti, C. C., Sardenberg, R. A. S., & Da Cruz, J. A. S. (2025). Effect of GLP-1 agonists on testosterone levels: A systematic review and meta-analysis. BMC Urology, 25(1), 311. https://pubmed.ncbi.nlm.nih.gov/41291666/
- Peng, W., Zhou, R., Sun, Z. F., Long, J. W., & Gong, Y. Q. (2022). Novel insights into the roles and mechanisms of GLP-1 receptor agonists against aging-related diseases. Aging and Disease, 13(2), 468-490. https://pubmed.ncbi.nlm.nih.gov/35371594/
- Ren, Q., Zhi, L., & Liu, H. (2025). Semaglutide therapy and accelerated sarcopenia in older adults with type 2 diabetes: A 24-month retrospective cohort study. Drug Design, Development and Therapy, 19, 5645-5652. https://pubmed.ncbi.nlm.nih.gov/40631351/
- Vural, E. M. S., van Munster, B. C., & de Rooij, S. E. (2014). Optimal dosages for melatonin supplementation therapy in older adults: A systematic review of current literature. Drugs & Aging, 31(6), 441-451. https://pubmed.ncbi.nlm.nih.gov/24802882/
- Walston, J. D. (2012). Sarcopenia in older adults. Current Opinion in Rheumatology, 24(6), 623-627. https://pubmed.ncbi.nlm.nih.gov/22955023/
- Yang, X. D., & Yang, Y. Y. (2024). Clinical pharmacokinetics of semaglutide: A systematic review. Drug Design, Development and Therapy, 18, 2555-2570. https://pubmed.ncbi.nlm.nih.gov/38952487/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 19 references cited
