Wearable devices now report HRV to millions of users nightly, but most content treats HRV as a generic “recovery” metric. The science reveals something more specific: HRV is a real-time readout of the vagal brake holding systemic inflammation in check. When that brake weakens — especially during sleep — inflammatory markers rise on a predictable timeline.
This article covers the mechanistic pathway from vagal HRV to cytokine suppression, the prospective evidence that today’s HRV predicts tomorrow’s inflammation, the dose-duration threshold for inflammatory elevation, and why insomnia patients show impaired vagal tone at sleep onset. It does not cover HRV and vagal tone for sleep regulation broadly — for the full autonomic framework, see the autonomic sleep disruption pillar.
The vagal anti-inflammatory pathway is one mechanism within the broader autonomic sleep disruption framework. The pillar covers all autonomic causes; this article goes deep on the HRV-inflammation measurement angle.
What Does Heart Rate Variability Actually Measure About the Vagus Nerve and Inflammation?
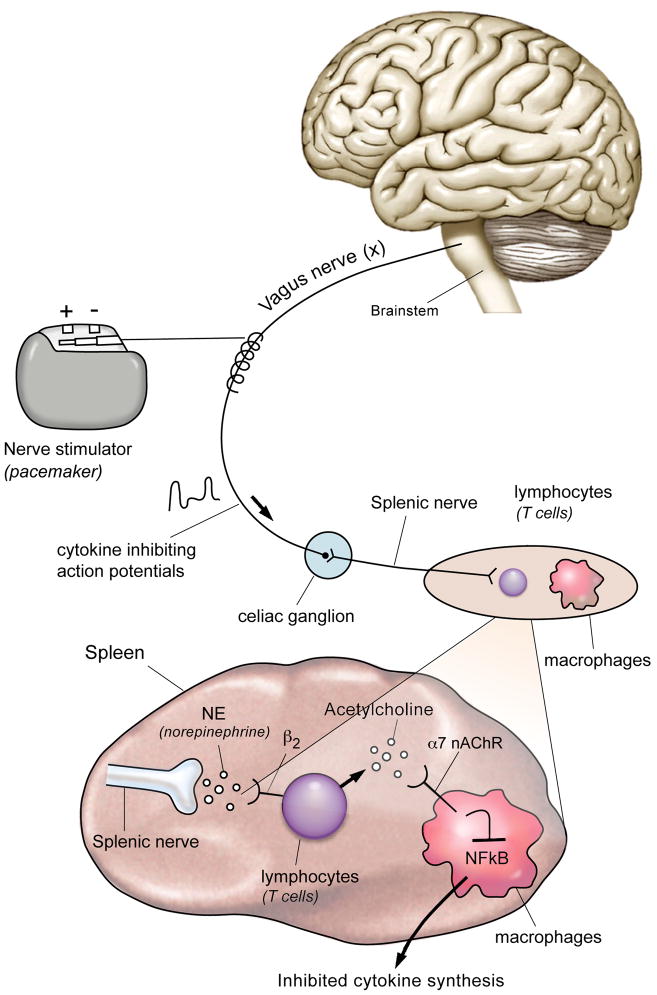
The vagus nerve does not reduce inflammation through a vague “calming” signal. The mechanism is biochemically defined. Action potentials traveling down vagal efferent fibers synapse in the celiac ganglion, which connects to the splenic nerve. The splenic nerve controls lymphocytes in the spleen that produce acetylcholine. That acetylcholine binds to alpha-7 nicotinic acetylcholine receptors (alpha-7 nAChR) expressed on cytokine-producing macrophages. Intracellular signal transduction through the alpha-7 receptor inhibits NF-kB activity, which suppresses transcription and translation of pro-inflammatory cytokines — specifically tumor necrosis factor (TNF), interleukin-1 (IL-1), and interleukin-6 (IL-6) (Huston & Tracey, 2011).
This means HRV is not a generic stress signal. It reflects the output of a defined receptor-mediated anti-inflammatory circuit. When vagal activity is high, acetylcholine release is high, and cytokine production is suppressed. When vagal activity drops — reflected as lower HRV — cytokine suppression weakens.
Williams et al. (2019) screened 2,283 studies and synthesized 51 that provided adequate quantitative data on the HRV-inflammation relationship. The results consistently showed negative associations between HRV indices and inflammatory markers across diverse populations. SDNN (standard deviation of normal-to-normal intervals) and high-frequency HRV (HF-HRV, the 0.15-0.40 Hz band reflecting parasympathetic modulation) demonstrated the strongest and most robust inverse associations with inflammatory biomarkers compared to other HRV measures (Williams et al., 2019).
Sloan et al. (2007) provided one of the largest epidemiological confirmations in 757 subjects from the Coronary Artery Risk Development in Young Adults (CARDIA) study. R-R interval variability was significantly inversely related to IL-6 (HF and LF power b = -0.08 and -0.17, respectively, p < 0.001) and to CRP (HF and LF power b = -0.14 and -0.26, respectively, p < 0.001). These relationships remained statistically significant after full multivariate adjustment for gender, race, age, smoking, physical activity, systolic blood pressure, BMI, and disease status.
Alen et al. (2021) examined a nationally representative sample of midlife adults (N = 738 with complete HRV data) and found that HF-HRV inversely predicted IL-6, CRP, and fibrinogen, while LF-HRV inversely predicted IL-6 and CRP — but showed no meaningful association with adhesion molecules (E-selectin, ICAM-1) or soluble IL-6 receptor. The vagal brake targets specific inflammatory molecules through the alpha-7 nAChR mechanism rather than suppressing inflammation globally. This selectivity confirms the pathway is receptor-mediated, not a nonspecific dampening effect.
Can Heart Rate Variability Today Predict Inflammation Tomorrow?
Most studies linking HRV to inflammation are cross-sectional — they capture both measurements at the same time and cannot determine which came first. Jarczok et al. (2014) addressed this limitation with a 4-year prospective design following 106 nonsmoking employees (mean age 44.4 years; 91% male) at baseline in 2007 and at follow-up in 2011.
At baseline, higher HF-HRV was associated with lower CRP (r = -0.19, p < 0.05). At 4-year follow-up, the same relationship had strengthened substantially (r = -0.34, p < 0.001). After controlling for sex, age, and BMI, baseline HF-HRV remained a significant independent predictor of future CRP levels (partial correlation = -0.20, p < 0.05).
The temporal structure of this study is critical: vagal tone measured at one time point predicts inflammatory burden measured four years later. The relationship did not weaken over time — it strengthened. This pattern positions vagal HRV as an upstream regulator of systemic CRP elevation, not merely a marker that co-occurs with inflammation.
Alen et al. (2021) reinforced the prospective argument with selectivity data. In 738 midlife adults, HF-HRV inversely predicted IL-6, CRP, and fibrinogen, while LF-HRV inversely predicted IL-6 and CRP — molecules directly suppressed by the cholinergic anti-inflammatory pathway. But neither HRV measure predicted adhesion molecules (E-selectin, ICAM-1) or soluble IL-6 receptor.
This selectivity is mechanistically informative. The vagal brake suppresses cytokines produced by macrophages expressing alpha-7 nAChR (TNF, IL-1, IL-6, and downstream acute-phase proteins like CRP and fibrinogen). Adhesion molecules are produced by endothelial cells through different signaling cascades — and the vagal pathway does not modulate those cascades. The selective prediction pattern maps precisely onto what the receptor biology would predict.
Low HRV readings on a wearable device are not just reflecting current sympathetic activation from a stressful day. The prospective data indicate that persistently low vagal HRV signals a trajectory toward elevated inflammatory burden across months and years. Longitudinal HRV tracking — the kind now available through consumer wearables recording nightly — could identify individuals at elevated inflammatory risk before blood markers like CRP and IL-6 reach clinical thresholds.

How Many Nights of Poor Sleep Does It Take to Raise Inflammatory Markers?
Ballesio et al. (2026) conducted the largest meta-analysis to date on sleep deprivation and peripheral inflammation, synthesizing 35 human studies comprising 887 healthy adults. The results identified a clear dose-duration threshold:
- Single night of total or partial sleep deprivation: No statistically significant changes in IL-6 or CRP.
- Three or more consecutive nights of partial sleep deprivation (~4.3 hours per night): Significant elevations in IL-6 (Cohen’s d = 0.42, 95% CI 0.11-0.73, p < 0.01) and CRP (Cohen's d = 0.76, 95% CI 0.09-1.43, p = 0.03).
The 3-night threshold exists because CRP has slow acute-phase kinetics. CRP is synthesized in the liver in response to sustained cytokine signaling — primarily IL-6. A single night of poor sleep may transiently reduce vagal output, but hepatic CRP production requires cumulative IL-6 elevation over multiple days to reach detectable levels. Brief vagal withdrawal recovers before enough cytokine signaling accumulates to trigger measurable CRP synthesis.
Zhang et al. (2025) quantified the autonomic consequences of sleep loss in a meta-analysis of 11 randomized controlled trials involving 549 participants. Sleep deprivation produced:
- Significant decrease in RMSSD (p < 0.05) — the primary time-domain index of vagal activity
- Significant increase in LF/HF ratio (p < 0.05) — indicating a shift toward sympathetic dominance
- Trend toward decreased HF power (not statistically significant)
The combined pattern — reduced RMSSD, elevated LF, elevated LF/HF — represents a clear parasympathetic withdrawal and sympathetic activation signature. Sleep loss directly suppresses the vagal output that maintains the cholinergic anti-inflammatory brake.
The two meta-analyses together complete a four-step causal chain:
1. Poor sleep (Zhang et al., 2025) suppresses vagal HRV (reduced RMSSD, elevated LF/HF)
2. Suppressed vagal HRV means reduced acetylcholine release at splenic macrophages (Huston & Tracey, 2011)
3. Reduced acetylcholine means diminished alpha-7 nAChR activation, permitting cytokine production
4. After three or more consecutive nights (Ballesio et al., 2026), cumulative cytokine signaling produces measurable IL-6 (d = 0.42) and CRP (d = 0.76) elevation
One bad night is recoverable — the vagal brake can reset within a day or two if sleep normalizes. A pattern of short sleep across a week is not recoverable through a single good night. The 3-night threshold marks the point where cumulative vagal withdrawal permits sustained cytokine escalation beyond the body’s compensatory capacity.
Why Do People With Insomnia Show Lower Heart Rate Variability at Sleep Onset?
Ma et al. (2024) compared HRV during the sleep-onset transition in 328 participants across four groups: insomnia without apnea (n = 69), insomnia with mild apnea (n = 70), insomnia with moderate/severe apnea (n = 66), and healthy controls (n = 123).
Insomnia patients exhibited:
- Elevated heart rate compared to controls (p = 0.004)
- Reduced SDNN (p = 0.003)
- Reduced pNN20 (p < 0.001)
- Reduced pNN50 (p = 0.010)
All four measures reflect the same underlying deficit: vagal activity is measurably lower in insomnia patients at the exact moment they transition from wakefulness to sleep. The parasympathetic surge that normally accompanies sleep onset — the vagal brake engaging as arousal systems quiet — is blunted in insomnia.
The study included participants with comorbid sleep apnea at varying severity levels specifically to test whether the HRV impairment was driven by respiratory disruption rather than insomnia. After adjustment for age, gender, BMI, and apnea severity, heart rate elevation, reduced pNN20, and reduced high-frequency power remained significant — confirming vagal impairment independent of respiratory factors. Comorbid moderate-to-severe apnea was associated with further HRV deterioration, but incremental differences between insomnia subgroups did not reach statistical significance. The vagal impairment at sleep onset is a feature of insomnia, not an artifact of body habitus or airway obstruction.
Reduced vagal HRV at sleep onset means diminished cholinergic anti-inflammatory signaling during the overnight period — the hours when vagal tone is normally highest and inflammatory suppression is strongest. The mechanistic chain from poor sleep quality to elevated inflammatory burden now has evidence at every step:
1. Insomnia patients show reduced vagal HRV at sleep onset (Ma et al., 2024)
2. Reduced vagal HRV means less acetylcholine at alpha-7 nAChR on macrophages (Huston & Tracey, 2011)
3. Less receptor activation permits overnight cytokine production
4. Repeated across three or more nights (Ballesio et al., 2026), cumulative cytokine elevation reaches clinical significance
Olivieri et al. (2024) reviewed the evidence positioning age-related HRV decline as a biomarker of the autonomic imbalance that contributes to inflammaging — the chronic low-grade sterile inflammation recognized as a primary risk factor for age-related disease. As sympathetic activity increases and parasympathetic function declines with age, the cholinergic anti-inflammatory brake progressively weakens. This explains a clinical observation: insomnia prevalence and circulating inflammatory markers both increase with age. They are not merely co-occurring age-related changes — they share a common mechanism in progressive vagal withdrawal.
The authors propose sleep-onset HRV as a physiological biomarker of insomnia during bedtime wakefulness. For wearable devices that record HRV continuously through the night, the sleep-onset transition may represent the most informative measurement window for detecting impaired vagal anti-inflammatory function.
Vagal tone is one measurable factor in the inflammation-sleep cycle, but it rarely operates alone. Autonomic hyperarousal, cortisol dysregulation, metabolic disruption, and gut-brain signaling can each amplify the same inflammatory burden that suppresses HRV. When multiple causes overlap, identifying which ones might be active is a useful next step.
Find out which causes might be driving your 3am wakeups ->
Frequently Asked Questions
Which Heart Rate Variability Metric Best Tracks Inflammation?
Can Wearables Detect Inflammation Through Nighttime Heart Rate Variability?
Does Improving Heart Rate Variability Reduce Inflammation?
Does Heart Rate Variability Decline With Age Explain Rising Inflammation?
Is a Single Low Heart Rate Variability Reading Concerning for Inflammation?
Related Reading
- How Does Autonomic Nervous System Dysfunction Disrupt Sleep?
- Why Does Inflammation Cause Insomnia?
- Can Vagus Nerve Stimulation Stop Mast Cell Insomnia?
- How Does Your Vagus Nerve Actively Suppress Inflammation?
- Can Inflammation Damage Your Vagus Nerve?
References
1. Williams, D. P., Koenig, J., Carnevali, L., Sgoifo, A., Jarczok, M. N., Sternberg, E. M., & Thayer, J. F. (2019). Heart rate variability and inflammation: A meta-analysis of human studies. Brain, Behavior, and Immunity, 80, 219-226. https://pubmed.ncbi.nlm.nih.gov/30872091/
2. Huston, J. M., & Tracey, K. J. (2011). The pulse of inflammation: heart rate variability, the cholinergic anti-inflammatory pathway and implications for therapy. Journal of Internal Medicine, 269(1), 45-53. https://pubmed.ncbi.nlm.nih.gov/21158977/
3. Sloan, R. P., McCreath, H., Tracey, K. J., Sidney, S., Liu, K., & Seeman, T. (2007). RR interval variability is inversely related to inflammatory markers: the CARDIA study. Molecular Medicine, 13(3-4), 178-184. https://pubmed.ncbi.nlm.nih.gov/17592552/
4. Alen, N. V., Parenteau, A. M., Sloan, R. P., & Hostinar, C. E. (2021). Heart Rate Variability and Circulating Inflammatory Markers in Midlife. Brain, Behavior, & Immunity – Health, 15, 100273. https://pubmed.ncbi.nlm.nih.gov/34268499/
5. Jarczok, M. N., Koenig, J., Mauss, D., Fischer, J. E., & Thayer, J. F. (2014). Lower heart rate variability predicts increased level of C-reactive protein 4 years later in healthy, nonsmoking adults. Journal of Internal Medicine, 276(6), 667-671. https://pubmed.ncbi.nlm.nih.gov/25141771/
6. Ballesio, A., Fiori, V., & Lombardo, C. (2026). Effects of experimental sleep deprivation on peripheral inflammation: An updated meta-analysis of human studies. Journal of Sleep Research, 35(1), e70099. https://pubmed.ncbi.nlm.nih.gov/40474574/
7. Zhang, S., Niu, X., Ma, J., Wei, X., Zhang, J., & Du, W. (2025). Effects of sleep deprivation on heart rate variability: A systematic review and meta-analysis. Frontiers in Neurology, 16, 1556784. https://pubmed.ncbi.nlm.nih.gov/40895095/
8. Ma, Y., Mullington, J. M., Wayne, P. M., & Yeh, G. Y. (2024). Heart rate variability during sleep onset in patients with insomnia with or without comorbid sleep apnea. Sleep Medicine, 122, 92-98. https://pubmed.ncbi.nlm.nih.gov/39137665/
9. Olivieri, F., Biscetti, L., Pimpini, L., Pelliccioni, G., Sabbatinelli, J., & Giunta, S. (2024). Heart rate variability and autonomic nervous system imbalance: Potential biomarkers and detectable hallmarks of aging and inflammaging. Ageing Research Reviews, 101, 102521. https://pubmed.ncbi.nlm.nih.gov/39341508/
10. Usui, H. (2026). Relationship between very low frequency heart rate variability and interleukin-6 levels in healthy young individuals. Autonomic Neuroscience: Basic & Clinical, 265, 103419. https://pubmed.ncbi.nlm.nih.gov/41967146/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 10 references cited
