Semaglutide delays gastric emptying in a way that can leave more food in the stomach later after a meal. In one small trial of women with PCOS and obesity, semaglutide increased half-emptying time from 118 to 171 minutes, a roughly 45% increase. When you lie down to sleep, that retained gastric volume can make reflux more likely, contributing to nighttime nausea, acid reflux, and repeated awakenings. This is a known gastrointestinal effect of how GLP-1 drugs slow gastric emptying, and it often appears during dose escalation.
Nausea is one of the most common semaglutide side effects. In Ozempic glycemic-control trials, nausea affected about 16-20% of semaglutide-treated participants; in Wegovy weight-management trials, nausea was reported by 44% of semaglutide-treated participants versus 16% with placebo. One sleep-relevant pathway is gastrointestinal: delayed gastric emptying can raise the chance of reflux when you lie down, and nocturnal reflux can fragment sleep.
This article explains the gastrointestinal pathway from delayed gastric emptying to nocturnal reflux to sleep disruption — and how long GI side effects can persist during dose escalation. It does not cover non-GI sleep mechanisms; for those, see the parent overview article on GLP-1 drugs and insomnia.
GLP-1 drugs may affect sleep through several distinct pathways. This article covers the GI and reflux mechanism. Other articles in this series cover the orexin-arousal paradox, blood sugar crashes, and appetite-related sleep chemistry disruption.
Why Does Ozempic Nausea Get Worse When You Lie Down at Night
Semaglutide slows the rate at which your stomach empties food into the small intestine. In one randomized controlled trial in women with PCOS and obesity, gastric content retention at 4 hours was 37% with semaglutide versus zero with placebo, and half-emptying time increased from 118 to 171 minutes — a 45% increase in half-emptying time. When you lie flat with a stomach still holding food, gravity no longer helps keep gastric contents below the lower esophageal sphincter. Intragastric pressure can make reflux more likely, contributing to the burning, nausea, and discomfort that fragments sleep.
Under normal conditions, a solid meal clears the stomach within approximately 4 hours. Semaglutide disrupts this timeline. In a placebo-controlled trial of 20 women with PCOS and obesity, Jensterle et al. (2023) measured gastric emptying after 12 weeks of weekly semaglutide at 1.0 mg. At the 4-hour mark, 37% of gastric contents remained in the semaglutide group versus 0% in the placebo group (p = 0.002). Half-emptying time extended from 118 to 171 minutes. Those gastric-emptying values are the accessible PubMed-verified figures from this abstract-only paper (Jensterle et al., 2023).
When you stand or sit upright, gravity keeps stomach contents below the lower esophageal sphincter. When you lie flat, this gravitational advantage disappears. The retained food mass can become a refluxate reservoir, exerting upward pressure against the sphincter.
Shepherd et al. (2020) reported the temporal mechanics using simultaneous esophageal pH monitoring and polysomnography in 25 participants. They found that 82% of nighttime reflux events occurred during wake periods rather than sleep, and 96% were preceded by brief arousals — meaning waking came first, and reflux followed in this study (Shepherd et al., 2020).
This creates a self-reinforcing loop. Any nighttime awakening — from a full bladder, noise, or natural sleep cycle transitions — may be followed by a reflux event when the stomach still holds food. The resulting burning and nausea prolongs wakefulness, which increases the window for additional reflux events. On semaglutide, delayed emptying can increase the retained meal volume available for reflux, as shown by the 37% four-hour retention finding in Jensterle et al. (2023).
Does Semaglutide’s Effect on Gastric Emptying Cause Nighttime Acid Reflux
Several large studies suggest that GLP-1 receptor agonists increase gastroesophageal reflux disease risk at the population level. A meta-analysis of 55 randomized controlled trials with 106,395 participants found GLP-1 drugs roughly doubled the risk of gastroesophageal reflux disease compared to placebo. A separate population-based cohort study comparing GLP-1 users to SGLT-2 inhibitor users found a 27% elevated reflux disease risk and a 55% elevated risk of reflux disease complications including erosive esophagitis.
Chiang et al. (2025) analyzed 55 randomized controlled trials encompassing 106,395 participants — a large placebo-controlled synthesis. GLP-1 receptor agonist use was associated with a risk ratio of 2.19 for reflux disease (95% CI 1.48-3.25), corresponding to approximately 4 additional cases per 1,000 participants compared to placebo (Chiang et al., 2025).
Noh et al. (2025) compared 24,708 GLP-1 receptor agonist users against 89,096 SGLT-2 inhibitor users, isolating the GLP-1 effect from baseline diabetic reflux risk. Over 3 years of median follow-up, GLP-1 users had a risk ratio of 1.27 for reflux disease (95% CI 1.14-1.42) and 1.55 for complications including erosive esophagitis (95% CI 1.12-2.29) (Noh et al., 2025).
Liu et al. (2024) examined 177,666 propensity-matched people per cohort. Short-acting GLP-1 receptor agonists were associated with erosive reflux disease (HR 1.215), esophageal stricture (HR 1.284), and Barrett’s esophagus (HR 1.372-1.505). In that cohort, long-acting GLP-1 receptor agonists were not associated with increased erosive reflux disease (HR 0.994; 95% CI 0.924-1.069). The complication signal in Liu et al. was concentrated in shorter-acting GLP-1 receptor agonists (Liu et al., 2024).
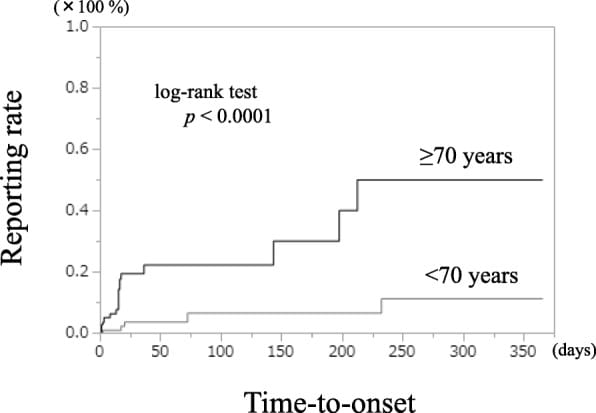
This connection was first detected through Japanese pharmacovigilance data. Noguchi et al. (2018) identified a reporting odds ratio of 5.61 (95% CI 3.95-7.96) for reflux symptoms with GLP-1 receptor agonists, years before Western cohort confirmation. DPP-4 inhibitors showed no comparable signal, supporting a GLP-1 receptor agonist-specific signal in that adverse-event database. Adults over 70 experienced significantly earlier symptom onset (Noguchi et al., 2018).
Quast et al. (2020) measured gastric emptying delay with a gastric-emptying breath test, while reflux was assessed by 24-hour lower-esophageal pH monitoring in 57 participants: +25 minutes with liraglutide and +52 minutes with lixisenatide. Despite the delay, reflux episode counts did not increase significantly over 10 weeks, suggesting measurable reflux disease may manifest over longer treatment durations or in people with other reflux-promoting factors (Quast et al., 2020).
How Long Do Ozempic Gastrointestinal Side Effects Disrupt Sleep
Nausea and GI side effects often occur during dose escalation and may lessen after a stable dose is tolerated. Each increase can bring a new wave of nausea or reflux vulnerability. Ozempic and Wegovy use different maintenance-dose schedules: Ozempic starts at 0.25 mg weekly and may increase to 0.5, 1.0, or 2.0 mg, while Wegovy escalates through 0.25, 0.5, 1.0, and 1.7 mg before maintenance at 1.7 or 2.4 mg. That means GI-mediated sleep disruption can recur across the escalation period.
Why Does Each Dose Increase Reset the Nausea Cycle
Semaglutide is prescribed using a stepped titration schedule: 0.25 mg for the first 4 weeks, then 0.5 mg, then 1.0 mg, and for weight management (Wegovy), continuing to 1.7 mg and 2.4 mg. At each step, the body encounters a higher concentration that can increase the inhibitory effect on gastric motility. The GI tract partially adapts over several weeks, but the next titration step can bring back nausea-related symptoms spanning the escalation period.
How Common Are Gastrointestinal Side Effects in Real-World Users
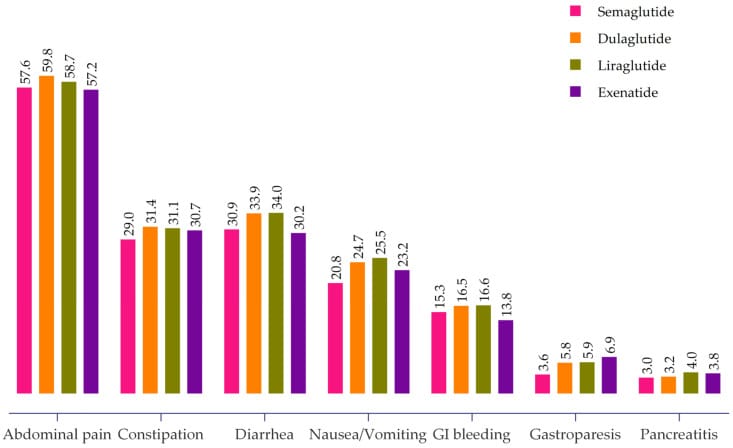
Aldhaleei et al. (2024) analyzed 10,328 GLP-1 receptor agonist users in the NIH All of Us Research Program, providing real-world event rates that complement clinical trial data. Nausea and vomiting occurred in 23.4% of users. Gastroparesis — a more severe form of delayed gastric emptying — occurred in 5.1% of the cohort. Abdominal pain was the most common complaint at 57.6%, followed by diarrhea (32.7%) and constipation (30.4%) (Aldhaleei et al., 2024).
The 5.1% gastroparesis rate is particularly relevant to nighttime reflux. Gastroparesis represents a degree of motility impairment that can keep gastric contents present for longer after meals. Persistent nausea, vomiting, early fullness, or reflux on semaglutide deserves medical evaluation rather than assuming it will fade on the usual escalation timeline.
How Does Semaglutide Compare to Other Weight Loss Drugs for Nausea
Not all GLP-1 receptor agonists carry equal nausea risk. Ismaiel et al. (2025) conducted a network meta-analysis of 39 studies with 33,354 non-diabetic participants and ranked agents by nausea probability: orforglipron carried the highest risk, followed by exenatide, tirzepatide, semaglutide, and liraglutide. Semaglutide falls in the middle of the class for nausea (Ismaiel et al., 2025).
The same analysis identified that semaglutide was specifically associated with constipation — a finding with possible reflux implications. Delayed colon transit can add abdominal pressure in some people, which may matter when delayed gastric emptying is already increasing retained stomach volume.
Can Changing When You Eat on Ozempic Reduce Nighttime Nausea
Because one core problem is retained food in the stomach at bedtime, the most direct step is ensuring the stomach is as empty as possible before lying down. A minimum 3-hour gap between your last meal and bedtime gives the slowed digestive system additional time to clear gastric contents. Smaller, more frequent meals reduce peak gastric volume at any given time. Raising the head of the bed by 6-8 inches uses gravity to keep refluxate below the esophageal sphincter.
Why Does a 3-Hour Pre-Bed Gap Reduce Nighttime Nausea
If semaglutide extends the half-emptying time to approximately 170 minutes, a 3-hour gap is the floor, not the ceiling. At 3 hours, roughly half the meal may still be present. A 4-5 hour gap allows more complete clearance (Jensterle et al., 2023). In practical terms, this means finishing dinner by 6:00 or 7:00 PM for a 10:00 PM bedtime.
Do Smaller More Frequent Meals Reduce Reflux Risk at Night
Reducing meal size lowers peak gastric distension. A distended stomach exerts more pressure against the lower esophageal sphincter, increasing the probability of transient sphincter relaxation — a primary mechanism through which reflux occurs. Three smaller meals with small snacks between them can produce lower peak volumes than two or three large meals, even if total caloric intake remains the same. This aligns with the appetite suppression many semaglutide users already experience.
Does Raising the Head of the Bed Reduce Nighttime Reflux on Ozempic
Raising the head of the bed by 6-8 inches keeps stomach contents below the esophageal sphincter even in a reclined position. Use bed risers under the headboard legs or a dedicated wedge pillow. Stacking regular pillows bends the neck without raising the torso enough — the entire upper body needs to be on an incline. Left-side sleeping may also reduce reflux exposure because of the anatomical position of the stomach relative to the esophagus.
Should You Adjust Eating on Ozempic Injection Day
Semaglutide reaches peak plasma concentration 1-3 days after injection, but I found no trial showing that GI side effects peak at a specific time of day or that injection-day meal timing prevents nighttime nausea. If you notice reflux or nausea clustering in the day or two after a dose, lighter evening meals during that personal high-risk window may reduce the gastric volume available for reflux.
Why Don’t Antacids Fix the Root Cause of Nighttime Nausea on Ozempic
Antacids neutralize stomach acid but do nothing about retained food volume. Proton pump inhibitors reduce acid production but do not improve gastric motility. These medications may reduce the burning sensation but do not address the mechanical cause of nighttime nausea on semaglutide.
Can Sleep Apnea Improvement Partially Offset Reflux-Related Sleep Loss
Li et al. (2025) found that GLP-1 receptor agonists reduce the apnea-hypopnea index by approximately 9.5 events per hour alongside weight loss. For people with both obstructive sleep apnea and reflux-related disruption, the sleep apnea improvement may partially offset reflux-mediated fragmentation. The net effect on sleep quality depends on which condition is more severe in a given person (Li et al., 2025).
Nighttime nausea and acid reflux are one of several gastrointestinal pathways through which metabolic changes can disrupt sleep. GLP-1 drugs also affect blood sugar stability, appetite-related neurotransmitter production, and the brain’s wakefulness circuits — each of which may compound the GI-mediated disruption. Sleep fragmentation from metabolic causes often overlaps with hormonal, circadian, or autonomic factors.
Find out which causes might be driving your 3am wakeups.
Frequently Asked Questions
Does Ozempic Cause Acid Reflux That Wakes You Up at Night
It can. A meta-analysis of 55 randomized controlled trials found that GLP-1 receptor agonists roughly double the risk of gastroesophageal reflux disease compared to placebo (RR 2.19; 95% CI 1.48-3.25). Nocturnal reflux can disrupt sleep because lying flat removes the gravitational barrier that keeps stomach acid below the esophageal sphincter. In one sleep-lab study, 96% of nighttime reflux events were preceded by brief arousals, creating a cycle where any waking may increase reflux risk when gastric volume is elevated (Chiang et al., 2025; Shepherd et al., 2020).
Any arousal — from noise, a sleep stage change, or a bathroom trip — can be followed by reflux. With a stomach still partially full, that relaxation allows acid to move upward. The resulting discomfort prolongs wakefulness and increases the window for further reflux events.
Can Taking Ozempic in the Morning Reduce Nighttime Nausea
Semaglutide is injected once weekly, and peak plasma concentration occurs 1-3 days post-injection. Ozempic can be administered at any time of day, with or without meals. I found no clinical trial showing that morning injection reduces nighttime nausea. Because the pharmacokinetic peak occurs over days rather than hours, meal timing, dose-escalation tolerance, and the timing of personal side-effect patterns are more relevant than assuming morning dosing changes the sleep window (Ozempic Prescribing Information, 2026).
Injection timing affects the calendar timing of the weekly dose, but it does not remove the delayed gastric-emptying mechanism.
Does Ozempic Nausea Improve Over Time or Stay Constant
GI adverse reactions are commonly reported during dose escalation and may lessen once a stable dose is tolerated. Each dose increase can bring symptoms back. In real-world All of Us data, gastroparesis was recorded in 5.1% of GLP-1 receptor agonist users, which makes persistent nausea, vomiting, early fullness, or reflux worth discussing with a qualified medical professional. Semaglutide carries intermediate nausea risk within the GLP-1 class, ranking below orforglipron and exenatide but above liraglutide (Ismaiel et al., 2025; Aldhaleei et al., 2024).
Improvement is not linear. A person may feel better after 6-8 weeks at 0.5 mg, only to experience a return of nausea upon stepping up to 1.0 mg. Persistent nausea beyond 12 weeks at a stable dose warrants medical evaluation for gastroparesis or another motility disorder.
Should You Raise Your Head When Sleeping on Ozempic
Raising the head of the bed by 6-8 inches is a standard reflux disease management strategy, and it may be especially relevant on semaglutide because the pharmacologically increased gastric volume creates a larger refluxate reservoir. The incline uses gravity to keep stomach contents below the lower esophageal sphincter. This is achieved with bed frame risers or a wedge pillow — stacking regular pillows typically bends the neck without raising the torso enough to provide the gravitational benefit (Katz et al., 2022; Schuitenmaker et al., 2022).
Bed risers tilt the entire sleeping surface. Wedge pillows raise the upper torso on a gradual incline. Either works. Stacking standard pillows does not — the esophagus needs to be on a downward slope relative to the stomach. Left-side sleeping may also help, as it positions the sphincter above the gastric contents.
Does Mounjaro Cause Less Nighttime Nausea Than Ozempic
In a network meta-analysis of 39 studies with 33,354 non-diabetic participants, tirzepatide (Mounjaro/Zepbound) and semaglutide (Ozempic/Wegovy) showed comparable nausea risk, with both ranking in the middle of the GLP-1 class. Neither agent has been studied head-to-head specifically for nighttime nausea. The gastric emptying delay mechanism can occur with GLP-1 activity, meaning the nocturnal reflux pathway may apply to both drugs even when overall nausea rates differ (Ismaiel et al., 2025).
Tirzepatide’s dual GIP/GLP-1 mechanism does not change the gastric emptying pathway. Both drugs slow gastric motility through GLP-1 receptor activation, producing the same retained-food-volume problem at bedtime.
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Aldhaleei, W. A., Abegaz, T. M., & Bhagavathula, A. S. (2024). Glucagon-like Peptide-1 Receptor Agonists Associated Gastrointestinal Adverse Events: A Cross-Sectional Analysis of the National Institutes of Health All of Us Cohort. Pharmaceuticals (Basel, Switzerland), 17(2), 199. https://pubmed.ncbi.nlm.nih.gov/38399414/
- Bertin, L., Savarino, V., Marabotto, E., Ghisa, M., de Bortoli, N., & Savarino, E. V. (2026). Pathophysiology of Gastroesophageal Reflux Disease. Digestion, 107(2), 185-201. https://pubmed.ncbi.nlm.nih.gov/40562014/
- Chiang, C. H., Jaroenlapnopparat, A., Colak, S. C., Yu, C. C., Xanthavanij, N., Wang, T. H., See, X. Y., Lo, S. W., Ko, A., Chang, Y. C., Song, J., Hsia, Y. P., & Chiang, C. H. (2025). Glucagon-Like Peptide-1 Receptor Agonists and Gastrointestinal Adverse Events: A Systematic Review and Meta-Analysis. Gastroenterology, 169(6), 1268-1281. https://pubmed.ncbi.nlm.nih.gov/40499738/
- Ismaiel, A., Scarlata, G. G. M., Boitos, I., Leucuta, D. C., Popa, S. L., Al Srouji, N., Abenavoli, L., & Dumitrascu, D. L. (2025). Gastrointestinal adverse events associated with GLP-1 RA in non-diabetic patients with overweight or obesity: a systematic review and network meta-analysis. International Journal of Obesity, 49(10), 1946-1957. https://pubmed.ncbi.nlm.nih.gov/40804463/
- Jensterle, M., Ferjan, S., Ležaič, L., Sočan, A., Goričar, K., Zaletel, K., & Janez, A. (2023). Semaglutide delays 4-hour gastric emptying in women with polycystic ovary syndrome and obesity. Diabetes, Obesity & Metabolism, 25(4), 975-984. https://pubmed.ncbi.nlm.nih.gov/36511825/
- Jung, H. K., Choung, R. S., & Talley, N. J. (2010). Gastroesophageal reflux disease and sleep disorders: evidence for a causal link and therapeutic implications. Journal of Neurogastroenterology and Motility, 16(1), 22-29. https://pubmed.ncbi.nlm.nih.gov/20535322/
- Katz, P. O., Dunbar, K. B., Schnoll-Sussman, F. H., Greer, K. B., Yadlapati, R., & Spechler, S. J. (2022). ACG Clinical Guideline for the Diagnosis and Management of Gastroesophageal Reflux Disease. The American Journal of Gastroenterology, 117(1), 27-56. https://pubmed.ncbi.nlm.nih.gov/34807007/
- Li, M., Lin, H., Yang, Q., Zhang, X., Zhou, Q., Shi, J., & Ge, F. (2025). Glucagon-like peptide-1 receptor agonists for the treatment of obstructive sleep apnea: a meta-analysis. Sleep, 48(4), zsae280. https://pubmed.ncbi.nlm.nih.gov/39626095/
- Liu, B. D., Udemba, S. C., Liang, K., Tarabichi, Y., Hill, H., Fass, R., & Song, G. (2024). Shorter-acting glucagon-like peptide-1 receptor agonists are associated with increased development of gastro-oesophageal reflux disease and its complications in patients with type 2 diabetes mellitus: a population-level retrospective matched cohort study. Gut, 73(2), 246-254. https://pubmed.ncbi.nlm.nih.gov/37739778/
- Noguchi, Y., Katsuno, H., Ueno, A., Otsubo, M., Yoshida, A., Kanematsu, Y., Sugita, I., Esaki, H., Tachi, T., Tsuchiya, T., & Teramachi, H. (2018). Signals of gastroesophageal reflux disease caused by incretin-based drugs: a disproportionality analysis using the Japanese adverse drug event report database. Journal of Pharmaceutical Health Care and Sciences, 4, 15. https://pubmed.ncbi.nlm.nih.gov/29946474/
- Noh, Y., Yin, H., Yu, O. H. Y., Bitton, A., & Azoulay, L. (2025). Glucagon-Like Peptide-1 Receptor Agonists and Risk for Gastroesophageal Reflux Disease in Patients With Type 2 Diabetes: A Population-Based Cohort Study. Annals of Internal Medicine, 178(9), 1268-1278. https://pubmed.ncbi.nlm.nih.gov/40658955/
- Novo Nordisk. (2026). Ozempic (semaglutide) injection prescribing information (Revised May 2026). https://www.novo-pi.com/ozempic.pdf
- Quast, D. R., Schenker, N., Menge, B. A., Nauck, M. A., Kapitza, C., & Meier, J. J. (2020). Effects of Lixisenatide Versus Liraglutide (Short- and Long-Acting GLP-1 Receptor Agonists) on Esophageal and Gastric Function in Patients With Type 2 Diabetes. Diabetes Care, 43(9), 2137-2145. https://pubmed.ncbi.nlm.nih.gov/32647054/
- Schuitenmaker, J. M., Kuipers, T., Smout, A. J. P. M., Fockens, P., & Bredenoord, A. J. (2022). Systematic review: Clinical effectiveness of interventions for the treatment of nocturnal gastroesophageal reflux. Neurogastroenterology and Motility, 34(12), e14385. https://pubmed.ncbi.nlm.nih.gov/35445777/
- Shepherd, K., Ockelford, J., Ganasan, V., Holloway, R., Hillman, D., & Eastwood, P. (2020). Temporal Relationship Between Night-Time Gastroesophageal Reflux Events and Arousals From Sleep. The American Journal of Gastroenterology, 115(5), 697-705. https://pubmed.ncbi.nlm.nih.gov/32356952/
- Simadibrata, D. M., Lesmana, E., Amangku, B. R., Wardoyo, M. P., & Simadibrata, M. (2023). Left lateral decubitus sleeping position is associated with improved gastroesophageal reflux disease symptoms: A systematic review and meta-analysis. World Journal of Clinical Cases, 11(30), 7329-7336. https://pubmed.ncbi.nlm.nih.gov/37969463/
- U.S. National Library of Medicine. (2026). DailyMed: WEGOVY (semaglutide) injection and tablets label. https://dailymed.nlm.nih.gov/dailymed/lookup.cfmsetid=ee06186f-2aa3-4990-a760-757579d8f77b&version=16
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 17 references cited
