Restless legs syndrome affects roughly 5-10% of the general population, and obesity increases the risk by 44% according to a 197,204-participant meta-analysis (Lin et al., 2018). Pharmacovigilance databases contain adverse event reports for restless legs on GLP-1 drugs, but no randomized controlled trial has assessed this outcome. A 2026 retrospective cohort did include restless legs syndrome as a secondary outcome in GLP-1 receptor agonist users and did not find a statistically significant hazard association at 1 or 5 years.
This article explains three pathways through which semaglutide could contribute to or worsen restless legs – iron depletion, B12 deficiency, and dopaminergic modulation – and what to ask about checking if restless legs develop. It does not cover all GLP-1 sleep effects; for the full overview, see the parent article on Ozempic and insomnia.
GLP-1 drugs may affect sleep through several distinct pathways. This article covers the restless-legs-specific mechanisms. Other articles in this series cover orexin activation, blood sugar crashes, vivid dreams, and muscle-loss-related sleep disruption.
Is Restless Legs Syndrome a Side Effect of Ozempic
The FDA Adverse Event Reporting System contains reports of restless legs syndrome associated with semaglutide and liraglutide use. An openFDA query on 2026-05-16 returned 82 semaglutide/RLS reports, including 60 Ozempic reports and 12 Wegovy reports; a separate liraglutide query returned 6 reports. These databases capture voluntary reports from individuals and healthcare providers, which means they document associations but cannot prove causation. Reporting bias, confounding variables, and the absence of denominator data all limit what adverse event databases can demonstrate.
What makes the association worth examining is the epidemiological context. Lin et al. (2018) conducted a meta-analysis of 15 observational studies encompassing 197,204 participants and found that obesity increases restless legs risk: pooled odds ratio 1.44 (95% CI 1.31-1.58). A risk gradient appeared across BMI categories. Sex-stratified analyses found women with obesity at higher relative risk (odds ratio 1.42) than men (odds ratio 1.19).
This creates a useful hypothesis: if obesity is a risk factor for restless legs, then weight loss on semaglutide might eventually reduce the obesity-associated component of restless legs risk – but the rapid weight loss process itself may transiently worsen restless legs if reduced intake or gastrointestinal effects lower iron or B12 status.
Can Iron or B12 Deficiency From Ozempic Contribute to Restless Legs
The iron-dopamine mechanism. Brain iron deficiency in the substantia nigra and thalamus is linked to altered dopaminergic biology and may contribute to the evening pattern of restless legs. Khan et al. (2017) synthesized neuroimaging, cerebrospinal fluid, and postmortem studies demonstrating decreased iron concentrations in these sensorimotor brain regions among people with restless legs. Allen (2004) described the iron-dopamine framework: iron serves as an essential cofactor for tyrosine hydroxylase, meaning brain iron insufficiency may constrain the biochemical capacity for dopamine production.
Iron depletion on semaglutide. Reduced food intake from appetite suppression can reduce dietary iron intake. Gastrointestinal side effects – nausea, vomiting, diarrhea – can make consistent intake harder. The combination may create vulnerability for low iron status in people who are eating substantially less or tolerating fewer iron-rich foods during GLP-1 therapy.
The B12 deficiency pathway. Geng et al. (2022) compared 80 restless legs participants to 80 matched controls. Restless legs participants had lower serum B12 concentrations, and multivariate logistic regression confirmed B12 as independently associated with RLS presence after controlling for confounders. B12 levels correlated inversely with severity – lower B12 corresponded to more severe presentations. B12 is required for myelin synthesis and the homocysteine-to-methionine conversion; deficiency may disrupt peripheral nerve function, amplifying the sensorimotor features of restless legs.
Iron supplementation has randomized-trial evidence. A 2025 meta-analysis by Mahmoud et al. – 12 randomized controlled trials, 511 participants – confirmed that iron supplementation produces improvement in restless legs severity scores, quality of life, and sleep quality. The Cochrane meta-analysis by Trotti and Becker (2019) quantified the benefit across 428 participants: iron therapy improved International Restless Legs Scale scores by a mean difference of -3.78 points versus placebo (95% CI -6.25 to -1.31).

People eating substantially less during GLP-1 therapy may be vulnerable to lower iron and B12 intake, especially if they already carry obesity- or metabolic-disease-related RLS risk. The 2026 cohort data add one relevant signal: GLP-1 receptor agonist use was associated with higher iron-deficiency hazard at 5 years, while the RLS hazard itself was not statistically significant.
Does Semaglutide Affect the Dopamine Pathways Linked to Restless Legs
GLP-1 receptors are present in dopamine-related regions including the ventral tegmental area and nucleus accumbens, and animal work has described GLP-1 receptor effects in substantia nigra dopaminergic neurons. Badulescu et al. (2024) conducted a review showing that GLP-1 receptor agonists reduce reward-related neural activation and may modulate dopaminergic signaling in humans. Neuroimaging data demonstrated decreased activation in reward regions, consistent with modulation of mesolimbic dopamine circuitry rather than peripheral satiety effects alone.
The D2/D3 receptor question. Restless legs has historically been treated with dopamine agonists targeting D2 and D3 receptors. GLP-1 receptor modulation raises a narrower question: whether GLP-1 receptor agonists change RLS-relevant dopamine signaling at all. That question remains untested.
Circadian dopamine dysregulation. Restless legs peak in the evening because dopaminergic tone follows a circadian pattern: high during the day, declining at night. A semaglutide-specific effect on this evening RLS pattern has not been demonstrated. Anguizola et al. (2025) reviewed brain iron deficiency alongside dopaminergic, glutamatergic, and adenosinergic pathways in RLS pathophysiology.
The augmentation concern. The 2025 American Academy of Sleep Medicine guideline (Winkelman et al., 2025) cautions against chronic dopamine agonist therapy for restless legs due to augmentation risk – a paradoxical worsening that develops with long-term dopamine receptor stimulation. This underscores that chronic dopaminergic stimulation can worsen RLS over time in some people.
No study has directly measured semaglutide’s effect on the sensorimotor dopamine pathways implicated in restless legs. The evidence is mechanistic inference from receptor distribution and reward circuit data, not direct proof – but the biological plausibility supports asking about ferritin, transferrin saturation, and B12 when restless legs appear during GLP-1 therapy.
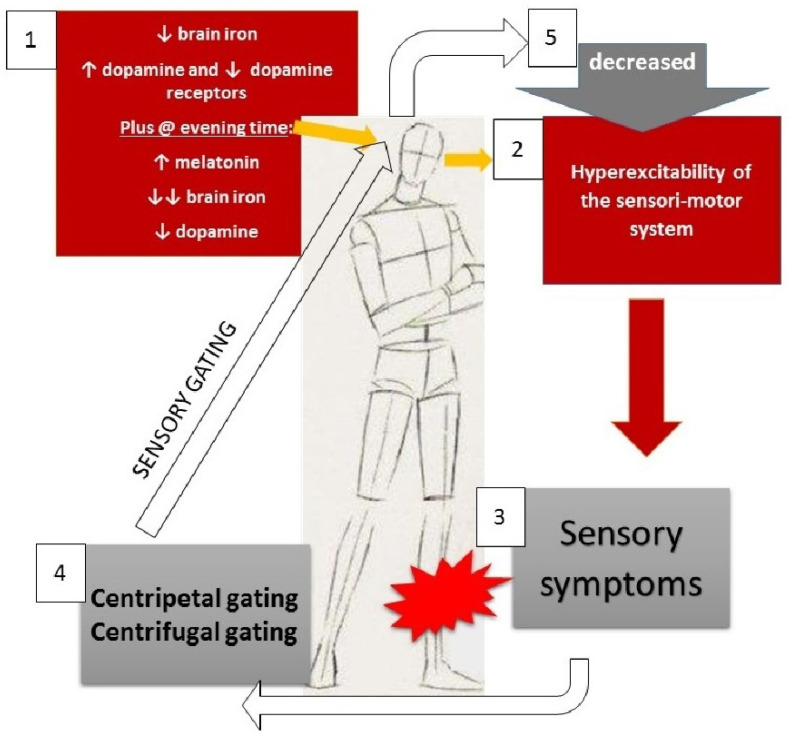
What Should You Check if You Develop Restless Legs on Ozempic
The ferritin threshold matters. The 2025 American Academy of Sleep Medicine guideline (Winkelman et al., 2025) uses a two-tier iron threshold for restless legs: ferritin at or below 75 micrograms per liter or transferrin saturation below 20% for oral or IV iron, with IV iron recommended when ferritin is between 75 and 100 micrograms per liter. Both thresholds are higher than the typical general-practice cutoff of below 30 micrograms per liter. A person can fall in the 30-75 range where standard screening may not flag iron deficiency, while RLS-specific guidelines still consider iron repletion. This discrepancy means that iron deficiency contributing to restless legs may be missed unless the sleep-medicine-specific thresholds are applied.
B12 and homocysteine. Serum B12 is reasonable to discuss given the independent association with restless legs severity (Geng et al., 2022) and the reduced dietary intake context of semaglutide therapy. Homocysteine may be elevated with B12 deficiency, serving as a functional marker that captures B12 insufficiency even when serum B12 levels appear borderline.
Iron formulation options. Short et al. (2024) conducted the first randomized double-blind trial comparing intravenous iron (ferumoxytol) versus oral iron (ferrous sulfate) for restless legs in people with comorbid iron deficiency anemia. Both routes produced improvement with no difference between groups, suggesting oral iron may be adequate in some people who can tolerate and absorb it. The 2025 meta-analysis (Mahmoud et al., 2025) highlighted ferric carboxymaltose as a particularly effective intravenous option when oral iron is insufficient.
If iron does not resolve restless legs. The 2025 American Academy of Sleep Medicine guideline gives strong recommendations for gabapentin, pregabalin, and gabapentin enacarbil in adults with RLS, while recommending against standard chronic use of several dopamine agonists because of augmentation risk. If iron repletion alone does not resolve restless legs during GLP-1 therapy, those guideline-supported medication options are part of the physician discussion.
Timing matters. Restless legs developing within weeks of starting or up-titrating semaglutide is a reasonable time to ask about ferritin, transferrin saturation, and B12. Restless legs present before GLP-1 therapy that worsen during use may indicate a lower threshold for a pre-existing vulnerability through the nutrient-intake and dopamine-modulation pathways described above.
Restless legs could be one of several ways metabolic changes during GLP-1 therapy can fragment sleep. Semaglutide also affects orexin-driven arousal, blood sugar regulation, and muscle mass – each of which may overlap with or compound the sensorimotor disruption of restless legs. Sleep disruption from metabolic causes often involves multiple simultaneous pathways.
Find out which contributors might be driving your 3am wakeups ->
Frequently Asked Questions
Does Ozempic Make Existing Restless Legs Syndrome Worse
People with a history of restless legs can ask about tracking ferritin and B12 during GLP-1 use. The 2025 American Academy of Sleep Medicine guideline’s ferritin threshold of at or below 75 micrograms per liter (for oral or IV iron) is the relevant cutoff – not the general-practice range of below 30 (Winkelman et al., 2025). A person whose ferritin was 60 before starting semaglutide might drop farther into the RLS-guideline treatment range through reduced dietary intake without triggering a standard lab flag.
Can Magnesium Supplements Help With Restless Legs on Ozempic
Iron and B12 status have a stronger rationale to discuss first. The Cochrane meta-analysis (Trotti & Becker, 2019) and the 2025 meta-analysis (Mahmoud et al., 2025) both show benefit from iron supplementation. No comparable evidence base exists for magnesium. It is a less-supported secondary discussion point after ferritin, transferrin saturation, and B12 have been considered.
Does Weight Loss From Ozempic Eventually Improve Restless Legs
No study has tracked restless legs outcomes across the full arc of GLP-1-mediated weight loss, nutrient status, and stabilization. The obesity-restless legs odds ratio of 1.44 (Lin et al., 2018) supports a hypothesis: reaching a lower body weight may reduce one obesity-associated contributor to restless legs risk. The transition period – during which rapid weight loss can lower intake of iron and B12 – is the vulnerable window. Managing nutrient status through this transition may reduce one preventable contributor to worsened restless legs.
Are Periodic Limb Movements During Sleep Related to Ozempic
Restless legs is a sensory-motor urge to move, experienced during wakefulness. Periodic limb movements are involuntary rhythmic movements during sleep that may cause arousals without the person being fully aware. If restless legs develop on semaglutide, polysomnography may reveal concurrent periodic limb movements contributing to sleep fragmentation. The 2025 guideline (Winkelman et al., 2025) distinguishes asymptomatic periodic limb movements from those with measurable consequences.
Does Mounjaro Cause the Same Restless Legs Risk as Ozempic
The same nutrient intake concerns – lower iron and B12 intake from appetite suppression or gastrointestinal side effects – may apply to both drugs. Badulescu et al. (2024) documented that GLP-1 receptor agonists as a class may modulate mesolimbic dopamine and reward circuits. Whether tirzepatide’s additional GIP receptor agonism adds further modulation of restless-legs-relevant dopaminergic circuits is unknown. The same discussion about ferritin, transferrin saturation, and B12 may be relevant regardless of which GLP-1-based drug is being used.
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Allen, R. (2004). Dopamine and iron in the pathophysiology of restless legs syndrome (RLS). Sleep Medicine, 5(4), 385-391. https://pubmed.ncbi.nlm.nih.gov/15222997/
- Anguizola, D., Garcia Aragón, A., Moncada, B., & Garcia-Borreguero, D. (2025). Restless Legs Syndrome: Neurochemistry and Therapy. Seminars in Neurology, 45(3), 368-382. https://pubmed.ncbi.nlm.nih.gov/40068886/
- Antelmi, E., Rocchi, L., Latorre, A., Belvisi, D., Magrinelli, F., Bhatia, K. P., & Tinazzi, M. (2022). Restless Legs Syndrome: Known Knowns and Known Unknowns. Brain Sciences, 12(1), 118. https://pubmed.ncbi.nlm.nih.gov/35053861/
- Badulescu, S., Tabassum, A., Le, G. H., Wong, S., Phan, L., Gill, H., Llach, C. D., McIntyre, R. S., Rosenblat, J., & Mansur, R. (2024). Glucagon-like peptide 1 agonist and effects on reward behaviour: A systematic review. Physiology & Behavior, 283, 114622. https://pubmed.ncbi.nlm.nih.gov/38945189/
- Geng, C., Yang, Z., Xu, P., & Zhang, H. (2022). Possible association between vitamin B12 deficiency and restless legs syndrome. Clinical Neurology and Neurosurgery, 223, 107477. https://pubmed.ncbi.nlm.nih.gov/36401952/
- Kamel-Abusalha, L., Afifi, A. M., Dawoud, A., Hayath, Z., Bouso, M. F., Aguillon, A., & Assaly, R. (2026). Association of GLP-1 receptor agonist use with hypersomnolence: A real-world cohort analysis. Journal of Diabetes and Metabolic Disorders, 25(1), 117. https://pubmed.ncbi.nlm.nih.gov/41867417/
- Khan, F. H., Ahlberg, C. D., Chow, C. A., Shah, D. R., & Koo, B. B. (2017). Iron, dopamine, genetics, and hormones in the pathophysiology of restless legs syndrome. Journal of Neurology, 264(8), 1634-1641. https://pubmed.ncbi.nlm.nih.gov/28236139/
- Lin, S., Zhang, H., Gao, T., Zhong, F., Sun, Y., Cai, J., & Ma, A. (2018). The association between obesity and restless legs syndrome: A systemic review and meta-analysis of observational studies. Journal of Affective Disorders, 235, 384-391. https://pubmed.ncbi.nlm.nih.gov/29674254/
- Liu, C., Liu, W. H., Yang, W., Chen, L., Xue, Y., & Chen, X. Y. (2024). GLP-1 modulated the firing activity of nigral dopaminergic neurons in both normal and parkinsonian mice. Neuropharmacology, 252, 109946. https://pubmed.ncbi.nlm.nih.gov/38599494/
- Mahmoud, A., Salamah, H. M., Alshaker, H., Ashraf Salah Ahmed, A., Abdelraouf, M. R., Suilik, H. A., Abuelazm, M., & Abdelazeem, B. (2025). Efficacy and safety of iron supplements for restless leg syndrome, a systematic review, meta-analysis, meta-regression, and trial sequential analysis of randomized controlled trials. Proceedings (Baylor University. Medical Center), 38(5), 732-746. https://pubmed.ncbi.nlm.nih.gov/40821477/
- Marshall, N. S., Serinel, Y., Killick, R., Child, J. M., Raisin, I., Berry, C. M., Lallukka, T., Wassing, R., Lee, R. W., Ratnavadivel, R., Vedam, H., Grunstein, R., Wong, K. K., Hoyos, C. M., Cayanan, E. A., Comas, M., Chapman, J. L., & Yee, B. J. (2019). Magnesium supplementation for the treatment of restless legs syndrome and periodic limb movement disorder: A systematic review. Sleep Medicine Reviews, 48, 101218. https://pubmed.ncbi.nlm.nih.gov/31678660/
- Moreira Alves, G. A., Teranishi, M., Ortega, A. C. T. C. G., James, F., & Perera Molligoda Arachchige, A. S. (2025). Mechanisms of GLP-1 in Modulating Craving and Addiction: Neurobiological and Translational Insights. Medical Sciences, 13(3), 136. https://pubmed.ncbi.nlm.nih.gov/40843757/
- Short, V., Allen, R., Earley, C. J., Bahrain, H., Rineer, S., Kashi, K., Gerb, J., & Auerbach, M. (2024). A randomized double-blind pilot study to evaluate the efficacy, safety, and tolerability of intravenous iron versus oral iron for the treatment of restless legs syndrome in patients with iron deficiency anemia. American Journal of Hematology, 99(6), 1077-1083. https://pubmed.ncbi.nlm.nih.gov/38476079/
- Trotti, L. M., & Becker, L. A. (2019). Iron for the treatment of restless legs syndrome. The Cochrane Database of Systematic Reviews, 1(1), CD007834. https://pubmed.ncbi.nlm.nih.gov/30609006/
- U.S. Food and Drug Administration. (2017). FDA Adverse Event Reporting System (FAERS) Database. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-adverse-event-reporting-system-faers-database
- U.S. Food and Drug Administration. (2026). openFDA drug/event API query: semaglutide and restless legs syndrome. https://api.fda.gov/drug/event.jsonsearch=patient.drug.openfda.generic_name:%22SEMAGLUTIDE%22+AND+patient.reaction.reactionmeddrapt:%22Restless%20legs%20syndrome%22&limit=1
- U.S. Food and Drug Administration. (2026). openFDA drug/event API query: Ozempic and restless legs syndrome. https://api.fda.gov/drug/event.jsonsearch=patient.drug.medicinalproduct:%22OZEMPIC%22+AND+patient.reaction.reactionmeddrapt:%22Restless%20legs%20syndrome%22&limit=1
- U.S. Food and Drug Administration. (2026). WEGOVY prescribing information via openFDA label API. https://api.fda.gov/drug/label.jsonsearch=openfda.brand_name:%22WEGOVY%22&limit=1
- U.S. Food and Drug Administration. (2026). MOUNJARO prescribing information via openFDA label API. https://api.fda.gov/drug/label.jsonsearch=openfda.brand_name:%22MOUNJARO%22&limit=1
- Winkelman, J. W., Berkowski, J. A., DelRosso, L. M., Koo, B. B., Scharf, M. T., Sharon, D., Zak, R. S., Kazmi, U., Falck-Ytter, Y., Shelgikar, A. V., Trotti, L. M., & Walters, A. S. (2025). Treatment of restless legs syndrome and periodic limb movement disorder: an American Academy of Sleep Medicine clinical practice guideline. Journal of Clinical Sleep Medicine, 21(1), 137-152. https://pubmed.ncbi.nlm.nih.gov/39324694/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 20 references cited
